Abstract
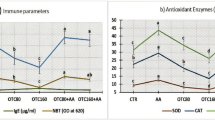
Ectoparasites Dactylogyrus spp. mainly infest fish gills and severely damage the host’s gill tissues. Correspondingly, the explanation of the interaction of fish with Dactylogyrus spp. infection is still insufficient. The present study describes the changes in hemato-biochemical, immune, antioxidant, genotoxic, and pathological indices response of Nile tilapia (Oreochromis niloticus) severely (n > 50), mildly infected (n = 1–50), and uninfected with Dactylogyrus spp. Data showed the adverse effect of hemato-biochemical indices in infected fish compared to uninfected, notably in severely infected O. niloticus. Compared to uninfected fish, there is a significantly decreased serum lysozyme and complement C3 and increased IgM and phagocytic activity along with significant upregulation of (COX-2), (IL-1β), (TNF-α), and (IL-10) genes in infected fish partially, in severely infected fish. Concisely, indices of antioxidants in the liver and gills marked an increased level of MDA in the infected fish compared to the uninfected fish. Conversely, levels of SOD, CAT, and GSH were decreased significantly with damaged DNA in the gills and liver of infected groups, particularly in severely infected (P < 0.05). Histopathologically investigating livers and gills in infected Nile tilapia indicated damaging and degenerative alterations, particularly with severe infection. Findings showed that Dactylogyrus spp.–infected Nile tilapia were effective in improving our knowledge of fish-pathogen interactions, which may be essential for fish defense against parasite infection.







Similar content being viewed by others
Data availability
No datasets were generated or analyzed during the current study.
Code availability
Not applicable.
Change history
23 March 2024
A Correction to this paper has been published: https://doi.org/10.1007/s10499-024-01474-8
References
Abbas WT, Abumourad IM, Mohamed LA, Abbas HH, Authman M, Soliman WS, Elgendy MY (2019) The role of the dietary supplementation of fenugreek seeds in growth and immunity in Nile Tilapia with or without cadmium contamination. Jordan J Biol Sci 12(5):649–656
Abdelhamid AF, Ayoub HF, Abd El-Gawad EA, Abdelghany MF, Abdel-Tawwab M (2021) Potential effects of dietary seaweeds mixture on the growth performance, antioxidant status, immunity response, and resistance of striped catfish (Pangasianodon hypophthalmus) against Aeromonas hydrophila infection. Fish Shellfish Immunol 119:76–83. https://doi.org/10.4194/1303-2712-v16_4_04
Abdelmonem AA, Metwally MM, Hussein HS, Elsheikha HM (2010) Gross and microscopic pathological changes associated with parasitic infection in European eel (Anguilla anguilla, Linnaeus 1758). Parasitol Res 106:463–469. https://doi.org/10.1007/s00436-009-1688-2
Abou-Okada M, AbuBakr HO, Hassan A, Abdel-Radi S, Aljuaydi SH, Abdelsalam M, Taha E, Younis NA, Abdel-Moneam DA (2021) Efficacy of Acriflavine for controlling parasitic diseases in farmed Nile tilapia with emphasis on fish health, gene expression analysis, oxidative stress, and histopathological alterations. Aquaculture 541:736791. https://doi.org/10.1016/j.aquaculture.2021.736791
