Abstract
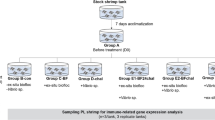
Globally , Vibrio parahaemolyticus AHPND is the major disease outbreaks in Litopenaeus vannamei shrimp ponds and has reduced shrimp production, significantly. This study aimed to simulate the outbreak of Vibrio spp. by immersion infection at 106 CFU mL−1. After 7 days of immersion, the survived L. vannamei were then orally supplemented and treated with alginate, spirulina, and alginate + spirulina for 12 days to recover the innate immune and reduce mortality. The cellular compound, namely total hemocyte count and phagocytic activity and index, and the humoral compound, namely phenol oxidase and superoxide dismutase, were determined. The q-RT PCR was applied to measure the immune-related gene expression (ProPO, LGBP, and lectin type C). Challenged by VpAHPND intra-muscularly at 109 CFU mL−1 was the administered. After challenged, all survival rate of shrimps from supplemented diet treatments were better than control (22% ± 7.78) (p < 0.05). The best shrimp survival rate was reached at Alg 3.0 mg g−1 (50% ± 2.12) treatment. THC showed that all supplemented shrimps were significantly different (p < 0.05) from control. PA and PI parameters were not significantly (p < 0.05) different from the control. SOD activity showed that the effective treatments were SWE 5.0 µg g−1 and Alg 3.0 mg g−1 + SWE 5.0 µg g−1. Before IM VpAHPND injection at 109 CFU mL−1, PO and gene expression (ProPO and LGBP (lipopolysaccharide and β-1,3-glucan binding protein)) shrimps from Alg 3.0 mg g−1 treatment reached the best results significantly (p < 0.05) over others and all treatments decreased extremely after injection. After challenge, lectin type C gene expression was upregulated (p < 0.05) in all treatments, except control. This study shows that alginate has the ability to recover the L. vannamei survival and immune system in a dose-dependent manner.






Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
References
Adilah RN, Chiu ST, Hu SY, Ballantyne R, Happy N, Cheng AC, Liu CH (2022) Improvement in the probiotic efficacy of Bacillus subtilis E20-stimulates growth and health status of white shrimp, Litopenaeus vannamei via encapsulation in alginate and coated with chitosan. Fish Shellfish Immunol 125:74–83. https://doi.org/10.1016/j.fsi.2022.05.002
Alagawany M, Taha AE, Noreldin A, El-Tarabily KA, Abd El-Hack ME (2021) Nutritional applications of species of Spirulina and Chlorella in farmed fish: A review. Aquaculture 542:736841. https://doi.org/10.1016/j.aquaculture.2021.736841
Amparyup P, Charoensapsri W, Tassanakajon A (2013) Prophenoloxidase system and its role in shrimp immune responses against major pathogens. Fish Shellfish Immunol 34:990–1001. https://doi.org/10.1016/j.fsi.2012.08.019
Aranguren LF, Han JE, Tang KFJ (2017) Enterocytozoon hepatopenaei (EHP) is a risk factor for acute hepatopancreatic necrosis disease (AHPND) and septic hepatopancreatic necrosis (SHPN) in the Pacific white shrimp Penaeus vannamei. Aquaculture 471:37–42. https://doi.org/10.1016/j.aquaculture.2016.12.038
Asche F, Anderson JL, Botta R, Kumar G, Abrahamsen EB, Nguyen LT, Valderrama D (2021) The economics of shrimp disease. J Invertebr Pathol 186:107397. https://doi.org/10.1016/j.jip.2020.107397
Baker-Austin C, McArthur JV, Lindell AH, Wright MS, Tuckfield RC, Gooch J, Warner L, Oliver J, Stepanauskas R (2009) Multi-site analysis reveals widespread antibiotic resistance in the marine pathogen Vibrio vulnificus. Microb Ecol 57:151–159. https://doi.org/10.1007/s00248-008-9413-8
