Abstract
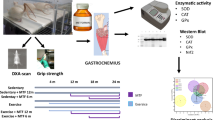
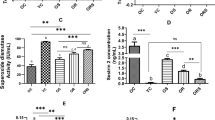
Here we investigate metabolic changes, the antioxidant system and the accumulation of oxidative damage in muscles with different fiber types during the aging process in Wistar rats and try to map how sooner the changes occur. To do so, 30 male Wistar rats were submitted to behavioral evaluation to determine voluntary strength in the 11, 15, and 19 month old rats, measuring the energy metabolism, antioxidant system, oxidative damage and structure in the soleus and extensor digitorum longus muscles. We detected structural and metabolic changes in both muscles, especially in the EDL of 15 month old rats and in the soleus of 19 month old rats. In the 15 month old rats, there was a reduction in the cross-sectional area of the fibers, and a reduction in the proportion of type I fibers, accompanied by an increase in fiber density and the amount of type IIA fibers. This change in the fiber profile was followed by an increase in the activity of anaerobic metabolism enzymes, suggesting a reduction in the oxidative capacity of the muscle. In addition, there was an increase in the rate of lipid peroxidation, accompanied by a reduced antioxidant capacity. In the 19 month old rats, these disturbances got stronger. In summary, the present study demonstrated that before functional disturbances, there was an accumulation of oxidative damage and structural changes in the skeletal muscle beginning at 15 months old in the EDL and the soleus only in the biochemical parameters. Therefore, the metabolic alterations occurred at 15 months old and not before.



Similar content being viewed by others
References
Abdel-Mageid AD, Abou-Salem MES, Salaam NMHA, El-Garhy HAS (2018) The potential effect of garlic extract and curcumin nanoparticles against complication accompanied with experimentally induced diabetes in rats. Phytomedicine 43:126–134. https://doi.org/10.1016/J.PHYMED.2018.04.039
Aebi H (1985) Catalase in vitro. Methods Enzimol 105:121–126
Ahn B, Ranjit R, Premkumar P et al (2019) Mitochondrial oxidative stress impairs contractile function but paradoxically increases muscle mass via fibre branching. J Cachexia Sarcopenia Muscle 10:411–428. https://doi.org/10.1002/jcsm.12375
Alway SE, Mohamed JS, Myers MJ (2017) Mitochondria initiate and regulate Sarcopenia. Exerc Sport Sci Rev 45:58
Baumann CW, Kwak D, Liu HM, Thompson LV (2016) Age-induced oxidative stress: how does it influence skeletal muscle quantity and quality? J Appl Physiol 121:1047
Blaauw B, Schiaffino S, Reggiani C (2013) Mechanisms modulating skeletal muscle phenotype. Compr Physiol 3:1645–1687. https://doi.org/10.1002/cphy.c130009
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Braggion GF, Ornelas E, Cury JCS et al (2015) Morphological and biochemical effects on the skeletal muscle of Ovariectomized old female rats submitted to the intake of diets with vegetable or animal protein and resistance training. Oxid Med Cell Longev 2016:10. https://doi.org/10.1155/2016/9251064
Bratic A, Larsson NG (2013) The role of mitochondria in aging. J Clin Invest 123:951
Brunetta HS, Holwerda AM, van Loon LJC, Holloway GP (2020) Mitochondrial ROS and aging: understanding exercise as a preventive tool. J Sci Sport Exerc 2:15
Cacciatore F, Della-Morte D, Basile C et al (2015) Butyryl-cholinesterase is related to muscle mass and strength. A new biomarker to identify elderly subjects at risk of Sarcopenia. Biomark Med 9:669. https://doi.org/10.2217/bmm.15.28
Calcinotto A, Kohli J, Zagato E et al (2019) Cellular senescence: aging, cancer, and injury. Physiol Rev 99:1047. https://doi.org/10.1152/physrev.00020.2018
Carlberg I, Mannervik B (1985) [59] Glutathione reductase. Methods Enzymol. https://doi.org/10.1016/S0076-6879(85)13062-4
Carter CS, Justice JN, Thompson LD (2019) Lipotoxicity, aging, and muscle contractility: does fiber type matter? GeroScience 41:297–308. https://doi.org/10.1007/s11357-019-00077-z
Correa-de-Araujo R, Harris-Love MO, Miljkovic I et al (2017) The need for standardized assessment of muscle quality in skeletal muscle function deficit and other aging-related muscle dysfunctions: a symposium report. Front Physiol 8:87
Crouch RK, Gandy SE, Kimsey G et al (1981) The inhibition of islet superoxide dismutase by diabetogenic drugs. Diabetes 30:235–241. https://doi.org/10.2337/diab.30.3.235
Cutler AA, Jackson JB, Corbett AH, Pavlath GK (2018) Non-equivalence of nuclear import among nuclei in multinucleated skeletal muscle cells. J Cell Sci 131:1–14. https://doi.org/10.1242/jcs.207670
