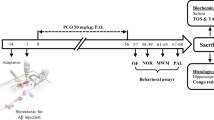
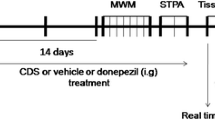
This study examined the effect of curcumin on pathological manifestations and clearance of amyloid β peptide (Aβ) in the hippocampus of 8-month-old transgenic APP/PS1 mice with inherent Alzheimer’s disease. APP/PS1 mice and the age-matched wild-type controls were subjected to 3 behavioral tests: open field, new object recognition, and Morris water maze. Expression of Aβ, APP, CTF, BACE1, IDE, NEP, and LRP1 proteins in the extracted hippocampal tissue was evaluated by Western blotting. The distribution and the quantity of amyloid plaques and the spread of microglia in the hippocampus were determined by immunofluorescence. The contents of Aβ40 and Aβ42 in the hippocampus were assayed and analyzed on Simoa HD-1 analyzer. The proteins interacting with Aβ in the hippocampus of APP/PS1 mice were detected by co-immunoprecipitation. Curcumin significantly reduced motor hyperactivity in the open-field test, improved short-term recognition memory, spatial learning, and reference memory in APP/PS1 mice. In the hippocampus of APP/PS1 mice, curcumin significantly diminished the elevated Aβ levels and inhibited microglia proliferation. At the same time, curcumin had no effect on Aβ production, extracellular enzymatic hydrolysis, and LRP1-mediated outward transport, but enhanced Aβ clearance by activation of the intracellular ubiquitin-proteasome system and related peripheral mechanisms. Thus, curcumin improves the learning and memory abilities of APP/PS1 mice and reduces the pathological accumulation of Aβ in the brain.
Similar content being viewed by others
References
Xin SH, Tan L, Cao X, Yu JT, Tan L. Clearance of Amyloid Beta and Tau in Alzheimer’s Disease: from Mechanisms to Therapy. Neurotox. Res. 2018;34(3):733-748. https://doi.org/10.1007/s12640-018-9895-1
Nalivaeva NN, Turner AJ. Targeting amyloid clearance in Alzheimer’s disease as a therapeutic strategy. Br. J. Pharmacol. 2019;176(18):3447-3463. https://doi.org/10.1111/bph.14593
Zhao Y, Wu X, Li X, Jiang LL, Gui X, Liu Y, Sun Y, Zhu B, Piña-Crespo JC, Zhang M, Zhang N, Chen X, Bu G, An Z, Huang TY, Xu H. TREM2 Is a Receptor for β-Amyloid that Mediates Microglial Function. Neuron. 2018;97(5):1023-1031.e7. https://doi.org/10.1016/j.neuron.2018.01.031
Li T, Jiao JJ, Hölscher C, Wu MN, Zhang J, Tong JQ, Dong XF, Qu XS, Cao Y, Cai HY, Su Q, Qi JS. A novel GLP-1/GIP/Gcg triagonist reduces cognitive deficits and pathology in the 3xTg mouse model of Alzheimer’s disease. Hippocampus. 2018;28(5):358-372. https://doi.org/10.1002/hipo.22837
Bar-Sela G, Epelbaum R, Schaffer M. Curcumin as an anti-cancer agent: review of the gap between basic and clinical applications. Curr. Med. Chem. 2010;17(3):190-197. https://doi.org/10.2174/092986710790149738
Yang W, Chauhan A, Mehta S, Mehta P, Gu F, Chauhan V. Trichostatin A increases the levels of plasma gelsolin and amyloid beta-protein in a transgenic mouse model of Alzheimer’s disease. Life Sci. 2014;99(1-2):31-36. https://doi.org/10.1016/j.lfs.2014.01.064
Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science. 2002;297(5580):353-6. https://doi.org/10.1126/science.1072994
Yang W, Chauhan A, Wegiel J, Kuchna I, Gu F, Chauhan V. Effect of trichostatin A on gelsolin levels, proteolysis of amyloid precursor protein, and amyloid beta-protein load in the brain of transgenic mouse model of Alzheimer’s disease. Curr. Alzheimer. Res. 2014;11(10):1002-1011. https://doi.org/10.2174/1567205011666141107125531
Van Gool B, Storck SE, Reekmans SM, Lechat B, Gordts PLSM, Pradier L, Pietrzik CU, Roebroek AJM. LRP1 Has a Predominant Role in Production over Clearance of Aβ in a Mouse Model of Alzheimer’s Disease. Mol. Neurobiol. 2019;56(10):7234-7245. https://doi.org/10.1007/s12035-019-1594-2
Lucin KM, O’Brien CE, Bieri G, Czirr E, Mosher KI, Abbey RJ, Mastroeni DF, Rogers J, Spencer B, Masliah E, Wyss-Coray T. Microglial beclin 1 regulates retromer trafficking and phagocytosis and is impaired in Alzheimer’s disease. Neuron. 2013;79(5):873-886. https://doi.org/10.1016/j.neuron.2013.06.046
Norden DM, Godbout JP. Review: microglia of the aged brain: primed to be activated and resistant to regulation. Neuropathol. Appl. Neurobiol. 2013;39(1):19-34. https://doi.org/10.1111/j.1365-2990.2012.01306.x
Mandrekar S, Jiang Q, Lee CY, Koenigsknecht-Talboo J, Holtzman DM, Landreth GE. Microglia mediate the clearance of soluble Abeta through fluid phase macropinocytosis. J. Neurosci. 2009;29(13):4252-4262. https://doi.org/10.1523/JNEUROSCI.5572-08.2009
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 175, No. 3, pp. 314-320, March, 2023
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, M.J., Zhong, J.H. & Yi, X.J. Effect of Curcumin on Cognitive Behavior and Pathological Characteristics of the Hippocampus in Mice with Inherent Alzheimer’s Disease. Bull Exp Biol Med 175, 335–340 (2023). https://doi.org/10.1007/s10517-023-05863-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-023-05863-6