Aebi H (1984) [13] Catalase in vitro. In Methods in enzymology (Vol. 105, pp. 121–126). Academic press. https://doi.org/10.1016/S0076-6879(84)05016-3
Bernet D, Schmidt H, Meier W, Burkhardt-Holm P, Wahli T (1999) Histopathology in fish: proposal for a protocol to assess aquatic pollution. J Fish Dis 22(1):25–34. https://doi.org/10.1046/j.1365-2761.1999.00134.x
Burgos-Aceves MA, Cohen A, Paolella G, Lepretti M, Smith Y, Faggio C, Lionetti L (2018a) Modulation of mitochondrial functions by xenobiotic-induced microRNA: from environmental sentinel organisms to mammals. Sci Total Environ 645:79–88. https://doi.org/10.1016/j.scitotenv.2018.07.109
Burgos-Aceves MA, Cohen A, Smith Y, Faggio C (2018b) MicroRNAs and their role on fish oxidative stress during xenobiotic environmental exposures. Ecotoxicol Environ Safety 148:995–1000. https://doi.org/10.1016/j.ecoenv.2017.12.001
Cai WQ, Li SF, Ma JY (2004) Diseases resistance of Nile tilapia (Oreochromis niloticus), blue tilapia (Oreochromis aureus) and their hybrid (female Nile tilapia× male blue tilapia) to Aeromonas sobria. Aquaculture 229(1–4):79–87. https://doi.org/10.1016/S0044-8486(03)00357-0
Chen YB, Hu J, Lyu QJ, Liu LJ, Wen LF, Yang XK, Zhao HH (2016) The effects of Natucin C-Natucin P mixture on blood biochemical parameters, antioxidant activity and non-specific immune responses in tilapia (Oreochromis niloticus). Fish Shellfish Immunol 55:367–373. https://doi.org/10.1016/j.fsi.2016.06.016
Chuang WL, Sun Pan B (2011) Anti-stress effects of Glycine tomentella Hayata in tilapia: inhibiting COX-2 expression and enhancing EPA synthesis in erythrocyte membrane and fish growth. J Agric Food Chem 59(17):9532–9541. https://doi.org/10.1021/jf2017308
Coulombe JJ, Favreau L (1963) A new simple semimicro method for colorimetric determination of urea. Clin Chem 9(1):102–108. https://doi.org/10.1093/clinchem/9.1.102
Del Rio-Zaragoza OB, Fajer-Avila EJ (1869) Almazán-Rueda P (2010) Haematological and gill responses to an experimental infection of dactylogyrid monogeneans on the spotted rose snapper Lutjanus guttatus (Steindachner. Aquac Res 41(11):1592–1601. https://doi.org/10.1111/j.1365-2109.2009.02471.x
Della Torre C, Petochi T, Corsi I, Dinardo MM, Baroni D, Alcaro L, Focardi S, Tursi A, Marino G, Frigeri A, Amato E (2010) DNA damage, severe organ lesions and high muscle levels of As and Hg in two benthic fish species from a chemical warfare agent dumping site in the Mediterranean Sea. Sci Total Environ 408(9):2136–2145. https://doi.org/10.1016/j.scitotenv.2010.01.001
Demers NE, Bayne CJ (1997) The immediate effects of stress on hormones and plasma lysozyme in rainbow trout. Dev Comp Immunol 21:363–373. https://doi.org/10.1016/S0145-305X(97)00009-8
Doumas BT, Watson WA, Biggs HG (1971) Albumin standards and the measurement of serum albumin with bromcresol green. Clin Chim Acta 31(1):87–96. https://doi.org/10.1016/0009-8981(71)90365-2
