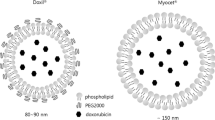
Preclinical study of therapeutic properties of an innovative drug Doxorubicin-NPh (doxorubicin in the form of ultrafine suspension of phospholipid liposomes) in comparison with free doxorubicin (Doxorubicin-Teva) and protected doxorubicin (Caelyx) was performed on transplanted murine tumor models. All these drugs were efficient in Ca755 breast carcinoma model (tumor growth inhibition ≈100%, increase in lifespan 90.6-114.3%). In P388 lymphocytic leukemia and LLC lung carcinoma, advantages of the protected doxorubicin by the benefit/risk ratio (width of therapeutic interval) were demonstrated: Caelyx>Doxorubicin-NPh>Doxorubicin-Teva. Doxorubicin-NPh and Caelyx exhibited similar therapeutic activity in the LLC model, especially when administered 3 times with 3-day intervals; for Doxorubicin-Teva, the optimal interval between the injections was 7 days.
Similar content being viewed by others
References
Malignant Neoplasms in Russia in 2017 (Morbidity and Mortality). Kaprin AD, Starinskii VV, Petrova GV, eds. Moscow, 2018. Russian.
Medvedeva NV, Torkhovskaya TI, Kostryukova LV, Zakharova TS, Kudinov VA, Kasatkina EO, Prozorovskiy VN, Ipatova OM. Influence of doxorubicin inclusion into phospholipid nanoparticles on tumor accumulation and specific activity. Biomed. Khimiya. 2017;63(1):56-61. Russian.
Archakov AI, Ipatova OM, Medvedeva NV, Prozorovskij VN, Torkhovskaja TI, Tikhonova EG, Zykova MG, Voskresenskaja AA. Patent Ru No. 2411935. Pharmaceutical composition based on doxorubicine and phospholipid nanoparticles for treatment of oncologic diseases. Bull. No. 5. Published February 20, 2011.
Handbook of Cancer Chemotherapy. Skeel RT, ed. Moscow, 2011. Russian.
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Long-term outcomes for neoadjuvant versus adjuvant chemotherapy in early breast cancer: meta-analysis of individual patient data from ten randomised trials. Lancet Oncol. 2018;19(1):27-39. https://doi.org/10.1016/S1470-2045(17)30777-5
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 169, No. 6, pp. 720-726, June, 2020
Rights and permissions
About this article
Cite this article
Nemtsova, E.R., Tikhonova, E.G., Bezborodova, O.A. et al. Preclinical Study of Pharmacological Properties of Doxorubicin-NPh. Bull Exp Biol Med 169, 778–782 (2020). https://doi.org/10.1007/s10517-020-04977-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-020-04977-5