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Cerenius L, Lee BL, Söderhäll K (2008) The proPO-system: pros and cons for its role in invertebrate immunity. Trends Immunol 29:263–271. https://doi.org/10.1016/j.it.2008.02.009
Chen YY, Chen JC, Kuo YH, Lin YC, Chang YH, Gong HY, Huang CL (2016a) Lipopolysaccharide and β-1,3-glucan-binding protein (LGBP) bind to seaweed polysaccharides and activate the prophenoloxidase system in white shrimp Litopenaeus vannamei. Dev Comp Immunol 55:144–151. https://doi.org/10.1016/j.dci.2015.10.023
Chen YY, Chen JC, Tayag CM, Li HF, Putra DF, Kuo YH, Bai JC, Chang YH (2016b) Spirulina elicits the activation of innate immunity and increases resistance against Vibrio alginolyticus in shrimp. Fish Shellfish Immunol 55:690–698. https://doi.org/10.1016/j.fsi.2016.06.042
Cheng W, Liu CH, Yeh ST, Chen JC (2004) The immune stimulatory effect of sodium alginate on the white shrimp Litopenaeus vannamei and its resistance against Vibrio alginolyticus. Fish Shellfish Immunol 17:41. https://doi.org/10.1016/j.fsi.2003.11.004
Cheng W, Liu CH, Kuo CM, Chen JC (2005) Dietary administration of sodium alginate enhances the immune ability of white shrimp Litopenaeus vannamei and its resistance against Vibrio alginolyticus. Fish Shellfish Immunol 18:1–12. https://doi.org/10.1016/j.fsi.2004.03.002
Choi M, Stevens AM, Smith SA, Taylor DP, Kuhn DD (2017) Strain and dose infectivity of Vibrio parahaemolyticus: the causative agent of early mortality syndrome in shrimp. Aquac Res 48:3719–3727. https://doi.org/10.1111/are.13197
Chung M-Y, Liu C-H, Chen Y-N, Cheng W (2011) Enhancing the reproductive performance of tiger shrimp, Penaeus monodon, by incorporating sodium alginate in the broodstock and larval diets. Aquaculture 312:180–184. https://doi.org/10.1016/j.aquaculture.2010.11.047
da Silva LV, Ossai S, Chigbu P, Parveen S (2021) Antimicrobial and genetic profiles of Vibrio vulnificus and Vibrio parahaemolyticus isolated from the Maryland Coastal Bays. United States. Front Microbiol 12:676249. https://doi.org/10.3389/fmicb.2021.676249
de la Peña LD, Cabillon NA, Catedral DD, Amar EC, Usero RC, Monotilla WD, Calpe AT, Fernandez DD, Saloma CP (2015) Acute hepatopancreatic necrosis disease (AHPND) outbreaks in Penaeus vannamei and P. monodon cultured in the Philippines. Dis Aquat Organ 116:251–254. https://doi.org/10.3354/dao02919
Dhar AK, Piamsomboon P, Aranguren Caro LF, Kanrar S, Adami R Jr, Juan YS (2019) First report of acute hepatopancreatic necrosis disease (AHPND) occurring in the USA. Dis Aquat Organ 132:241–247. https://doi.org/10.3354/dao03330
Fagutao FF, Koyama T, Kaizu A, Saito-Taki T, Kondo H, Aoki T, Hirono I (2009) Increased bacterial load in shrimp hemolymph in the absence of prophenoloxidase. Febs J 276:5298–5306. https://doi.org/10.1111/j.1742-4658.2009.07225.x
FAO (2021) Fishery and Aquaculture Statistics 2019/FAO Yearbook. Food and Agricultural Organization of the United Nations, Rome
Febriani D, Marlina E, Oktaviana A (2018) Total haemocytes of Pacific white shrimp (Litopenaeus vannamei) cultured at salinity of 10 ppt in various stocking density. J Aquacult Sci 3:1–8. https://doi.org/10.31093/joas.v3i1.35
Han JE, Tang KF, Tran LH, Lightner DV (2015) Photorhabdus insect-related (Pir) toxin-like genes in a plasmid of Vibrio parahaemolyticus, the causative agent of acute hepatopancreatic necrosis disease (AHPND) of shrimp. Dis Aquat Organ 113:33–40. https://doi.org/10.3354/dao02830