Doumas BT, Bayse DD, Carter RJ, Peters T Jr, Schaffer R (1981) A candidate reference method for determination of total protein in serum. I Development and Validation Clinical Chemistry 27(10):1642–1650. https://doi.org/10.1093/clinchem/27.10.1642
Eissa IAM, Abd El-Wahab MM, El-Genaidy HM, Abd El-Hady OK, Abd El-Fatah WA (2012) Studies on the prevailing nematodiasis among some wild freshwater fishes in Ismailia governorate. Egypt the Global Journal of Fisheries and Aquatic Research 5(5):71–85
Eissa AE, Attia MM, Elgendy MY, Ismail GA, Sabry NM, Prince A, Mahmoud MA, El-Demerdash GO, Abdelsalam M, Derwa HI (2021) Streptococcus, Centrocestus formosanus and Myxobolus tilapiae concurrent infections in farmed Nile tilapia (Oreochromis niloticus). Microb Pathog 158:105084. https://doi.org/10.1016/j.micpath.2021.105084
El Gamal SA, Adawy RS, Zaki VH, Zahran E (2023) Host–pathogen interaction unveiled by immune, oxidative stress, and cytokine expression analysis to experimental Saprolegnia parasitica infection in Nile tilapia. Sci Rep 13(1):9888. https://doi.org/10.1038/s41598-023-36892-w.10.1038/s41598-023-36892-w
FAO (2022) The State of World Fisheries and Aquaculture 2022. Towards Blue Transformation, Rome, FAO. https://doi.org/10.4060/cc0461en
Gewaily MS, Kassab M, Farrag FA, Almadaly EA, Atta MS, Abd-Elmaksoud A, Wakayama T (2020) Comparative expression of cell adhesion molecule1 (CADM1) in the testes of experimental mice and some farm animals. Acta Histochem 122:151456. https://doi.org/10.1016/j.acthis.2019.151456
Ghelichpour M, Taheri Mirghaed A, Hoseinifar SH, Khalili M, Yousefi M, Van Doan H, Perez-Jimenez A (2019) Expression of immune, antioxidant and stress related genes in different organs of common carp exposed to indoxacarb. Aquat Toxicol 208:208–216. https://doi.org/10.1016/j.aquatox.2019.01.011
Heinegård D, Tiderström G (1973) Determination of serum creatinine by a direct colorimetric method. Clin Chim Acta 43(3):305–310. https://doi.org/10.1016/0009-8981(73)90466-X
Henry RJ (1964) Colorimetric determination of total protein. Clinical Chemistry. Harper and Row Publ, New York, USA, p 181
Houston AH (1990) Blood and circulation. In: Schreck CC, Moyle PB (eds) Methods in fish biology. American Fisheries Society, Maryland, p 415–488
Ibrahim D, Abd El-Hamid MI, Al-Zaban MI, ElHady M, El-Azzouny MM, ElFeky TM, Omar AE (2022) Impacts of fortifying Nile tilapia (Oreochromis niloticus) diet with different strains of microalgae on its performance, fillet quality and disease resistance to Aeromonas hydrophila considering the interplay between antioxidant and inflammatory response. Antioxidants 11(11):2181. https://doi.org/10.3390/antiox11112181
Igeh PC (2019) Avenant-Oldewage A (2019) Pathological effects of Cichlidogyrus Philander Dou¨ellou, 1993 (Monogenea, Ancyrocephalidae) on the gills of Pseudocrenilabrus philander (Weber, 1897) (Cichlidae). J Fish Dis 00:1–8. https://doi.org/10.1111/jfd.13121
Jaiswal N, Srivastava R, Srivastava R, Mishra S, Jaiswal K, Malhotra S (2022) Assessment of genotoxicity induced by helminthes parasites in freshwater fishes of river Ganges. Indian J Experiment Biol (IJEB) 60(09):719–726. https://doi.org/10.56042/ijeb.v60i09.65148
