Abstract
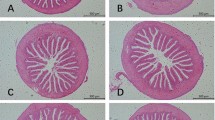
This study aimed to evaluate the different effects of dietary microencapsulated synbiotics on the growth, intestinal histology, immune responses, and gut microbiota diversity of white shrimp (Penaeus vannamei). This study used a completely randomized design with five treatments and three replications. Five different feed treatments were used to feed experimental shrimp for 8 weeks, namely, SBM (1% Bacillus NP5 108 CFU g−1 and 0.4% mannan-oligosaccharides (MOS)), SPM (1% Pseudoalteromonas piscicida 1Ub 108 CFU g−1 and 0.4% MOS), and SBPM (0.5% Bacillus NP5 108 CFU g−1, 0.5%, P. piscicida 1Ub 108 CFU g−1, and 0.4% MOS) and the controls without synbiotics administration consisted of the positive control (PC) and the negative control (NC). Pacific white shrimp from all treatments, except NC, were challenged with 106 CFU mL−1 Vibrio parahaemolyticus, injected intramuscularly. In this study, an in vitro test was performed that showed that P. piscicida 1Ub could utilize MOS as a prebiotic (synergistic effects). The results showed that dietary synbiotics with different effects for 60 days resulted in higher growth performance, wider gut microbiota diversity, improved intestinal histology, and elevated immune responses compared with controls (p < 0.05). Dietary synbiotics with synergistic effects also affected the composition of intestinal microflora. Beneficial bacteria, such as Bacillus, Glycomyces, and Weissella, were more abundant in SPM and SBPM treatments than in the other groups. After the challenge test, SPM and SBPM treatments showed significantly higher levels of immune response than in the PC. SPM and SBPM treatments (synbiotics with synergistic effects) showed a significantly higher survival rate (SR) than the other treatments.








Similar content being viewed by others
References
Cerezuela RC, Meseguer J, Esteban MA (2011) Current knowledge in synbiotic use for fish aquaculture: A review. J Aquac Res Dev s1:1–7. https://doi.org/10.4172/2155-9546.S1-008
Duan Y, Zhang Y, Dong H et al (2017) Effect of dietary Clostridium butyricum on growth, intestine health status and resistance to ammonia stress in Pacific white shrimp Litopenaeus vannamei. Fish Shellfish Immunol 65:25–33. https://doi.org/10.1016/j.fsi.2017.03.048
Dudek M, Adams J, Swain M et al (2014) Metaphylogenomic and potential functionality of the limpet Patella pellucida’s gastrointestinal tract microbiome. Int J Mol Sci 15:18819–18839. https://doi.org/10.3390/ijms151018819
Fan L, Li QX (2019) Characteristics of intestinal microbiota in the Pacific white shrimp Litopenaeus vannamei differing growth performances in the marine cultured environment. Aquaculture 505:450–461. https://doi.org/10.1016/j.aquaculture.2019.02.075
FAO (2022) The state of world fisheries and aquaculture 2022. FAO, Rome
Febrianti D, Yuhana M, Widanarni (2016) Dietary synbiotic microcapsule influence the immune responses, growth performance and microbial populations to white spot syndrome virus in Pacific white shrimp (Litopenaeus vannamei). J Fish Aquat Sci 11:28–42. https://doi.org/10.3923/jfas.2016.28.42
Hamsah H, Widanarni W, Alimuddin A et al (2019) Immune response and resistance of Pacific white shrimp larvae administered probiotic, prebiotic, and synbiotic through the bio-encapsulation of Artemia sp. Aquacult Int 27:567–580. https://doi.org/10.1007/s10499-019-00346-w
Hasyimi W, Widanarni W, Yuhana M (2020) Growth performance and intestinal microbiota diversity in Pacific white shrimp Litopenaeus vannamei fed with a probiotic bacterium, honey prebiotic, and synbiotic. Curr Microbiol 77:2982–2990. https://doi.org/10.1007/s00284-020-02117-w
Huynh TG, Cheng AC, Chi CC, Chiu KH, Liu CH (2018) A synbiotic improves the immunity of white shrimp, Litopenaeus vannamei: metabolomic analysis reveal compelling evidence. Fish Shellfish Immunol 79:284–293. https://doi.org/10.1016/j.fsi.2018.05.031
Huynh TG, Shiu YL, Nguyen TP et al (2017) Current applications, selection, and possible mechanisms of actions of Synbiotics in improving the growth and health status in aquaculture: a review. Fish Shellfish Immunol 64:367–382. https://doi.org/10.1016/j.fsi.2017.03.035
Kolida S, Gibson GR (2011) Synbiotics in health and disease. Annu Rev Food Sci Technol 2:373–393. https://doi.org/10.1146/annurev-food-022510-133739
