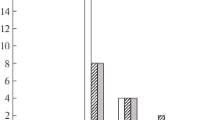
The antiviral activity of recombinant human IFN-lambda type 1 (IFNλ-1) against culture strain of SARS-CoV-2 virus was determined by infecting a highly sensitive VeroE6 coronavirus cell culture after preincubation test (the cell monolayer was incubated with 4-fold dilutions of IFNλ-1 in a concentration range of 0.16-42,500 ng/ml in a culture medium for 12 h at 37°C) and without preincubation (simultaneous addition of different concentrations of IFNλ-1 and SARS-CoV-2 infection in a dose of 102 TCID50). The created recombinant human IFNλ-1 demonstrated obvious antiviral activity against SARS-CoV-2 virus in vitro. In the tests with and without preincubation, IFNλ-1 exhibited significant activity, although somewhat lower in variant with simultaneous addition of IFNλ-1 and virus to the cell culture. It should be noted that the antiviral effect of IFNλ-1 was observed in a wide range of concentrations.
Article PDF
Similar content being viewed by others
References
Kirilyuk IA, Svyatchenko VA, Morozov DA, Kazachinskaya EI, Kiselev NN, Bakunova SM, Voinov MA, Loktev VB, Grigoryev IA. In vitro cytotoxicity of nitroxyl radicals with respect to tumor and diploid human cells and estimation of their antiviral activity. Antibiotiki Khimioterapiya. 2012;57(1-2):3-12. Russian.
Busnadiego I, Fernbach S, Pohl MO, Karakus U, Huber M, Trkola A, Stertz S, Hale BG. Antiviral activity of type I, II, and III interferons counterbalances ACE2 inducibility and restricts SARS-CoV-2. mBio. 2020;11(5):e01928-20. doi: https://doi.org/10.1128/mBio.01928-20
Dinnon KH 3rd, Leist SR, Schäfer A, Edwards CE, Martinez DR, Montgomery SA, West A, Yount BL Jr, Hou YJ, Adams LE, Gully KL, Brown AJ, Huang E, Bryant MD, Choong IC, Glenn JS, Gralinski LE, Sheahan TP, Baric RS. A mouse-adapted model of SARS-CoV-2 to test COVID-19 countermeasures. Nature. 2020;586:560-566. doi: https://doi.org/10.1038/s41586-020-2708-8
O’Brien TR, Thomas DL, Jackson SS, Prokunina-Olsson L, Donnelly RP, Hartmann R. Weak Induction of interferon expression by severe acute respiratory syndrome coronavirus 2 supports clinical trials of interferon-λ to treat early coronavirus disease 2019. Clin. Infect. Dis. 2020;71(6):1410-1412. doi: https://doi.org/10.1093/cid/ciaa453
Peignier A, Parker D. Pseudomonas aeruginosa can degrade interferon λ, thereby repressing the antiviral response of bronchial epithelial cells. J. Interferon Cytokine Res. 2020;40(8):429-431. doi: https://doi.org/10.1089/jir.2020.0057
Salka K, Arroyo M, Chorvinsky E, Abutaleb K, Perez GF, Wolf S, Xuchen X, Weinstock J, Gutierrez MJ, Pérez-Losada M, Pillai DK, Nino G. Innate IFN-lambda responses to dsRNA in the human infant airway epithelium and clinical regulatory factors during viral respiratory infections in early life. Clin. Exp. Allergy. 2020;50(9):1044-1054. doi: https://doi.org/10.1111/cea.13701
Svyatchenko VA, Nikonov SD, Mayorov AP, Gelfond ML, Loktev VB. Antiviral photodynamic therapy: Inactivation and inhibition of SARS-CoV-2 in vitro using methylene blue and Radachlorin. Photodiagnosis Photodyn. Ther. 2021;33:102112. doi: https://doi.org/10.1016/j.pdpdt.2020.102112
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 172, No. 7, pp. 65-69, July, 2021
Rights and permissions
About this article
Cite this article
Madonov, P.G., Svyatchenko, V.A., Legostaev, S.S. et al. Evaluation of the Anti-Viral Activity of Human Recombinant Interferon Lambda-1 against SARS-CoV-2. Bull Exp Biol Med 172, 53–56 (2021). https://doi.org/10.1007/s10517-021-05330-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-021-05330-0