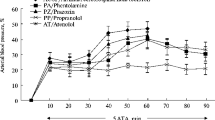
We studied the involvement of the autonomic nervous system in the antiarrhythmic effect of intermittent hypobaric hypoxia modeled by daily placing the rats into an altitude chamber at 405 mm Hg (5000 m above sea level). The antiarrhythmic effect of hypoxia was observed on the model of acute coronary occlusion/reperfusion in vivo, but not during simulation of total ischemia/reperfusion of the isolated myocardium. Intravenous injection of ganglionic blocker hexamethonium (30 mg/kg) 15 min prior to in vivo coronary occlusion modeling abolished the antiarrhythmic effect of intermittent hypobaric hypoxia, which suggests that this effect is mediated via activation of the autonomic nervous system.
Similar content being viewed by others

References
Ishekov АN, Mosyagin IG. Dynamics of cardiorespiratory system indices in students during adaptation to atmospheric hypoxic hypoxia in European North of Russia. Ekol. Chel. 2009;(1):38-42. Russian.
Lishmanov YuB, Naryzhnaya NV, Maslov LN, Gross GJ. Opiategic component of the antiarrhythmic effect of adaptation to hypoxia in in vivo model of ischemia and reperfusion. Patol. Fiziol. Eksp. Ter. 2003;(1):19-21. Russian.
Lishmanov IB, Uskina EV, Krylatov AV, Kondrat’ev BI, Ugdyzhekova DS, Maslov LN. Modulation of the antiarrhythmic effect by endogenous opioids during adaptation to hypoxia in rats. Ross. Fiziol. Zh. 1998;84(4):363-372. Russian.
Maslov LN, Krylatov AV, Lishmanov YuB. Antiarrhythmic effects of μ-opiate receptor agonists in rats with epinephrine-induced arrhythmias: Role of the vegetative nervous system. Bull. Exp. Biol. Med. 1996;122(1):671-673.
Mikhailova SD, Storozhakov GI, Bebyakova NA, Semushkina TM. Role of vagus nerves in antiarrhythmic effect of DAGO in acute myocardial ischemia. Bull. Exp. Biol. Med. 1997;124(4):952-954.
Boushel R, Calbet JA, Rådegran G, Sondergaard H, Wagner PD, Saltin B. Parasympathetic neural activity accounts for the lowering of exercise heart rate at high altitude. Circulation. 2001;104(15):1785-1791.
Estrada JA, Barlow MA, Yoshishige D, Williams AG Jr, Downey HF, Mallet RT, Caffrey JL. d-Opioid receptors: pivotal role in intermittent hypoxia-augmentation of cardiac parasympathetic control and plasticity. Auton. Neurosci. 2016;198:38-49.
Kent KM, Smith ER, Redwood DR, Epstein SE. Electrical stability of acutely ischemic myocardium. Influences of heart rate and vagal stimulation. Circulation. 1973;47(2):291-298.
Laubie M, Schmitt H, Vincent M. Vagal bradycardia produced by microinjections of morphine-like drugs into the nucleus ambiguus in anaesthetized dogs. Eur. J. Pharmacol. 1979;59(3-4):287-291.
Meerson FZ, Ustinova EE, Orlova EH. Prevention and elimination of heart arrhythmias by adaptation to intermittent high altitude hypoxia. Clin. Cardiol. 1987;10(12):783-789.
Schultz JE, Yao Z, Cavero I, Gross GJ. Glibenclamide-induced blockade of ischemic preconditioning is time dependent in intact rat heart. Am. J. Physiol. 1997;272(6, Pt 2):H2607-H2615.
Wang X, Dergacheva O, Griffioen KJ, Huang ZG, Evans C, Gold A, Bouairi E, Mendelowitz D. Action of kappa and Delta opioid agonists on premotor cardiac vagal neurons in the nucleus ambiguus. Neuroscience. 2004;129(1):235-241.
Wolfe BB, Voelkel NF. Effects of hypoxia on atrial muscarinic cholinergic receptors and cardiac parasympathetic responsiveness. Biochem. Pharmacol. 1983;32(13):1999-2002.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 163, No. 3, pp. 275-278, March, 2017
Rights and permissions
About this article
Cite this article
Naryzhnaya, N.V., Mukhamedzyanov, A.V., Lasukova, T.V. et al. Involvement of Autonomic Nervous System in Antiarrhythmic Effect of Intermittent Hypobaric Hypoxia. Bull Exp Biol Med 163, 299–301 (2017). https://doi.org/10.1007/s10517-017-3789-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-017-3789-8