Abstract
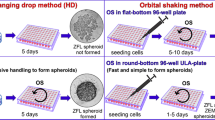
The present study aimed to generate 3D spheroid cultures from three rainbow trout (RTE, RTK and RTS) cell lines by simple and reproducible methods and evaluated them for the propagation of fish viral pathogen for the first time. The 3D spheroids were generated in the present study by scaffold-free method using 3D-orbital shaker. The growth of spheroids was documented on each day until 15 days and their viability was assessed by using AO/EB and Hoechst 33258 fluorescent staining under bright field microscope. The successfully generated RTK spheroids (RTKs) from the RTK cells were further used for the propagation of SJNNV virus in direct comparison of viral growth with conventional 2D monolayer culture of RTK cell line. Immunofluorescent staining confirms the expression of SJNNV major capsid protein in infected RTK spheroid earlier than the RTK monolayer. The samples collected from both RTK spheroid and RTK monolayer at different days of post infection was confirmed by RT-PCR and their viral copy numbers found high in RTK spheroid while quantified by qRT-PCR. Further, SJNNV major capsid protein was confirmed by western blot and ELISA. The viral load obtained using RTK spheroid was found to be high when compared to the monolayer and this study suggests that the spheroid generated from RTK cells is more suitable in vitro tool that can be used for propagation of fish viral pathogens in large-scale for production of whole virus vaccine.







Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Baron MG, Purcell WM, Jackson SK, Owen SF, Jha AN (2012) Towards a more representative in vitro method for fish ecotoxicology: morphological and biochemical characterisation of three-dimensional spheroidal hepatocytes. Ecotoxicol 21:2419–2429. https://doi.org/10.1007/s10646-012-0965-5
Baron MG, Mintram KS, Owen SF, Hetheridge MJ, Moody AJ, Purcell WM, Jackson SK, Jha AN (2017) Pharmaceutical metabolism in fish: using a 3-D hepatic in vitro model to assess clearance. PLoS One 12(1):e0168837. https://doi.org/10.1371/journal.pone.0168837
Barré-Sinoussi F, Montagutelli X (2015) Animal models are essential to biological research: issues and perspectives. Future Sci OA 1(4). https://doi.org/10.4155/fso.15.63
Barrila J, Radtke AL, Crabbé A, Sarker SF, Herbst-Kralovetz MM, Ott CM, Nickerson CA (2010) Organotypic 3D cell culture models: using the rotating wall vessel to study host–pathogen interactions. Nat Rev Microbiol 8(11):791–801. https://doi.org/10.1038/nrmicro2423
Caiazzo M, Okawa Y, Ranga A, Piersigilli A, Tabata Y, Lutolf MP (2016) Defined three-dimensional microenvironments boost induction of pluripotency. Nat Mat 15(3):344–52. https://doi.org/10.1038/nmat4536
Cesarz Z, Tamama K (2016) Spheroid culture of mesenchymal stem cells. Stem Cells Int 2016:9176357. https://doi.org/10.1155/2016/9176357
Chang TT, Hughes-Fulford M (2009) Monolayer and spheroid culture of human liver hepatocellular carcinoma cell line cells demonstrate distinct global gene expression patterns and functional phenotypes. Tissue Eng A 15(3):559–567. https://doi.org/10.1089/ten.tea.2007.0434
de Souza IR, Canavez ADPM, Schuck DC, Gagosian VSC, de Souza IR, Vicari T, da Silva TE, Cestari MM, Lorencini M, Leme DM (2021) Development of 3D cultures of zebrafish liver and embryo cell lines: a comparison of different spheroid formation methods. Ecotoxicol 30(9):1893–1909. https://doi.org/10.1007/s10646-021-02459-6
Faber MN, Sojan JM, Saraiva M, van West P, Secombes CJ (2021) Development of a 3D spheroid cell culture system from fish cell lines for in vitro infection studies: evaluation with Saprolegnia parasitica. J Fish Dis 44(6):701–710. https://doi.org/10.1111/jfd.13331