Li H, Tian X, Dong S (2019) Growth performance, non-specific immunity, intestinal histology and disease resistance of Litopenaeus vannamei fed on a diet supplemented with live cells of Clostridium butyricum. Aquaculture 498:470–481. https://doi.org/10.1016/j.aquaculture.2018.09.003
Li J, Tan B, Mai K (2009) Dietary probiotic Bacillus OJ and isomaltooligosaccharides influence the intestine microbial populations, immune responses and resistance to white spot syndrome virus in shrimp (Litopenaeus vannamei). Aquaculture 291:35–40. https://doi.org/10.1016/j.aquaculture.2009.03.005
Liu CH, Chen JC (2004) Effect of ammonia on the immune response of white shrimp Litopenaeus vannamei and its susceptibility to Vibrio alginolyticus. Fish Shellfish Immunol 16:321–334. https://doi.org/10.1016/S1050-4648(03)00113-X
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Muharrama ARW, Widanarni W, Alimuddin A, Yuhana M (2021) Gene expression and immune response of pacific white shrimp given Bacillus NP5 probiotic and honey prebiotic and Vibrio parahaemolyticus infection. J Appl Aquac. https://doi.org/10.1080/10454438.2021.1873888
Munaeni W, Widanarni YM, Yuhana M, Setiawati M, Wahyudi AT (2020) Effect in white shrimp Litopenaeus vannamei of Eleutherine bulbosa (Mill.) Urb. Powder on immune genes expression and resistance against Vibrio parahaemolyticus infection. Fish Shellfish Immunol 102:218–227. https://doi.org/10.1016/j.fsi.2020.03.066
Rengpipat S, Rukpratanporn S, Piyatiratitivorakul S, Menasaveta P (2000) Immunity enhancement in black tiger shrimp (Penaeus monodon) by a probiont bacterium (Bacillus S11). Aquaculture 191:271–288. https://doi.org/10.1016/S0044-8486(00)00440-3
Rodriguez-Estrada U, Satoh S, Haga Y et al (2009) Effects of single and combined supplementation of Enterococcus faecalis, mannan oligosaccharide and polyhydroxybutyrate acid on growth performance and immune response of rainbow trout Oncorhynchus mykiss. Aquac Sci 57:609–617. https://doi.org/10.11233/aquaculturesci.57.609
Tamura K, Hemsworth GR, Déjean G et al (2017) Molecular mechanism by which prominent human gut Bacteroidetes utilize mixed-linkage beta-glucans, major health-promoting cereal polysaccharides. Cell Rep 21:417–430. https://doi.org/10.1016/j.celrep.2017.09.049
Tang K, Lin Y, Han Y, Jiao N (2017) Characterization of potential polysaccharide utilization systems in the marine Bacteroidetes Gramella flava JLT2011 using a multi-omics approach. Front Microbiol 8:220. https://doi.org/10.3389/fmicb.2017.00220
Tran L, Nunan L, Redman RM et al (2013) Determination of the infectious nature of the agent of acute hepatopancreatic necrosis syndrome affecting penaeid shrimp. Dis Aquat Organ 105:45–55. https://doi.org/10.3354/dao02621
Yao W, Li X, Zhang C et al (2021) Effects of dietary Synbiotics supplementation methods on growth, intestinal health, non-specific immunity and disease resistance of Pacific white shrimp, Litopenaeus vannamei. Fish Shellfish Immunol 112:46–55. https://doi.org/10.1016/j.fsi.2021.02.011
Zokaeifar H, Balcázar JL, Saad CR et al (2012) Effects of Bacillus subtilis on the growth performance, digestive enzymes, immune gene expression and disease resistance of white shrimp, Litopenaeus vannamei. Fish Shellfish Immunol 33:683–689. https://doi.org/10.1016/j.fsi.2012.05.027
Funding
This work was fully funded by the Indonesian Endowment Fund for Education/Lembaga Pengelola Dana Pendidikan (LPDP), Republic of Indonesia (grant number KET-2459/LPDP.4/2020).
Author information
Authors and Affiliations
Contributions
Maria Agustina Pardede performed the experiment, analyzed the data, and wrote the first version of the manuscript. Widanarni designed the study, wrote the first version of the manuscript, and approved it for publication. Sukenda designed the study, reviewed the first version of the manuscript, and approved the final manuscript for publication. Munti Yuhana reviewed the first version of the manuscript, designed the study, and approved the final manuscript for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Daniel Merrifield
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pardede, M.A., Widanarni, W., Sukenda, S. et al. Evaluation of dietary microencapsulated synbiotics Pseudoalteromonas piscicida 1UB, Bacillus NP5, and mannan-oligosaccharides to prevent Vibrio parahaemolyticus infection in Pacific white shrimp Penaeus vannamei. Aquacult Int 32, 2077–2091 (2024). https://doi.org/10.1007/s10499-023-01258-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-023-01258-6