Abstract
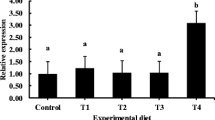
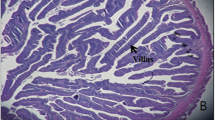
Silymarin is phytochemical extracted from milk thistle (Silybum marianum). It has been used as a natural hepatoprotector having antioxidant and detoxicant capacities. Micelle form of silymarin is one of the methods to increase its solubility and absorptivity in the intestine. Therefore, this study was performed to estimate the effects of dietary micelle silymarin on olive flounder (Paralichthys olivaceus). Experimental diets were prepared by adding dietary micelle silymarin by 0.025, 0.05, 0.1, 0.2 and 0.4% and regular silymarin by 0.1 and 0.2% to a Con diet (designated as M0025, M005, M01, M02, M04, S01 and S02, respectively). After the feeding trial for 70 days, dietary micelle silymarin significantly enhanced growth performance and feed intake of olive flounder. Innate immune responses such as lysozyme, anti-protease, myeloperoxidase and total immunoglobulin were significantly improved by micelle silymarin. Micelle silymarin groups were shown significantly higher antioxidant capacities as superoxide dismutase and glutathione peroxidase, and lower lipid peroxidation as malondialdehyde. Intestine and liver tissues were more positively developed by micelle silymarin. These results indicate that dietary micelle silymarin can enhance the growth performance, feed utilization, innate immune response, antioxidant capacity and histological development of olive flounder. In conclusion, the optimal supplementation level of micelle silymarin is likely to be 0.05–0.1% in olive flounder feed. Compared to regular form, dietary micelle silymarin showed similar or higher effects on olive flounder even at low supplementation levels. Therefore, dietary micelle silymarin is expected to be more effective and economical supplement for olive flounder than regular silymarin.


Similar content being viewed by others
Data availability
The data in this study will be available from the corresponding author upon reasonable request.
References
Abdel-Latif HM, Shukry M, Noreldin AE, Ahmed HA, El-Bahrawy A, Ghetas HA, Khalifa E (2023) Milk thistle (Silybum marianum) extract improves growth, immunity, serum biochemical indices, antioxidant state, hepatic histoarchitecture, and intestinal histomorphometry of striped catfish, Pangasianodon hypophthalmus Aquaculture 562:738761. https://doi.org/10.1016/j.aquaculture.2022.738761
Ahmadi K, Banaee M, Vosoghei AR, Mirvaghefei AR, Ataeimehr B (2012) Evaluation of the immunomodulatory effects of silymarin extract (Silybum marianum) on some immune parameters of rainbow trout, Oncorhynchus mykiss (Actinopterygii: Salmoniformes: Salmonidae). Acta Ichthyol Piscat 42(2):113–120. https://doi.org/10.3750/aip2011.42.2.04
AOAC (2005) Official methods of analysis, 18th edn. Association of Official Analytical Chemists Inc, Arlington
Banaee M, Sureda A, Mirvaghefi AR, Rafei GR (2011) Effects of long-term silymarin oral supplementation on the blood biochemical profile of rainbow trout (Oncorhynchus mykiss). Fish Physiol Biochem 37(4):885–896. https://doi.org/10.1007/s10695-011-9486-z
Bouderba S, Sanchez-Martin C, Villanueva GR, Detaille D, Koceïr EA (2014) Beneficial effects of silibinin against the progression of metabolic syndrome, increased oxidative stress, and liver steatosis in Psammomys obesus, a relevant animal model of human obesity and diabetes. J Diabetes 6(2):184–192. https://doi.org/10.1111/1753-0407.12083
Brown BA (1980) Routine hematology procedures. In: Williams L, Wilkins (eds) Hematology: principles and procedures. Lea & Febiger, Philadelphia, pp 71–112
Crocenzi FA, Pellegrino JM, Pozzi EJS, Mottino AD, Garay EAR, Roma MG (2000) Effect of silymarin on biliary bile salt secretion in the rat. Biochem Pharmacol 59(8):1015–1022. https://doi.org/10.1016/s0006-2952(99)00407-4
Dwivedi PS, Patil VS, Khanal P, Bhandare VV, Gurav S, Harish DR, Patil BM, Roy S (2022) System biology-based investigation of silymarin to trace hepatoprotective effect. Comput Biol Med 142:105223. https://doi.org/10.1016/j.compbiomed.2022.105223