Fey SJ, Wrzesinski K (2012) Determination of drug toxicity using 3D spheroids constructed from an immortal human hepatocyte cell line. Toxicol Sci 127(2):403–411. https://doi.org/10.1093/toxsci/kfs122
Friedrich J, Seidel C, Ebner R, Kunz-Schughart LA (2009) Spheroid-based drug screen: considerations and practical approach. Nat Protoc 4(3):309–324. https://doi.org/10.1038/nprot.2008.226
Ghosh S, Spagnoli GC, Martin I, Ploegert S, Demougin P, Heberer M, Reschner A (2005) Three-dimensional culture of melanoma cells profoundly affects gene expression profile: a high density oligonucleotide array study. J Cell Physiol 204(2):522–531. https://doi.org/10.1002/jcp.20320
Heiss M, Hellström M, Kalén M, May T, Weber H, Hecker M, Augustin HG, Korff (2015) Endothelial cell spheroids as a versatile tool to study angiogenesis in vitro. FASEB J 29(7):3076–3084. https://doi.org/10.1096/fj.14-267633
Hultman MT, Løken KB, Grung M, Reid MJ, Lillicrap A (2019) Performance of three-dimensional rainbow trout (Oncorhynchus mykiss) hepatocyte spheroids for evaluating biotransformation of pyrene. Environ Toxicol Chem 38(8):1738–1747. https://doi.org/10.1002/etc.4476
Lammel T, Tsoukatou G, Jellinek J, Sturve J (2019) Development of three-dimensional (3D) spheroid cultures of the continuous rainbow trout liver cell line RTL-W1. Ecotoxicol 167:250–258. https://doi.org/10.1016/j.ecoenv.2018.10.009
Langan LM, Dodd NJ, Owen SF, Purcell WM, Jackson SK, Jha AN (2016) Direct measurements of oxygen gradients in spheroid culture system using electron parametric resonance oximetry. PLoS one 11(2):e0149492. https://doi.org/10.1371/journal.pone.0149492
Langan LM, Owen SF, Jha AN (2018) Establishment and long-term maintenance of primary intestinal epithelial cells cultured from the rainbow trout, Oncorhynchus mykiss. Biol Open 7(3):bio032870. https://doi.org/10.1242/bio.032870
Laschke MW, Menger MD (2017) Life is 3D: boosting spheroid function for tissue engineering. Trend Biotechnol 35(2):133–144. https://doi.org/10.1016/j.tibtech.2016.08.004
Lutolf MP, Lauer-Fields JL, Schmoekel HG, Metters AT, Weber FE, Fields GB, Hubbell JA (2003) Synthetic matrix metalloproteinase-sensitive hydrogels for the conduction of tissue regeneration: engineering cell-invasion characteristics. Proc Natl Acad Sci U S A 100(9):5413–5418
Malenovská H (2016) 3D rotating wall vessel and 2D cell culture of four veterinary virus pathogens: a comparison of virus yields, portions of infectious particles and virus growth curves. J Virol Methods 228:10–15. https://doi.org/10.1016/j.jviromet.2015.11.002
Molina-Jimenez F, Benedicto I, Thi VLD, Gondar V, Lavillette D, Marin JJ, Briz O, Moreno-Otero R, Aldabe R, Baumert TF, Cosset FL (2012) Matrigel-embedded 3D culture of Huh-7 cells as a hepatocyte-like polarized system to study hepatitis C virus cycle. Virol 425(1):31–39. https://doi.org/10.1016/j.virol.2011.12.021
Nishizawa T, Mori K, Nakai T, Furusawa I, Muroga K (1994) Polymerase chain reaction (PCR) amplification of RNA of striped jack nervous necrosis virus (SJNNV). Dis Aquat Organ 18:103–107
Nitta S, Hisasue M, Horiguchi Y, Yamada Y, Kikuchi K, Kubo T, Igarashi H, Neo S (2020) Three-dimensional spheroid culture of canine hepatocyte-like cells derived from bone marrow mesenchymal stem cells. Regen Ther 15:210–215. https://doi.org/10.1016/j.reth.2020.09.002
Pampaloni F, Reynaud EG, Stelzer EH (2007) The third dimension bridges the gap between cell culture and live tissue. Nat Rev Mol Cell Biol 8(10):839–845. https://doi.org/10.1038/nrm2236
Robinson NB, Krieger K, Khan FM, Huffman W, Chang M, Naik A, Yongle R, Hameed I, Krieger K, Girardi LN, Gaudino M (2019) The current state of animal models in research: a review. Inter J Surg 72:9–13. https://doi.org/10.1016/j.ijsu.2019.10.015
