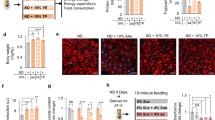
Abstract
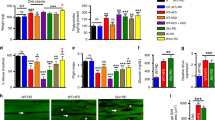
Aging entails the progressive decline in the body’s self-regulation and functionality over time. Notably, obesity and aging exhibit parallel phenotypes, with obesity further accelerating the aging process across multiple dimensions and diminishing lifespan. In this study, we explored the impact of trans fatty acid (TFA) consumption on the overall health and lifespan of male Drosophila melanogaster under an isocaloric high-sugar and high-fat diet. Our results indicate that TFA intake results in a shortened lifespan, elevated body weight, and increased triglyceride levels in flies fed a high-sugar and high-fat diet with equivalent caloric intake. Additionally, TFA exposure induces oxidative stress, locomotor deficits, and damage to the intestinal barrier in flies. Collectively, chronic TFA consumption expedites the aging process and reduces the lifespan of male Drosophila melanogaster. These results contribute supplementary evidence regarding the adverse health effects associated with TFAs.
Graphical Abstract
Trans fatty acid intake results in a shortened lifespan, elevated body weight, and increased triglyceride levels in male Drosophila melanogaster fed a high-sugar and high-fat diet with equivalent caloric intake. Additionally, Trans fatty acid exposure induces oxidative stress, locomotor deficits, and damage to the intestinal barrier in flies.







Similar content being viewed by others
References
Adams MD, Celniker SE, Holt RA, Evans CA, Gocayne JD, Amanatides PG, Scherer SE, Li PW, Hoskins RA, Galle RF, George RA, Lewis SE, Richards S, Ashburner M, Henderson SN et al (2000) The genome sequence of Drosophila melanogaster. Science 287(5461):2185–2195. https://doi.org/10.1126/science.287.5461.2185
Ali Abd El-Aal Y, Mohamed Abdel-Fattah D, El-Dawy Ahmed K (2019) Some biochemical studies on trans fatty acid-containing diet. Diabetes Metab Syndr 13(3):1753–1757. https://doi.org/10.1016/j.dsx.2019.03.029
Baenas N, Wagner AE (2022) Drosophila melanogaster as a model organism for obesity and type-2 diabetes mellitus by applying high-sugar and high-fat diets. Biomolecules 12(2):307. https://doi.org/10.3390/biom12020307
Birse RT, Choi J, Reardon K, Rodriguez J, Graham S, Diop S, Ocorr K, Bodmer R, Oldham S (2010) High-fat-diet-induced obesity and heart dysfunction are regulated by the TOR pathway in Drosophila. Cell Metab 12(5):533–544. https://doi.org/10.1016/j.cmet.2010.09.014
Bjorksten J (1968) The crosslinkage theory of aging. J Am Geriatr Soc 16(4):408–427. https://doi.org/10.1111/j.1532-5415.1968.tb02821.x
Buchon N, Osman D, David FP, Fang HY, Boquete JP, Deplancke B, Lemaitre B (2013) Morphological and molecular characterization of adult midgut compartmentalization in Drosophila. Cell Rep 3(5):1725–1738. https://doi.org/10.1016/j.celrep.2013.04.001
Cui X, Wang L, Zuo P, Han Z, Fang Z, Li W, Liu J (2004) D-galactose-caused life shortening in Drosophila melanogaster and Musca domestica is associated with oxidative stress. Biogerontology 5(5):317–325. https://doi.org/10.1007/s10522-004-2570-3
Dambroise E, Monnier L, Ruisheng L, Aguilaniu H, Joly JS, Tricoire H, Rera M (2016) Two phases of aging separated by the Smurf transition as a public path to death. Sci Rep 6:23523. https://doi.org/10.1038/srep23523
de Souza RJ, Mente A, Maroleanu A, Cozma AI, Ha V, Kishibe T, Uleryk E, Budylowski P, Schunemann H, Beyene J, Anand SS (2015) Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease, and type 2 diabetes: systematic review and meta-analysis of observational studies. BMJ 351:h3978. https://doi.org/10.1136/bmj.h3978
Demontis F, Piccirillo R, Goldberg AL, Perrimon N (2013) The influence of skeletal muscle on systemic aging and lifespan. Aging Cell 12(6):943–949. https://doi.org/10.1111/acel.12126
Dhibi M, Brahmi F, Mnari A, Houas Z, Chargui I, Bchir L, Gazzah N, Alsaif MA, Hammami M (2011) The intake of high fat diet with different trans fatty acid levels differentially induces oxidative stress and non alcoholic fatty liver disease (NAFLD) in rats. Nutr Metab (lond) 8(1):65. https://doi.org/10.1186/1743-7075-8-65