Jaruboonyakorn P, Tejangkura T, Chontananarth T (2022) Multiplex PCR development for the simultaneous and rapid detection of two pathogenic flukes, Dactylogyrus spp. and Centrocestus formosanus, in ornamental fishes. Aquaculture 548:737660. https://doi.org/10.1016/j.aquaculture.2021.737660
Khan MH, Shareef PAA, Rehman A, Ullah R, Rehman L, Abidi SMA (2017) Genotoxic potential of Fasciola gigantica infection in experimentally infected rabbits. J Parasit Dis 41:423–428. https://doi.org/10.1007/s12639-016-0820-3
Kumar S, Raman RP, Prasad KP, Srivastava PP, Kumar S, Rajendran KV (2017) Modulation of innate immune responses and induction of oxidative stress biomarkers in Pangasianodon hypophthalmus following an experimental infection with dactylogyrid monogeneans. Fish Shellfish Immunol 63:334–343. https://doi.org/10.1016/j.fsi.2017.02.033
Kumar S, Raman RP, Prasad KP, Srivastava PP, Kumar S, Rajendran KV (2018) Effects on haematological and serum biochemical parameters of Pangasianodon hypophthalmus to an experimental infection of Thaparocleidus sp. (Monogenea: dactylogyridae). Exp Parasitol 188:1–7. https://doi.org/10.1016/j.exppara.2018.02.007
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25:402–408
Mahmoud MA, Abdelsalam M, Mahdy OA, El Miniawy HM, Ahmed ZA, Osman AH, Mohamed HM, Khattab AM, Ewiss MZ (2016) Infectious bacterial pathogens, parasites and pathological correlations of sewage pollution as an important threat to farmed fishes in Egypt. Environ Pollut 219:939–948. https://doi.org/10.1016/j.envpol.2016.09.044
Martins ML, Vieira FN, Jerônimo GT, Mourino JL, Dotta G, Speck GM, Bezerra AJ, Pedrotti FS, Buglione-Neto CC, Pereira G (2009) Leukocyte response and phagocytic activity in Nile tilapia experimentally infected with Enterococcus sp. Fish Physiol Biochem 35:219–222. https://doi.org/10.1007/s10695-008-9262-x
McCord JM, Fridovich I (1969) Superoxide dismutase: an enzymic function for erythrocuprein (hemocuprein). J Biol Chem 244(22):6049–6055
Molbert N, Agostini S, Alliot F, Angelier F, Biard C, Decencière B, Leroux-Coyau M, Millot A, Ribout C, Goutte A (2021) Parasitism reduces oxidative stress of fish host experimentally exposed to PAHs. Ecotoxicol Environ Saf 219:112322. https://doi.org/10.1016/j.ecoenv.2021.112322
Mori Krinal J, Ad Viral G, Rather MA, Kumar S (2023) Fundamentals of genotoxicity and antiparasitic drugs associated with genotoxicity in fish. In Xenobiotics in Aquatic Animals: Reproductive and Developmental Impacts (pp. 399–413). Singapore: Springer Nature Singapore. https://doi.org/10.1007/978-981-99-1214-8_20
Moussa MA, Mohamed HRH, Abdel-Khalek AA (2022) Metal accumulation and DNA damage in Oreochromis niloticus and Clarias gariepinus after chronic exposure to discharges of the batts drain: potential risk to human health. Bull Environ Contam Toxicol 108(6):1064–1073. https://doi.org/10.1007/s00128-022-03512-8
Mzula A, Wambura PN, Mdegela RH, Shirima GM (2019) Phenotypic and molecular detection of Aeromonads infection in farmed Nile tilapia in Southern highland and Northern Tanzania. Heliyon, 5(8). https://doi.org/10.1016/j.heliyon.2019.e02220.