Deschenes MR, Roby MA, Glass EK (2011) Aging influences adaptations of the neuromuscular junction to endurance training. Neuroscience 190:56. https://doi.org/10.1016/j.neuroscience.2011.05.070
Deus APL, Bassi D, Simes RP et al (2012) MMP 2 expression in skeletal muscle after strength training. Int J Sports Med. https://doi.org/10.1055/s-0031-1291224
Drew B, Dirks PA, Selman C et al (2003) Effects of aging and caloric restriction on mitochondrial energy production in gastrocnemius muscle and heart. Am J Physiol Regul Integr Comp Physiol. https://doi.org/10.1152/ajpregu.00455.2002
Dubowitz V, Brooke M (1973) Muscle biopsy: a mordern approach, 1st edn. Saunders College Publishing, London
Dungan CM, Peck BD, Walton RG et al (2020) In vivo analysis of γH2AX + cells in skeletal muscle from aged and obese humans. FASEB J 34:7018–7035. https://doi.org/10.1096/fj.202000111RR
Edwards MH, Dennison EM, Aihie Sayer A et al (2015) Osteoporosis and sarcopenia in older age. Bone 80:126–130. https://doi.org/10.1016/j.bone.2015.04.016
Ellman GL, Courtney KD, Adres-Jr V, Featherstone RM (1961) A new and rapid colorimetric of acetylcholinesterase determination. Biochem Pharmacol 7:88–95
Finkel T, Holbrook NJ (2000) Oxidants, oxidative stress and the biology of ageing. Nature 408:239
Frontera WR (2017) Physiologic changes of the musculoskeletal system with aging: a brief review. Phys Med Rehabil Clin N Am 28:705
Frontera WR, Rodriguez Zayas A, Rodriguez N (2012) Aging of human muscle: understanding Sarcopenia at the single muscle cell level. Phys Med Rehabil Clin N Am 23:201
Hanson MA, Gluckman PD (2014) Early developmental conditioning of later health and disease: physiology or pathophysiology? Physiol Rev 94:1027
Hunter SK, Pereira XHM, Keenan KG (2016) The aging neuromuscular system and motor performance. J Appl Physiol. https://doi.org/10.1152/japplphysiol.00475.2016
Jiang ZY, Woollard ACS, Wolff SP (1991) Lipid hydroperoxides measurement by oxidation of Fe2+ in the presence of xylenol orange. Comparison with the TBA assay and an iodometric method. Lipids 26:853–856
Junqueira LC, Junqueira LM (1983) Técnicas Básicas de Histologia e Citologia, 1st edn. Editora Santos, São Paulo
Keen JH, Habig WH, Jakoby WB (1976) Mechanism for several activities of the gluthathione S-transferases. J Biol Chem 251:6138–6188
Khosa S, Trikamji B, Khosa GS et al (2019) An overview of neuromuscular junction aging findings in human and animal studies. Curr Aging Sci 12:28. https://doi.org/10.2174/1874609812666190603165746
Lai N, Fealy CE, Kummitha CM et al (2020) Mitochondrial utilization of competing fuels is altered in insulin resistant skeletal muscle of non-obese rats (Goto-Kakizaki). Front Physiol 11:1–14. https://doi.org/10.3389/fphys.2020.00677
Larsson L, Degens H, Li M et al (2019) Sarcopenia: aging-related loss of muscle mass and function. Physiol Rev 99:427. https://doi.org/10.1152/physrev.00061.2017
Liu S, Yang D, Yu L et al (2021) Effects of lycopene on skeletal muscle-fiber type and high-fat diet-induced oxidative stress. J Nutr Biochem 87:108523. https://doi.org/10.1016/j.jnutbio.2020.108523
López-Otín C, Blasco MA, Partridge L et al (2013) The hallmarks of aging Europe PMC funders group. Cell. https://doi.org/10.1016/j.cell.2013.05.039
Martins HRF, Zotz TGG, Messa SP et al (2020) Morphometric and molecular muscle remodeling after passive stretching in Elderly female rats. Clinics. https://doi.org/10.6061/clinics/2020/e1769
McArdle A, Jackson MJ (2000) Exercise, oxidative stress and ageing. J Anat 197:539
Niccoli T, Partridge L (2012) Ageing as a risk factor for disease. Curr Biol 22:R741
Niki E (2016) Oxidative stress and antioxidants: distress or eustress? Arch Biochem Biophys 595:19–24. https://doi.org/10.1016/J.ABB.2015.11.017
Nilwik R, Snijders T, Leenders M et al (2013) The decline in skeletal muscle mass with aging is mainly attributed to a reduction in type II muscle fiber size. Exp Gerontol 48:492–498. https://doi.org/10.1016/j.exger.2013.02.012
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Reid MB, Khawli FA, Moody MR (1993) Reactive oxygen in skeletal muscle. III. Contractility of unfatigued muscle. J Appl Physiol 75:1081. https://doi.org/10.1152/jappl.1993.75.3.1081
Rodacki CLN, Rodacki ALF, Pereira G et al (2012) Fish-oil supplementation enhances the effects of strength training in elderly women. Am J Clin Nutr. https://doi.org/10.3945/ajcn.111.021915
Romeu Montenegro K, Amarante Pufal M, Newsholme P (2021) Vitamin D supplementation and impact on skeletal muscle function in cell and animal models and an aging population: what do we know so far? Nutrients 13:1110