El-Houseiny W, Abd El-Hakim YM, Metwally MMM, Ghfar SSA, Khalil AA (2022) The single or combined Silybum marianum and co-enzyme Q10 role in alleviating fluoride-induced impaired growth, immune suppression, oxidative stress, histological alterations, and reduced resistance to Aeromonas sobria in African catfish (Clarias gariepinus). Aquaculture 548:737693. https://doi.org/10.1016/j.aquaculture.2021.737693
Ellis AE (1990) Serum antiproteases in fish and lysozyme assays. In: Stolen JS, Fletcher TC, Anderson DP, Roberson BS, Van Muiswinkel WB (eds) Techniques in fish immunology. SOS publications, Fair Haven, pp 95–103
Faisal Z, Mohos V, Fliszár-Nyúl E, Valentová K, Káňová K, Lemli B, Kunsági-Máté S, Poór M (2021) Interaction of silymarin components and their sulfate metabolites with human serum albumin and cytochrome P450 (2C9, 2C19, 2D6, and 3A4) enzymes. Biomed Pharmacother 138:111459. https://doi.org/10.1016/j.biopha.2021.111459
Folch J, Lees M, Stanley GS (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226(1):497–509. https://doi.org/10.1016/s0021-9258(18)64849-5
Gao X, Guo M, Zhang Z, Shen P, Yang Z, Zhang N (2017) Baicalin promotes the bacteriostatic activity of lysozyme on S. aureus in mammary glands and neutrophilic granulocytes in mice. Oncotarget 8(12):19894. https://doi.org/10.18632/oncotarget.15193
Gillessen A, Schmidt HHJ (2020) Silymarin as supportive treatment in liver diseases: a narrative review. Adv Ther 37(4):1279–1301. https://doi.org/10.1007/s12325-020-01251-y
Han X, Wang Z, Wang M, Li J, Xu Y, He R, Guan H, Yue Z, Gong M (2016) Liver-targeting self-assembled hyaluronic acid-glycyrrhetinic acid micelles enhance hepato-protective effect of silybin after oral administration. Drug Deliv 23(5):1818–1829. https://doi.org/10.3109/10717544.2015.1108374
Hassaan MS, Mohammady EY, Soaudy MR, El-Garhy HA, Moustafa MM, Mohamed SA, El-Haroun ER (2019) Effect of Silybum marianum seeds as a feed additive on growth performance, serum biochemical indices, antioxidant status, and gene expression of Nile tilapia, Oreochromis niloticus (L.) fingerlings. Aquaculture 509:178–187. https://doi.org/10.1016/j.aquaculture.2019.05.006
Jahanian E, Mahdavi AH, Asgary S, Jahanian R (2017) Effects of dietary inclusion of silymarin on performance, intestinal morphology and ileal bacterial count in aflatoxin-challenged broiler chicks. J Anim Physiol Anim Nutr 101(5):e43–e54. https://doi.org/10.1111/jpn.12556
Jia R, Cao L, Du J, Xu P, Jeney G, Yin G (2013) The protective effect of silymarin on the carbon tetrachloride (CCl4)-induced liver injury in common carp (Cyprinus carpio). In Vitro Cell Dev Biol Anim 49(3):155–161. https://doi.org/10.1007/s11626-013-9587-3
Jiang X, Lin S, Lin Y, Fang Z, Xu S, Feng B, Zhuo Y, Li J, Che L, Jiang X, Wu D (2020) Effects of silymarin supplementation during transition and lactation on reproductive performance, milk composition and haematological parameters in sows. J Anim Physiol Anim Nutr 104(6):1896–1903. https://doi.org/10.1111/jpn.13425
Jutfelt F (2011) Barrier function of the gut. In: Farrell AP (ed) Encyclopedia of fish physiology: from genome to environment, vol 2. Academic, San Diego, pp 1322–1331
Kesharwani SS, Jain V, Dey S, Sharma S, Mallya P, Kumar VA (2020) An overview of advanced formulation and nanotechnology-based approaches for solubility and bioavailability enhancement of silymarin. J Drug Deliv Sci Technol 60:102021. https://doi.org/10.1016/j.jddst.2020.102021
Kim SS, Rahimnejad S, Kim KW, Lee BJ, Lee KJ (2013) Effects of dietary supplementation of spirulina and quercetin on growth, innate immune responses, disease resistance against Edwardsiella tarda, and dietary antioxidant capacity in the juvenile olive flounder Paralichthys olivaceus Fish Aquat Sci 16(1):7–14. https://doi.org/10.5657/fas.2013.0007