Naito HK (1984) High-density lipoprotein (HDL) Cholesterol. In: Kaplan A (ed) Clinical chemistry: theory, analysis and correlation, vol 437. The CV Mosby Co, St Louis, Toronto, Princeton, pp 1207–1213
Noor El-Deen AI, Abd El-Hady OK, Kenawy AM, Mona SZ (2015) Study of the prevailing external parasitic diseases in cultured freshwater tilapia (Oreochromis niloticus) Egypt. Life Sci J 12(8):30–37
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95(2):351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Okon EM, Okocha RC, Taiwo AB, Michael FB, Bolanle AM (2023) Dynamics of co-infection in fish: a review of pathogen-host interaction and clinical outcome. Fish Shellfish Immunol Rep, p.100096. https://doi.org/10.1016/j.fsirep.2023.100096
Overkamp D, Mohammed SA, Cartledge C, Landon J (1988) Production of polyclonal antibodies in ascitic fluid of mice: technique and applications. J Immunoassay 9:51–68. https://doi.org/10.1080/01971528808053210
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70(1):158–169. https://doi.org/10.5555/uri:pii:0022214367900765
Panjvini F, Abarghuei S, Khara H, Parashkoh HM (2016) Parasitic infection alters haematology and immunity parameters of common carp, Cyprinus carpio, Linnaeus, 1758. J Parasit Dis 40:1540–1543. https://doi.org/10.1007/s12639-015-0723-8
Radwan M, El-Sharkawy MA, Alabssawy AN, Ghanem SF, Mohammadein A, Al Malki JS, Al-Thomali AW, Manaa EA, Soliman RA, Yassir S, Mekky AE (2023) The synergy between serious parasitic pathogens and bacterial infestation in the cultured Nile tilapia (Oreochromis niloticus): a severe threat to fish immunity, causing mass mortality and significant economic losses. Aquacult Int, pp.1–29. https://doi.org/10.1007/s10499-023-01093-9
Radwan M (2022) Vital economic threat of predatory birds and parasites to cultivated fishes in Egypt. Aquaculture 548:737666 https://doi.org/10.1016/j.aquaculture.2021.737666
Rastiannasab A, Afsharmanesh S, Rahimi R, Sharifian I (2016) Alternations in the liver enzymatic activity of common carp, Cyprinus carpio in response to parasites, Dactylogyrus spp. and Gyrodactylus spp. J Parasit Dis 40:1146–1149. https://doi.org/10.1007/s12639-014-0638-9
Recio L, Hobbs C, Caspary W, Witt KL (2010) Dose-response assessment of four genotoxic chemicals in a combined mouse and rat micronucleus (MN) and comet assay protocol. J Toxicol Sci 35(2):149–162. https://doi.org/10.2131/jts.35.149
Salazar AM, Mendlovic F, Cruz-Rivera M, Chávez-Talavera O, Sordo M, Avila G, Flisser A, Ostrosky-Wegman P (2013) Genotoxicity induced by Taenia solium and its reduction by immunization with calreticulin in a hamster model of taeniosis. Environ Mol Mutagen 54(5):347–353. https://doi.org/10.1002/em.21782
Saraiva A, Costa J, Serrão J, Cruz C, Eiras JC (2015) A histology-based fish health assessment of farmed seabass (Dicentrarchus labrax L.). Aquaculture 448:375–381. https://doi.org/10.1016/j.aquaculture.2015.06.028
Sigh J, Lindenstrøm T, Buchmann K (2004) The parasitic ciliate Ichthyophthirius multifiliis induces expression of immune relevant genes in rainbow trout, Oncorhynchus mykiss (Walbaum). J Fish Dis 27(7):409–417. https://doi.org/10.1111/j.1365-2761.2004.00558.x
Silvestre F (2020) Signaling pathways of oxidative stress in aquatic organisms exposed to xenobiotics. Journal of Experimental Zoology Part a: Ecological and Integrative Physiology 333(6):436–448. https://doi.org/10.1002/jez.2356
Souza CDF, Baldissera MD, Verdi CM, Santos RC, Da Rocha MIU, da Veiga ML, da Silva AS, Baldisserotto B (2019) Oxidative stress and antioxidant responses in Nile tilapia Oreochromis niloticus experimentally infected by Providencia rettgeri. Microb Pathog 131:164–169. https://doi.org/10.1016/j.micpath.2019.04.007