Han JE, Choi S-K, Han S-H, Lee SC, Jeon HJ, Lee C, Kim KY, Lee YS, Park SC, Rhee G, Park SY, Kim J-S, Park S, Kim JH, Lee K-J (2020) Genomic and histopathological characteristics of Vibrio parahaemolyticus isolated from an acute hepatopancreatic necrosis disease outbreak in Pacific white shrimp (Penaeus vannamei) cultured in Korea. Aquaculture 524:735284. https://doi.org/10.1016/j.aquaculture.2020.735284
Hidayati JR, Yudiati E, Pringgenies D, Oktaviyanti DT, Kusuma AP (2020) Comparative study on antioxidant activities, total phenolic compound and pigment contents of tropical Spirulina platensis, Gracilaria arcuata and Ulva lactuca extracted in different solvents polarity. E3S Web of Conferences 147:03012. https://doi.org/10.1051/e3sconf/202014703012
Hisatsune K, Kiuye A, Kondo S (1981) A comparative study of the sugar composition of o-antigenic lipopolysaccharides isolated from Vibrio alginolyticus and Vibrio parahaemolyticus. Microbiol Immunol 25:127–136. https://doi.org/10.1111/j.1348-0421.1981.tb00016.x
Hong To TT, Yanagawa H, Khanh Thuan N, Hiep DM, Cuong DV, Khai LTL, Taniguchi T, Kubo R, Hayashidani H (2020) Prevalence of Vibrio parahaemolyticus causing acute hepatopancreatic necrosis disease of shrimp in shrimp, molluscan shellfish and water samples in the Mekong Delta, Vietnam. Biology 9(10):312. https://doi.org/10.3390/biology9100312
Jahromi ST, Pourmozaffar S, Jahanbakhshi A, Rameshi H, Gozari M, Khodadadi M, Sohrabipour J, Behzadi S, Barzkar N, Nahavandi R, Zahedi MR, Moezzi M (2021) Effect of different levels of dietary Sargassum cristaefolium on growth performance, hematological parameters, histological structure of hepatopancreas and intestinal microbiota of Litopenaeus vannamei. Aquaculture 533:736130. https://doi.org/10.1016/j.aquaculture.2020.736130
Ji P-F, Yao C-L, Wang Z-Y (2009) Immune response and gene expression in shrimp (Litopenaeus vannamei) hemocytes and hepatopancreas against some pathogen-associated molecular patterns. Fish Shellfish Immunol 27:563–570. https://doi.org/10.1016/j.fsi.2009.08.001
Jiang Y, Jiang X, Wang P, Hu X (2017) Effects of alginate-derived oligosaccharides on immune ability of farm-cultured shrimp Penaeus vannamei and its resistance to Vibrio harveyi. N Am J Aquac 79:317. https://doi.org/10.1080/15222055.2017.1365787
Joshi J, Srisala J, Truong VH, Chen IT, Nuangsaeng B, Suthienkul O, Lo CF, Flegel TW, Sritunyalucksana K, Thitamadee S (2014) Variation in Vibrio parahaemolyticus isolates from a single Thai shrimp farm experiencing an outbreak of acute hepatopancreatic necrosis disease (AHPND). Aquaculture 428–429:297–302. https://doi.org/10.1016/j.aquaculture.2014.03.030
Kongchum P, Chimtong S, Prapaiwong N (2022) Association between single nucleotide polymorphisms of nLvALF1 and PEN2-1 genes and resistance to Vibrio parahaemolyticus in the Pacific white shrimp Litopenaeus vannamei. Aquac Fish 7:373–381. https://doi.org/10.1016/j.aaf.2021.08.003
Kumar R, Ng TH, Wang H-C (2020) Acute hepatopancreatic necrosis disease in penaeid shrimp. Rev Aquac 12:1867–1880. https://doi.org/10.1111/raq.12414
Kumar V, Roy S, Behera BK, Bossier P, Das BK (2021) Acute hepatopancreatic necrosis disease (AHPND): virulence, pathogenesis and mitigation strategies in shrimp aquaculture. Toxins 13. https://doi.org/10.3390/toxins13080524
Kurniawinata MI, Sukenda S, Wahjuningrum D, Widanarni W (2022) Bacterial diversity and community composition in the gut and rearing water of Pacific White shrimp Penaeus vannamei during an outbreak of white feces disease. Aquaculture 559:738431. https://doi.org/10.1016/j.aquaculture.2022.738431