Rosellini A, Freer G, Quaranta P, Dovere V, Menichini M, Maggi F, Mazzetti P, Pistello M (2019) Enhanced in vitro virus expression using 3-dimensional cell culture spheroids for infection. J Virol Methods 265:99–104. https://doi.org/10.1016/j.jviromet.2018.12.017
Sainz B, TenCate V, Uprichard SL (2009) Three-dimensional Huh7 cell culture system for the study of Hepatitis C virus infection. Virol j 6:1–8. https://doi.org/10.1186/1743-422X-6-103
Saleh F, Harb A, Soudani N, Zaraket H (2020) A three-dimensional A549 cell culture model to study respiratory syncytial virus infections. J Infec Public Health 13(8):1142–1147. https://doi.org/10.1016/j.jiph.2020.03.011
Shoval H, Karsch-Bluman A, Brill-Karniely Y, Stern T, Zamir G, Hubert A, Benny O (2017) Tumor cells and their crosstalk with endothelial cells in 3D spheroids. Sci Rep 7(1):10428. https://doi.org/10.1038/s41598-017-10699-y
Suryakodi S, Majeed SA, Taju G, Vimal S, Sivakumar S, Ahmed AN, Shah FA, Bhat SA, Sarma D, Begum A, Hameed AS (2021) Development and characterization of novel cell lines from kidney and eye of rainbow trout, Oncorhynchus mykiss for virological studies. Aqua 532:736027. https://doi.org/10.1016/j.aquaculture.2020.736027
Uchea C, Owen SF, Chipman JK (2015) Functional xenobiotic metabolism and efflux transporters in trout hepatocyte spheroid cultures. Toxicol Res 4(2):494–507. https://doi.org/10.1039/c4tx00160e
Van Weemen B, Schuurs AHWM (1971) Immunoassay using antigen—enzyme conjugates. FEBS Lett 15(3):232–236
Vimal S, Madan N, Farook MA, Nambi KSN, Majeed SA, Rajkumar T, Venu S, Thirunavukkarasu AR, Hameed AS (2014) Production of recombinant vaccine using capsid gene of nodavirus to protect Asian sea bass, Lates calcarifer (Bloch, 1790). Aqua 418:148–154. https://doi.org/10.1016/j.aquaculture.2013.10.017
Weeber F, Ooft SN, Dijkstra KK, Voest EE (2017) Tumor organoids as a pre-clinical cancer model for drug discovery. Cell Chem Biol 24(9):1092–1100. https://doi.org/10.1016/j.chembiol.2017.06.012
Zhu J (2010) Bioactive modification of poly (ethylene glycol) hydrogels for tissue engineering. Biomat 31(17):4639–4656
Acknowledgements
The authors are thankful to the management and Principal of C. Abdul Hakeem College, Melvisharam, for providing the facilities to carry out this work. Prof. Thomas John (Former Professor & Head, PG & Research Department of Zoology, C. Abdul Hakeem College) is acknowledged for correcting the MS.
Funding
This study was funded by a grant from the Department of Biotechnology, Government of India, New Delhi, India, under the scheme of R&D projects on Biotechnology Network Programme, Grant/Award Number: BT/PR40452/NDB/39/750/2020 and BT/PR41899/AAQ/3/1006/2021.
Author information
Authors and Affiliations
Contributions
Suryakodi S: Investigation, Methodology, Data curation, Writing—original draft, Mithra S: Investigation, Formal analysis, Data curation, Nafeez Ahmed A: Methodology, Data curation, Kanimozhi K: Formal analysis, Data curation, Rajkumar V—Formal analysis, Data curation; Taju G: Methodology and Data curation, Abdul Majeed S: Methodology, Resources and Supervision, Sahul Hameed AS: Conceptualization, Methodology, Supervision, Project administration, Writing—review and editing.
Corresponding authors
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Brian Austin
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Suryakodi, S., Mithra, S., Nafeez Ahmed, A. et al. Development of three-dimensional (3D) spheroid culture system from rainbow trout kidney cell line (RTK) for in vitro production of fish viral pathogen. Aquacult Int (2024). https://doi.org/10.1007/s10499-024-01414-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10499-024-01414-6