Sripa B, Kaewkes S, Sithithaworn P, Mairiang E, Laha T, Smout M, Pairojkul C, Bhudhisawasdi V, Tesana S, Thinkamrop B, Bethony JM (2007) Liver fluke induces cholangiocarcinoma. PLoS Med 4(7):e201. https://doi.org/10.1371/journal.pmed.0040201
Stankevičiūtė M, Sauliutė G, Makaras T, Čapukoitienė B, Vansevičiūtė G, Markovskaja S (2022) Biomarker responses in perch (Perca fluviatilis) under multiple stress: parasite co-infection and multicomponent metal mixture exposure. Environ Res 207:112170. https://doi.org/10.1016/j.envres.2021.112170
Sunyer JO, Tort L (1995) Natural hemolytic and bactericidal activities of sea bream Sparus aurata serum are effected by the alternative complement pathway. Vet Immunol Immunopathol 45:333–345. https://doi.org/10.1016/0165-2427(94)05430-Z
Syahputra K, Kania PW, Al-Jubury A, Marnis H, Setyawan AC, Buchmann K (2019) Differential immune gene response in gills, skin, and spleen of rainbow trout Oncorhynchus mykiss infected by Ichthyophthirius multifiliis. PLoS ONE 14(6):e0218630. https://doi.org/10.1371/journal.pone.0218630
Tang Y, Zeng W, Wang Y, Wang Q, Yin J, Li Y, Hu H (2020) Comparison of the blood parameters and histopathology between grass carp infected with a virulent and avirulent isolates of genotype II grass carp reovirus. Microb Pathog 139:103859. https://doi.org/10.1016/j.micpath.2019.103859
Tian JJ, Li YP, Zhang K, Wang GJ, Xia Y, Yu EM, Xie J (2021) Dietary Pennisetum sinese Roxb meal reduces lipid accumulation and alleviates enteritis in juvenile grass carp. Ctenopharyngodon Idella Aquaculture Reports 21:100915. https://doi.org/10.1016/j.aqrep.2021.100915
Tice RR, Agurell E, Anderson D, Burlinson B, Hartmann A, Kobayashi H, Miyamae Y, Rojas E, Ryu JC, Sasaki YF (2000) Single cell gel/comet assay: guidelines for in vitro and in vivo genetic toxicology testing. Environ Mol Mutagen 35(3):206–221. https://doi.org/10.1002/(SICI)1098-2280(2000)35:3%3c206::AID-EM8%3e3.0.CO;2-J
Wang G, Zhou Z, Cheng C, Yao J, Yang Z (2008) Osthol and isopimpinellin from Fructus cnidii for the control of Dactylogyrus intermedius in Carassius auratus. Vet Parasitol 158(1–2):144–151. https://doi.org/10.1016/j.vetpar.2008.07.034
Wang L, Zhang D, Xie J, Chang O, Wang Q, Shi C, Pan H (2023) Do ectoparasites on fish gills “talk” with gut microbiota far away? Aquaculture 562:738880. https://doi.org/10.1016/j.aquaculture.2022.738880
Witeska M, Kondera E, Bojarski B (2023) Hematological and hematopoietic analysis in fish toxicology—a review. Animals 13(16):2625. https://doi.org/10.3390/ani13162625
Yang BJ, Zou H, Zhou S, Wu SG, Wang GT, Li WX (2016) Seasonal dynamics and spatial distribution of the Dactylogyrus species on the gills of grass carp (Ctenopharyngodon idellus) from a fish pond in Wuhan. China J Parasitol 102(5):507–513. https://doi.org/10.1645/15-931
Yardimci B (1851) Pekmezci GZ (2012) Gill histopathology in cultured sea bass (Dicentrarchus labrax (L.) coinfected by Diplectanum aequans (Wagener, 1857) and Lernanthropus kroyeri (van Beneden. Ankara Üniversitesi Veteriner Fakültesi Dergisi 59(1):61–64. https://doi.org/10.1501/Vetfak_0000002502
Yilmaz S, Çelik EŞ, Ergün S, Ahmadifar E, Abdel-Latif HM (2023) Effects of dietary walnut (Juglans regia) leaves extract on immunity, gene expression responses, and disease resistance in Oreochromis niloticus. Fish Shellfish Immunol 135:108656. https://doi.org/10.1016/j.fsi.2023.108656
Yin X, Mu L, Fu S, Wu L, Han K, Wu H, Bian X, Wei X, Guo Z, Wang A, Ye J (2019) Expression and characterization of Nile tilapia (Oreochromis niloticus) secretory and membrane-bound IgM in response to bacterial infection. Aquaculture 508:214–222. https://doi.org/10.1016/j.aquaculture.2019.03.058