Lafarga T, Fernández-Sevilla JM, González-López C, Acién-Fernández FG (2020) Spirulina for the food and functional food industries. Food Res Int. 137:109356. https://doi.org/10.1016/j.foodres.2020.109356
Lin C, Zhang LJ, Li B, Zhang F, Shen QR, Kong GQ, Wang XF, Cui SH, Dai R, Cao WQ, Zhang P (2020) Selenium-containing protein from selenium-enriched Spirulina platensis attenuates high glucose-induced calcification of MOVAS cells by inhibiting ROS-mediated DNA damage and regulating MAPK and PI3K/AKT pathways. Front Physiol 11:791. https://doi.org/10.3389/fphys.2020.00791
Liu C-H, Yeh S-P, Kuo C-M, Cheng W, Chou C-H (2006) The effect of sodium alginate on the immune response of tiger shrimp via dietary administration: activity and gene transcription. Fish Shellfish Immunol 21:442–452. https://doi.org/10.1016/j.fsi.2006.02.003
Liu CH, Yeh ST, Cheng SY, Chen JC (2004) The immune response of the white shrimp Litopenaeus vannamei and its susceptibility to Vibrio infection in relation with the moult cycle. Fish Shellfish Immunol 16:151–161. https://doi.org/10.1016/s1050-4648(03)00058-5
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Muñoz M, Cedeño R, Rodríguez J, van der Knaap W, Mialhe E, Bachère E (2000) Measurement of reactive oxygen intermediate production in haemocytes of the penaeid shrimp, Penaeus vannamei. Aquaculture 191:89–107. https://doi.org/10.1016/S0044-8486(00)00420-8
Navaneeth KA, Bhuvaneswari T, Rajan JJS, Alavandi SV, Vijayan KK, Otta SK (2020) Characterization of Vibrio parahaemolyticus isolates from shrimp farms of Southeast coast of India with special reference to Acute Hepatopancreatic Necrosis Disease (AHPND) status. Aquaculture 518:734813. https://doi.org/10.1016/j.aquaculture.2019.734813
Nguyen HT, Van TN, Ngoc TT, Boonyawiwat V, Rukkwamsuk T, Yawongsa A (2021) Risk factors associated with acute hepatopancreatic necrosis disease at shrimp farm level in Bac Lieu Province, Vietnam. Vet World. 14:1050–1058. https://doi.org/10.14202/vetworld.2021.1050-1058
Onohuean H, Agwu E, Nwodo UU (2022) Systematic review and meta-analysis of environmental Vibrio species – antibiotic resistance. Heliyon 8:e08845. https://doi.org/10.1016/j.heliyon.2022.e08845
Peña-Navarro N, Castro-Vásquez R, Vargas-Leitón B, Dolz G (2020) Molecular detection of acute hepatopancreatic necrosis disease (A4HPND) in Penaeus vannamei shrimps in Costa Rica. Aquaculture 523:735190. https://doi.org/10.1016/j.aquaculture.2020.735190
Perazzolo LM, Barracco MA (1997) The prophenoloxidase activating system of the shrimp Penaeus paulensis and associated factors. Dev Comp Immunol 21:385–395. https://doi.org/10.1016/S0145-305X(97)00022-0
Schar D, Klein EY, Laxminarayan R, Gilbert M, Van Boeckel TP (2020) Global trends in antimicrobial use in aquaculture. Sci Rep 10:1. https://doi.org/10.1038/s41598-020-78849-3
Shaw KS, Rosenberg Goldstein RE, He X, Jacobs JM, Crump BC, Sapkota AR (2014) Antimicrobial susceptibility of Vibrio vulnificus and Vibrio parahaemolyticus recovered from recreational and commercial areas of Chesapeake Bay and Maryland Coastal Bays. PLOS ONE 9:e89616. https://doi.org/10.1371/journal.pone.0089616
Sirikharin R, Taengchaiyaphum S, Sanguanrut P, Chi TD, Mavichak R, Proespraiwong P, Nuangsaeng B, Thitamadee S, Flegel TW, Sritunyalucksana K (2015) Characterization and PCR detection of binary, Pir-like toxins from Vibrio parahaemolyticus isolates that cause acute hepatopancreatic necrosis disease (AHPND) in shrimp. PLoS One 10:e0126987. https://doi.org/10.1371/journal.pone.0126987