Effros RB (2005) Roy Walford and the immunologic theory of aging. Immun Ageing 2(1):7. https://doi.org/10.1186/1742-4933-2-7
Estadella D, da Penha Oller do Nascimento CM, Oyama LM, Ribeiro EB, Damaso AR, de Piano A (2013) Lipotoxicity: effects of dietary saturated and transfatty acids. Mediat Inflamm 2013:137579. https://doi.org/10.1155/2013/137579
Feany MB, Bender WW (2000) A Drosophila model of Parkinson’s disease. Nature 404(6776):394–398. https://doi.org/10.1038/35006074
Fernando R, Drescher C, Nowotny K, Grune T, Castro JP (2019) Impaired proteostasis during skeletal muscle aging. Free Radic Biol Med 132:58–66. https://doi.org/10.1016/j.freeradbiomed.2018.08.037
Gargano JW, Martin I, Bhandari P, Grotewiel MS (2005) Rapid iterative negative geotaxis (RING): a new method for assessing age-related locomotor decline in Drosophila. Exp Gerontol 40(5):386–395. https://doi.org/10.1016/j.exger.2005.02.005
Halim M, Halim A (2019) The effects of inflammation, aging and oxidative stress on the pathogenesis of diabetes mellitus (type 2 diabetes). Diabetes Metab Syndr 13(2):1165–1172. https://doi.org/10.1016/j.dsx.2019.01.040
Harman D (1993) Free radical involvement in aging pathophysiology and therapeutic implications. Drugs Aging 3(1):60–80. https://doi.org/10.2165/00002512-199303010-00006
Kruk PA, Rampino NJ, Bohr VA (1995) DNA damage and repair in telomeres: relation to aging. Proc Natl Acad Sci USA 92(1):258–262. https://doi.org/10.1073/pnas.92.1.258
Lennicke C, Rahn J, Heimer N, Lichtenfels R, Wessjohann LA, Seliger B (2016) Redox proteomics: methods for the identification and enrichment of redox-modified proteins and their applications. Proteomics 16(2):197–213. https://doi.org/10.1002/pmic.201500268
Li YM, Chan HY, Yao XQ, Huang Y, Chen ZY (2008) Green tea catechins and broccoli reduce fat-induced mortality in Drosophila melanogaster. J Nutr Biochem 19(6):376–383. https://doi.org/10.1016/j.jnutbio.2007.05.009
Liao S, Amcoff M, Nässel DR (2021) Impact of high-fat diet on lifespan, metabolism, fecundity and behavioral senescence in Drosophila. Insect Biochem Mol Biol 133:103495. https://doi.org/10.1016/j.ibmb.2020.103495
Liguori I, Russo G, Curcio F, Bulli G, Aran L, Della-Morte D, Gargiulo G, Testa G, Cacciatore F, Bonaduce D, Abete P (2018) Oxidative stress, aging, and diseases. Clin Interv Aging 13:757–772. https://doi.org/10.2147/CIA.S158513
Martins RR, McCracken AW, Simons MJP, Henriques CM, Rera M (2018) How to catch a Smurf? - ageing and beyond… In vivo assessment of intestinal permeability in multiple model organisms. Bio Protoc 8(3):e2722. https://doi.org/10.21769/BioProtoc.2722
Mozaffarian D, Katan MB, Ascherio A, Stampfer MJ, Willett WC (2006) Trans fatty acids and cardiovascular disease. N Engl J Med 354(15):1601–1613. https://doi.org/10.1056/NEJMra054035
Musselman LP, Fink JL, Narzinski K, Ramachandran PV, Hathiramani SS, Cagan RL, Baranski TJ (2011) A high-sugar diet produces obesity and insulin resistance in wild-type Drosophila. Dis Model Mech 4(6):842–849. https://doi.org/10.1242/dmm.007948
Ogienko AA, Omelina ES, Bylino OV, Batin MA, Georgiev PG, Pindyurin AV (2022) Drosophila as a model organism to study basic mechanisms of longevity. Int J Mol Sci 23(19):11244. https://doi.org/10.3390/ijms231911244
Pipoyan D, Stepanyan S, Stepanyan S, Beglaryan M, Costantini L, Molinari R, Merendino N (2021) The effect of trans fatty acids on human health: regulation and consumption patterns. Foods 10(10):2452. https://doi.org/10.3390/foods10102452
Reiter LT, Potocki L, Chien S, Gribskov M, Bier E (2001) A systematic analysis of human disease-associated gene sequences in Drosophila melanogaster. Genome Res 11(6):1114–1125. https://doi.org/10.1101/gr.169101
Rera M, Clark RI, Walker DW (2012) Intestinal barrier dysfunction links metabolic and inflammatory markers of aging to death in Drosophila. Proc Natl Acad Sci USA 109(52):21528–21533. https://doi.org/10.1073/pnas.1215849110