Younis NA, Thabit H, El-Samannoudy SI, Attia MM (2023) The immune responses of Oreochromis niloticus against Prohemistomum vivax encysted metacercariae infection with the evaluation of different biomarkers stressors. Sci Rep 13(1):11885. https://doi.org/10.1038/s41598-023-38809-z
Yusoff S F M, Christianus A N N I E, Matori F U A D, Talba M A, Hamid N H, Hamdan R H, Bakar S N A (2020) The impact of natural co-infection of dactylogyrus spp. and aeromonas hydrophila on behavioural, clinical, and histopathological changes of striped catfish, pangasianodon hypophthalmus (sauvage, 1878): a case study. J Sustain Sci Manag 10. https://doi.org/10.46754/jssm.2020.10.008
Zhi T, Huang C, Sun R, Zheng Y, Chen J, Xu X, Brown CL, Yang T (2020) Mucosal immune response of Nile tilapia Oreochromis niloticus during Gyrodactylus cichlidarum infection. Fish Shellfish Immunol 106:21–27. https://doi.org/10.1016/j.fsi.2020.07.025
Zhou Z, He S, Liu Y, Cao Y, Meng K, Yao B, Ringø E, Yoon I (2011) Gut microbial status induced by antibiotic growth promoter alters the prebiotic effects of dietary DVAQUA® on Aeromonas hydrophila-infected tilapia: production, intestinal bacterial community and non-specific immunity. Vet Microbiol 149(3–4):399–405. https://doi.org/10.1016/j.vetmic.2010.11.022
Zhou S, Zou H, Wu SG, Wang GT, Marcogliese DJ, Li WX (2017) Effects of goldfish (Carassius auratus) population size and body condition on the transmission of Gyrodactylus kobayashii (Monogenea). Parasitol 144(9):1221–1228. https://doi.org/10.1017/s0031182017000543
Acknowledgements
The researchers would like to acknowledge Deanship of Scientific Research, Taif University for funding this work. The authors would also like to thank the research staff of the Department of Zoology, Al-Azhar University, for their scientific guidance.
Author information
Authors and Affiliations
Contributions
Mahmoud Radwan: methodology, formal analysis, investigation, resources, data curation, writing—original draft, and project administration; Moussa Attia Moussa: formal analysis, statistical analysis, methodology, and editing; Mahmoud A. El-Sharkawy: formal analysis, statistical analysis, methodology, and editing; Salah M. El-Sharkawy: formal analysis, statistical analysis, methodology, and editing; Metwally G. Metwally: formal analysis, statistical analysis, methodology, and editing; Bassem E. Elaraby: formal analysis, statistical analysis, methodology, and editing; Kareem F. Darweesh: writing—original draft, methodology, and conceptualization; Marwa O. Abd El-Halim: writing—original draft, methodology, and conceptualization; Jamila S. Al malki: formal analysis, methodology, visualization, and editing; Amaal Mohammadein: formal analysis, methodology, conceptualization, and editing. Shahd Yassir: writing—original draft, methodology, and conceptualization; and Said M. A. Elraey: formal analysis, methodology, conceptualization, and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study was conducted according to the Ethics Committee of Institutional Animal Care and Use Committee guidelines, Zagazig University, Egypt (No. ZU-IACUC/1/F/367/2023).
Consent to participate
All of the authors consent to participate in the study above.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Brian Austin
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Radwan, M., Moussa, M.A., El-Sharkawy, M.A. et al. The inflammatory, genotoxicity, antioxidants, and pathological response to ectoparasite infection of cultured Nile tilapia. Aquacult Int (2024). https://doi.org/10.1007/s10499-024-01429-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10499-024-01429-z