Ryan MJ, Jackson JR, Hao Y et al (2010) Suppression of oxidative stress by resveratrol after isometric contractions in gastrocnemius muscles of aged mice. J Gerontol Ser A Biol Sci Med Sci. https://doi.org/10.1093/gerona/glq080
Santos AL, Sinha S, Lindner AB (2018) The good, the bad, and the ugly of ROS: new insights on aging and aging-related diseases from eukaryotic and prokaryotic model organisms. Oxid Med Cell Longev 2018:1941285
Sastre J, Pallardó FV, Viña J (2003) The role of mitochondrial oxidative stress in aging. Free Radic Biol Med. https://doi.org/10.1016/S0891-5849(03)00184-9
Schiaffino S, Reggiani C (2011) Fiber types in mammalian skeletal muscles. Physiol Rev 91:1447–1531. https://doi.org/10.1152/physrev.00031.2010
Schiaffino S, Reggiani C, Murgia M (2019) Fiber type diversity in skeletal muscle explored by mass spectrometry-based single fiber proteomics. Histol Histopathol 18:170
Schoffen JPF, Santi Rampazzo AP, Cirilo CP et al (2014) Food restriction enhances oxidative status in aging rats with neuroprotective effects on myenteric neuron populations in the proximal colon. Exp Gerontol 51:54. https://doi.org/10.1016/j.exger.2014.01.001
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205. https://doi.org/10.1016/0003-2697(68)90092-4
Shahidi B, Shah SB, Esparza M et al (2018) Skeletal muscle atrophy and degeneration in a mouse model of traumatic brain Injury. J Neurotrauma 35:398–401. https://doi.org/10.1089/neu.2017.5172
Shields HJ, Traa A, Van Raamsdonk JM (2021) Beneficial and detrimental effects of reactive oxygen species on lifespan: a comprehensive review of comparative and experimental studies. Front Cell Dev Biol. https://doi.org/10.3389/fcell.2021.628157. (Front Cell Dev Biol 9)
Sullivan-Gunn MJ, Lewandowski PA (2013) Elevated hydrogen peroxide and decreased catalase and glutathione peroxidase protection are associated with aging Sarcopenia. BMC Geriatr 13:1–9. https://doi.org/10.1186/1471-2318-13-104
UeA W, Hussain F, Kayani HUR (2020) Aging—oxidative stress, antioxidants and computational modeling. Heliyon 6:1–11
Vasilaki A, Mansouri A, Van Remmen H et al (2006) Free radical generation by skeletal muscle of adult and old mice: effect of contractile activity. Aging Cell 5:109. https://doi.org/10.1111/j.1474-9726.2006.00198.x
Wagner KH, Cameron-Smith D, Wessner B, Franzke B (2016) Biomarkers of aging: from function to molecular biology. Nutrients 8:338
Wendell A (1981) Glutathione peroxidase. Methods in enzimology. Elsevier, Amsterdam, pp 325–333
World Health Organization (2018) WHO | life expectancy. WHO
Wosczyna MN, Rando TA (2018) A muscle stem cell support group: coordinated cellular responses in muscle regeneration. Dev Cell 46:135–143. https://doi.org/10.1016/j.devcel.2018.06.018
Zhao SJ, Liu XJ, Tian JS et al (2020) Effects of Guilingji on aging rats and its underlying mechanisms. Rejuvenation Res 23:138. https://doi.org/10.1089/rej.2018.2118
Zotz TGG, Zotz R, Guimarães ATB et al (2019) The development of a mechanical device to stretch skeletal muscle of young and old rats. Clinics 74:e629. https://doi.org/10.6061/clinics/2019/e629
Acknowledgements
Authors thanks to Coordenação de Aperfeiçoamento de Pessoal de Nível Superior to Malu Cristina Araujo Montoro Lima fellowship and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) to Matheus Felipe Zazula fellowship.
Funding
This research was funded by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Author information
Authors and Affiliations
Contributions
MCAML: Conceptualization, Methodology, Formal Analysis and Investigation. MFZ: Methodology, Validation, Formal Analysis, Data Curation, Writing Original Draft and Writing—review and editing. LFM: Methodology, Visualization. SRC: Methodology, Visualization. ATBG: Funding acquisition, Formal analysis, Methodology. LCF: Conceptualization, Funding acquisition, Formal analysis, Methodology, Project administration, Validation, Visualization, Writing—original draft. KN: Conceptualization, Funding acquisition, Formal analysis, Methodology, Project administration, Supervision, Validation, Visualization, Writing—original draft, Writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lima, M.C.A.M., Zazula, M.F., Martins, L.F. et al. How soon do metabolic alterations and oxidative distress precede the reduction of muscle mass and strength in Wistar rats in aging process?. Biogerontology (2023). https://doi.org/10.1007/s10522-023-10078-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10522-023-10078-3