Korylak M, Vіshchur O, Hrytsyniak I (2019) The effect of milk thistle (Silybum marianum) on the state of T-and B-cell immunity and natural resistance of the immune system of two-year-old carp. Fish Sci Ukraine 3(49):89–100. https://doi.org/10.15407/fsu2019.03.089
KOSIS (Korean Statistical Information Service) (2021) Survey on the status of fish culture. https://kosis.kr/statisticsList/statisticsListIndex.do?vwcd=MT_ZTITLE&menuId=M_01_01#content-group. Accessed 20 June 2021
Kumar N, Rai A, Reddy ND, Shenoy RR, Mudgal J, Bansal P, Mudgal PP, Arumugam K, Udupa N, Sharma N, Rao CM (2019) Improved in vitro and in vivo hepatoprotective effects of liposomal silymarin in alcohol-induced hepatotoxicity in Wistar rats. Pharmacol Rep 71(4):703–712. https://doi.org/10.1016/j.pharep.2019.03.013
Kumari J, Sahoo PK (2005) Effects of cyclophosphamide on the immune system and disease resistance of asian catfish Clarias batrachus Fish Shellfish Immunol 19(4):307–316. https://doi.org/10.1016/j.fsi.2005.01.008
Lee JS, Kim ES, Lee HG (2017) Improving the water solubility and antimicrobial activity of silymarin by nanoencapsulation. Colloids Surf B Biointerfaces 154:171–177. https://doi.org/10.1016/j.colsurfb.2017.03.004
Li X, Yuan Q, Huang Y, Zhou Y, Liu Y (2010) Development of silymarin self-microemulsifying drug delivery system with enhanced oral bioavailability. AAPS PharmSciTech 11(2):672–678. https://doi.org/10.1208/s12249-010-9432-x
Li D, Zhang T, Xu C, Ji B (2011) Effect of pH on the interaction of baicalein with lysozyme by spectroscopic approaches. J Photochem Photobiol B 104(3):414–424. https://doi.org/10.1016/j.jphotobiol.2011.04.009
MacDonald-Ramos K, Michán L, Martínez-Ibarra A, Cerbón M (2021) Silymarin is an ally against insulin resistance: a review. Ann Hepatol 23:100255. https://doi.org/10.1016/j.aohep.2020.08.072
Mariappan B, Kaliyamurthi V, Binesh A (2023) Medicinal plants or plant derived compounds used in aquaculture. In: Jyothis M, Midhun SJ, Radhakrishnan EK, Ajay K (eds) Recent advances in aquaculture microbial technology. Academic, Cambridge, pp 153–207
Mohsen AM, Asfour MH, Salama AA (2017) Improved hepatoprotective activity of silymarin via encapsulation in the novel vesicular nanosystem bilosomes. Drug Dev Ind Pharm 43(12):2043–2054. https://doi.org/10.1080/03639045.2017.1361968
Nguyen MH, Yu H, Dong B, Hadinoto K (2016) A supersaturating delivery system of silibinin exhibiting high payload achieved by amorphous nano-complexation with chitosan. Eur J Pharm Sci 89:163–171. https://doi.org/10.1016/j.ejps.2016.04.036
Pang B, Bi S, Wang Y, Yan L, Wang T (2012) Investigation on the interactions of silymarin to bovine serum albumin and lysozyme by fluorescence and absorbance. J Lumin 132(4):895–900. https://doi.org/10.1016/j.jlumin.2011.11.021
Pepping J (1999) Milk thistle: Silybum marianum Am J Health Syst Pharm 56(12):1195–1197. https://doi.org/10.1093/ajhp/56.12.1195
Piazzini V, D’Ambrosio M, Luceri C, Cinci L, Landucci E, Bilia AR, Bergonzi MC (2019) Formulation of nanomicelles to improve the solubility and the oral absorption of silymarin. Molecules 24(9):1688. https://doi.org/10.3390/molecules24091688
Quade MJ, Roth JA (1997) A rapid, direct assay to measure degranulation of bovine neutrophil primary granules. Vet Immunol Immunopathol 58:239–248. https://doi.org/10.1016/S0165-2427(97)00048-2
Shin JH, Lee CL, Kim YJ, Shin JB, Lim HW, Yun KS, Lee KJ (2020) Dietary supplementation of phytosterol for olive flounder Paralichthys olivaceus Korean J Fish Aquat Sci 53(2):191–202. https://doi.org/10.5657/KFAS.2020.0191
Shriram RG, Moin A, Alotaibi HF, Khafagy ES, Al Saqr A, Abu Lila AS, Charyulu RN (2022) Phytosomes as a plausible nano-delivery system for enhanced oral bioavailability and improved hepatoprotective activity of silymarin. Pharmaceuticals 15(7):790. https://doi.org/10.3390/ph15070790
Siwicki AK, Anderson DP (1993) Nonspecific defense mechanisms assay in fish: II. Potential killing activity of neutrophils and macrophages, lysozyme activity in serum and organs and total immunoglobulin level in serum. In: Siwicki AK, Anderson DP, Waluga J (eds) Fish disease diagnosis and prevention methods. Wydawnictwo Instytutu Rybactwa Strodladowego, Olsztyn, pp 105–111