Söderhäll K, Cerenius L (1992) Crustacean immunity. Annu Rev Fish Dis 2:3–23. https://doi.org/10.1016/0959-8030(92)90053-Z
Soonthornchai W, Rungrassamee W, Karoonuthaisiri N, Jarayabhand P, Klinbunga S, Söderhäll K, Jiravanichpaisal P (2010) Expression of immune-related genes in the digestive organ of shrimp, Penaeus monodon, after an oral infection by Vibrio harveyi. Dev Comp Immunol 34:19–28. https://doi.org/10.1016/j.dci.2009.07.007
Soto-Rodriguez SA, Lozano-Olvera R, Ramos-Clamont Montfort G, Zenteno E, Sánchez-Salgado JL, Vibanco-Pérez N, Aguilar Rendón KG (2022) New insights into the mechanism of action of PirAB from Vibrio Parahaemolyticus. Toxins 14(4):243. https://doi.org/10.3390/toxins14040243
Sritunyalucksana K, Lee SY, Söderhäll K (2002) A beta-1,3-glucan binding protein from the black tiger shrimp, Penaeus monodon. Dev Comp Immunol 26:237–245. https://doi.org/10.1016/s0145-305x(01)00074-x
Stephens NL, Bough WA, Beuchat LR, Heaton EK (1976) Preparation and evaluation of two microbiological media from shrimp heads and hulls. Appl Environ Microbiol 31:1–6. https://doi.org/10.1128/aem.31.1.1-6.1976
Sun J, Wang L, Wang B, Guo Z, Liu M, Jiang K, Luo Z (2007) Purification and characterisation of a natural lectin from the serum of the shrimp Litopenaeus vannamei. Fish Shellfish Immunol 23:292–299. https://doi.org/10.1016/j.fsi.2006.11.001
Tang KFJ, Pantoja CR, Redman RM, Han JE, Tran LH, Lightner DV (2015) Development of in situ hybridization and PCR assays for the detection of Enterocytozoon hepatopenaei (EHP), a microsporidian parasite infecting penaeid shrimp. J Invertebr Pathol 130:37–41. https://doi.org/10.1016/j.jip.2015.06.009
Tang KFJ, Han JE, Aranguren LF, White-Noble B, Schmidt MM, Piamsomboon P, Risdiana E, Hanggono B (2016) Dense populations of the microsporidian Enterocytozoon hepatopenaei (EHP) in feces of Penaeus vannamei exhibiting white feces syndrome and pathways of their transmission to healthy shrimp. J Invertebr Pathol 140:1–7. https://doi.org/10.1016/j.jip.2016.08.004
Tayag CM, Lin YC, Li CC, Liou CH, Chen JC (2010) Administration of the hot-water extract of Spirulina platensis enhanced the immune response of white shrimp Litopenaeus vannamei and its resistance against Vibrio alginolyticus. Fish Shellfish Immunol 28:764–773. https://doi.org/10.1016/j.fsi.2010.01.023
Trang TT, Hung NH, Ninh NH, Knibb W, Nguyen NH (2019) Genetic variation in disease resistance against white spot syndrome virus (WSSV) in Liptopenaeus vannamei. Front Genet 10:264. https://doi.org/10.3389/fgene.2019.00264
Wang X-W, Zhang X-W, Xu W-T, Zhao X-F, Wang J-X (2009) A novel C-type lectin (FcLec4) facilitates the clearance of Vibrio anguillarum in vivo in Chinese white shrimp. Dev Comp Immunol 33:1039–1047. https://doi.org/10.1016/j.dci.2009.05.004
Wang YC, Chang PS, Chen HY (2007) Tissue expressions of nine genes important to immune defence of the Pacific white shrimp Litopenaeus vannamei. Fish Shellfish Immunol 23:1161–1177. https://doi.org/10.1016/j.fsi.2007.04.004
Warner HR (1994) Superoxide dismutase, aging, and degenerative disease. Free Radic Biol Med 17:249–258. https://doi.org/10.1016/0891-5849(94)90080-9
Yudiati E, Isnansetyo A, Murwantoko A, Triyanto HCR (2016) Innate immune-stimulating and immune genes up-regulating activities of three types of alginate from Sargassum siliquosum in Pacific white shrimp. Litopenaeus Vannamei Fish Shellfish Immunol 54:46. https://doi.org/10.1016/j.fsi.2016.03.022