Rubin GM, Yandell MD, Wortman JR, Gabor Miklos GL, Nelson CR, Hariharan IK, Fortini ME, Li PW, Apweiler R, Fleischmann W, Cherry JM, Henikoff S, Skupski MP, Misra S, Ashburner M et al (2000) Comparative genomics of the Eukaryotes. Science 287(5461):2204–2215. https://doi.org/10.1126/science.287.5461.2204
Saheera S, Krishnamurthy P (2020) Cardiovascular changes associated with hypertensive heart disease and aging. Cell Transpl 29:963689720920830. https://doi.org/10.1177/0963689720920830
Salazar AM, Aparicio R, Clark RI, Rera M, Walker DW (2023) Intestinal barrier dysfunction: an evolutionarily conserved hallmark of aging. Dis Model Mech 16(4):dmm049969. https://doi.org/10.1242/dmm.049969
Santos AL, Sinha S (2021) Obesity and aging: molecular mechanisms and therapeutic approaches. Ageing Res Rev 67:101268. https://doi.org/10.1016/j.arr.2021.101268
Sauvat A, Chen G, Müller K, Tong M, Aprahamian F, Durand S, Cerrato G, Bezu L, Leduc M, Franz J, Rockenfeller P, Sadoshima J, Madeo F, Kepp O, Kroemer G (2018) Trans-fats inhibit autophagy induced by saturated fatty acids. EBioMedicine 30:261–272. https://doi.org/10.1016/j.ebiom.2018.03.028
Shell BC, Schmitt RE, Lee KM, Johnson JC, Chung BY, Pletcher SD, Grotewiel M (2018) Measurement of solid food intake in Drosophila via consumption-excretion of a dye tracer. Sci Rep 8(1):11536. https://doi.org/10.1038/s41598-018-29813-9
Tam BT, Morais JA, Santosa S (2020) Obesity and ageing: two sides of the same coin. Obes Rev 21(4):e12991. https://doi.org/10.1111/obr.12991
Taormina G, Ferrante F, Vieni S, Grassi N, Russo A, Mirisola MG (2019) Longevity: lesson from model organisms. Genes (basel) 10(7):518. https://doi.org/10.3390/genes10070518
Trindade de Paula M, Poetini Silva MR, Machado Araujo S, Cardoso Bortolotto V, Barreto Meichtry L, Zemolin AP, Wallau GL, Jesse CR, Franco JL, Posser T, Prigol M (2016) High-fat diet induces oxidative stress and MPK2 and HSP83 gene expression in Drosophila melanogaster. Oxid Med Cell Longev 2016:4018157. https://doi.org/10.1155/2016/4018157
Wang HL, Sun ZO, Rehman RU, Wang H, Wang YF, Wang H (2017) Rosemary extract-mediated lifespan extension and attenuated oxidative damage in Drosophila melanogaster fed on high-fat diet. J Food Sci 82(4):1006–1011. https://doi.org/10.1111/1750-3841.13656
WHO (2020) WHO methods and data sources for country-level causes of death 2000–2019. World Health Organization. https://iris.who.int/handle/10665/374896
WHO (2023) WHO Book Saturated fatty acid and trans-fatty acid intake for adults and children: WHO guideline. World Health Organization. https://iris.who.int/handle/10665/370419
Zhang Y, Fischer KE, Soto V, Liu Y, Sosnowska D, Richardson A, Salmon AB (2015) Obesity-induced oxidative stress, accelerated functional decline with age and increased mortality in mice. Arch Biochem Biophys 576:39–48. https://doi.org/10.1016/j.abb.2014.12.018
Funding
This work was funded by Basic and Applied Basic Research Foundation of Guangdong Province (Grant no.: 2021B1515140057), National Natural Science Foundation of China (Grant no.: 82273622), BYHEALTH Nutrition and Health Research Foundation (Grant no.: TY202101001), Undergraduate Training Program for Innovation and Entrepreneurship (Grant no.: GDMU2023331), Discipline Construction Project of Guangdong Medical University (Grant no.: 4SG21016G).
Author information
Authors and Affiliations
Contributions
Qianhua Yuan: investigation, data curation, writing—original draft. Mengliu Luo: validation, writing—review and editing. Yutong Xie: investigation, visualization. Wanhan Song: formal analysis. Ya Wang: data curation, validation. Dazhang Deng: writing—review and editing. Shuyan Chen: investigation, writing—review and editing. Honghui Guo: resources, project administration, supervision. All authors have read and approved the submitted version of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yuan, Q., Luo, M., Xie, Y. et al. Chronic trans fatty acid consumption shortens lifespan in male Drosophila melanogaster on a high-sugar and high-fat diet. Biogerontology (2024). https://doi.org/10.1007/s10522-024-10101-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10522-024-10101-1