Soto C, Mena R, Luna J, Cerbon M, Larrieta E, Vital P, Uria E, Sanchez M, Recoba R, Barron H, Favari L, Lara A (2004) Silymarin induces recovery of pancreatic function after alloxan damage in rats. Life Sci 75(18):2167–2180. https://doi.org/10.1016/j.lfs.2004.04.019
Surai PF (2015) Silymarin as a natural antioxidant: an overview of the current evidence and perspectives. Antioxidants 4(1):204–247. https://doi.org/10.3390/antiox4010204
Veisi S, Johari SA, Tyler CR, Mansouri B, Esmaeilbeigi M (2021) Antioxidant properties of dietary supplements of free and nanoencapsulated silymarin and their ameliorative effects on silver nanoparticles induced oxidative stress in Nile tilapia (Oreochromis niloticus). Environ Sci Pollut Res 28(20):26055–26063. https://doi.org/10.1007/s11356-021-12568-8
Wang J, Zhou H, Wang X, Mai K, He G (2019) Effects of silymarin on growth performance, antioxidant capacity and immune response in turbot (Scophthalmus maximus L). J World Aquac Soc 50(6):1168–1181. https://doi.org/10.1111/jwas.12614
Wang X, Zhang Z, Wu SC (2020) Health benefits of Silybum marianum: Phytochemistry, pharmacology, and applications. J Agric Food Chem 68(42):11644–11664. https://doi.org/10.1021/acs.jafc.0c04791
Wei L, Wu P, Zhou XQ, Jiang WD, Liu Y, Kuang SY, Tang L, Feng L (2020) Dietary silymarin supplementation enhanced growth performance and improved intestinal apical junctional complex on juvenile grass carp (Ctenopharyngodon idella). Aquaculture 525:735311. https://doi.org/10.1016/j.aquaculture.2020.735311
Wu JP, Tsai CC, Yeh YL, Lin YM, Lin CC, Day CH, Shen CY, Padma VV, Pan LF, Huang CY (2015) Silymarin accelerates liver regeneration after partial hepatectomy. Evid Based Complement Alternat Med. https://doi.org/10.1155/2015/603529
Xiao P, Ji H, Ye Y, Zhang B, Chen Y, Tian J, Liu P, Chen L, Du Z (2017) Dietary silymarin supplementation promotes growth performance and improves lipid metabolism and health status in grass carp (Ctenopharyngodon idellus) fed diets with elevated lipid levels. Fish Physiol Biochem 43(1):245–263. https://doi.org/10.1007/s10695-016-0283-6
Yan-yu X, Yun-mei S, Zhi-peng C, Qi-neng P (2006) Preparation of silymarin proliposome: a new way to increase oral bioavailability of silymarin in beagle dogs. Int J Pharm 319(1–2):162–168. https://doi.org/10.1016/j.ijpharm.2006.03.037
Yao C, Huang W, Liu Y, Yin Z, Xu N, He Y, Wu X, Mai K, Ai Q (2020) Effects of dietary silymarin (SM) supplementation on growth performance, digestive enzyme activities, antioxidant capacity and lipid metabolism gene expression in large yellow croaker (Larimichthys crocea) larvae. Aquac Nutr 26(6):2225–2234. https://doi.org/10.1111/anu.13159
Funding
This study was supported by Synergen Inc. (Bucheon, South Korea) and the National Research Foundation of South Korea (no. 2019R1A6A1A03033553).
Author information
Authors and Affiliations
Contributions
Han-Se Kim and Sehee Jo participated to feed preparation, conducted the feeding trial and data collection.Han-Se Kim mainly contributed to sampling, analyses, writing and editing the manuscript.Kwan-Sik Yun designed the experiment and provided ingredients.Kyeong-Jun Lee organized and supervised the experiment and completed the manuscript.
Corresponding author
Ethics declarations
Ethical approval
Experimental procedures were accepted (approval number: 2021 − 0069) by the Animal Care and Use Committee of Jeju National University.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Daniel Merrifield.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, HS., Jo, S., Yun, KS. et al. Effects of dietary micelle silymarin on the growth performance, feed utilization and health of olive flounder (Paralichthys olivaceus). Aquacult Int 31, 3419–3436 (2023). https://doi.org/10.1007/s10499-023-01135-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-023-01135-2