Yudiati E, Isnansetyo A (2017) Characterizing the three different alginate type of Sargassum siliquosum. Ilmu Kelaut 22:7. https://doi.org/10.14710/ik.ijms.22.1.7-14
Yudiati E, Pringgenies D, Djunaedi A, Arifin Z, Sudaryono A (2018) Free radicals scavenging activities of low molecular weight sodium alginate (LMWSA) from Sargassum polycystum, produced by thermal treatment. Aquac Indones 19:21–27. https://doi.org/10.21534/ai.v19i1.121
Yudiati E, Isnansetyo A, Murwantoko T, Handayani CR (2019) Alginate from Sargassum siliquosum simultaneously stimulates innate immunity, upregulates immune genes, and enhances resistance of Pacific white shrimp (Litopenaeus vannamei) against white spot syndrome virus (WSSV). Mar Biotechnol 21:503. https://doi.org/10.1007/s10126-019-09898-7
Yudiati E, Sedjati S, Azhar N, Oktarima WA, Arifin Z (2021) Spirulina water extract and Lactobacillus bulgaricus FNCC– 0041, Streptococcus thermophilus FNCC–0040 secretion as immunostimulants in gnotobiotic Artemia challenge tests against pathogenic Vibrio parahaemolyticus, V vulnificus, and V harveyi. IOP Confer Series: Earth Environ Sci 890:012018. https://doi.org/10.1088/1755-1315/890/1/012018
Zhang Y, Qiu L, Song L, Zhang H, Zhao J, Wang L, Yu Y, Li C, Li F, Xing K, Huang B (2009) Cloning and characterization of a novel C-type lectin gene from shrimp Litopenaeus vannamei. Fish Shellfish Immunol 26:183–192. https://doi.org/10.1016/j.fsi.2008.03.008
Zhang Z, Zhang Q, Wang T, Xu N, Lu T, Hong W, Penuelas J, Gillings M, Wang M, Gao W, Qian H (2022) Assessment of global health risk of antibiotic resistance genes. Nat Commun 13:1553. https://doi.org/10.1038/s41467-022-29283-8
Zheng Z, Li R, Aweya JJ, Yao D, Wang F, Li S, Tuan TN, Zhang Y (2021) The PirB toxin protein from Vibrio parahaemolyticus induces apoptosis in hemocytes of Penaeus vannamei. Virulence 12:481–492. https://doi.org/10.1080/21505594.2021.1872171
Funding
This work was supported by the Indonesian Government’s Ministry of Research and Higher Education (Grant numbers: 187–64/UN7.6.1/PP/2022) and the Faculty of Fisheries and Marine Science, Diponegoro University (Grant numbers: 247/UN7.5.https://doi.org/10.2/PP/2022).
Author information
Authors and Affiliations
Contributions
Nuril Azhar: conceptualization, methodology, validation, formal analysis, investigation; Ervia Yudiati: resources, writing original draft preparation, writing review and editing, visualization, supervision, project administration, funding acquisition. Both authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The research is conducted in accordance with Indonesia’s current animal welfare legislation. This experimental use of Litopenaeus vannamei does not require approval from the Indonesian Ethics Committee for Animal Use. All authors are willing to take part in this experiment.
Human and animal ethics
The authors followed international and institutional animal management guidelines for the experiments.
Consent for publication
All the authors of this article agree to the publication.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Amany Abbass
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Azhar, N., Yudiati, E. Outbreak simulation of Litopenaeus vannamei recovery rate with oral alginate and spirulina diet supplementation against Vibrio parahaemolyticus AHPND. Aquacult Int 31, 1659–1676 (2023). https://doi.org/10.1007/s10499-023-01050-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-023-01050-6