Abstract
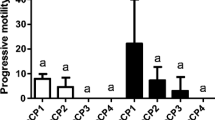
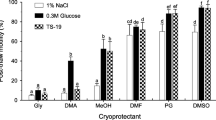
Although sperm cryopreservation is well developed, different methods can be used for freezing fish semen, mainly with the intention of reducing the use of liquid nitrogen. For this purpose, ultra-freezers could, among other advantages, facilitate the maintenance of semen compared to liquid nitrogen containers. Indeed, ultra-freezers have an increased capacity to store semen, and they can be available in places where there are no other technologies. The aim of this study was to develop an alternative protocol for freezing streaked prochilod Prochilodus lineatus semen using an ultra-freezer. The fish semen (n=10) was collected and frozen using an ultra-freezer (−80°C), with a solution containing 10% DMSO diluted in 5% Beltsville thawing solution in the proportion of 1:4 (semen/diluent) in 0.5-mL straws. Each week, a tube was randomly chosen, thawed, and used to evaluate seminal parameters using computerized semen analysis (CASA). Then, the data were subjected to analysis of variance followed by Tukey’s post hoc test. Motility, motility time, vitality, sperm velocity, and secondary morphology were influenced by storage in ultra-freezer (p < 0.05). However, these parameters were kept at levels considered adequate during the time tested. This is, to the best of our knowledge, the first report of frozen P. lineatus semen under ultra-freezer conditions (−80°C), demonstrating that this method is efficient in maintaining the semen quality for up to 10 weeks.

Similar content being viewed by others
Data availability
Available upon request.
References
Almeida-Monteiro PS, Oliveira-Araújo MS, Pinheiro RR et al (2017) Influence of vitamins C and E on the quality of cryopreserved semen Prochilodus brevis Prochilodontidae, Teleostei. Semin Ciências Agrárias 38:2669–2680. https://doi.org/10.5433/1679-0359.2017v38n4Supl1p2669
Assis IL, Palhares PC, Machado GJ et al (2019) Effect of melatonin on cryopreserved sperm of Prochilodus lineatus (Characiformes). Cryo Letters 40:152–158
Babiak I, Glogowski J, Luczynski MJ, Kucharczyk D, Luczynski M (1995) Cryopreservation of the milt of the northern pike. J Fish Biol 46:819–828. https://doi.org/10.1111/j.1095-8649.1995.tb01604.x
Babiak I, Glogowski J, Kujawa R, Kucharczyk D, Mamcarz A (1998) Cryopreservation of sperm from Asp Aspius aspius. Prog Fish Cult 60:146–148. https://doi.org/10.1577/1548-8640(1998)060<0146:COSFAA>2.0.CO;2
Batista M, Niño T, Alamo D et al (2009) Successful artificial insemination using semen frozen and stored by an ultrafreezer in the Majorera goat breed. Theriogenology 71:1307–1315. https://doi.org/10.1016/j.theriogenology.2008.12.024
Chen YK, Liu QH, Li J, Xiao ZZ, Xu SH, Shi XH, Ma DY (2010) Effect of long-term cryopreservation on physiological characteristics, antioxidant activities and lipid peroxidation of red seabream (Pagrus major) sperm. Cryobiology 61:189–193. https://doi.org/10.1016/j.cryobiol.2010.07.003
de Vazzoler AEAM (1996) Biologia da Reprodução de Peixes Teleósteos: Teoria e Prática. EDUEM, São Paulo, Maringá
Di Chiacchio IM, Almeida ILG, Leal MC, Viveiros ATM (2017) Sperm quality and its freezing ability throughout the spawning season in Prochilodus lineatus and Brycon orbignyanus. Theriogenology 90:284–288. https://doi.org/10.1016/j.theriogenology.2016.12.011
Dias GCM, Cassel M, Jesus LWO et al (2017) Spermatogonia, germline cells, and testicular organization in the Characiform Prochilodus lineatus studied using histological, stereological, and morphometric approaches. Anat Rec (Hoboken) 300:589–599. https://doi.org/10.1002/AR.23505
Egger RC, Motta NC, França TS et al (2021) Sperm cryopreservation of Prochilodus lineatus throughout the same reproductive season. Aquac Res 52:6453–6463. https://doi.org/10.1111/are.15513
Felizardo VO, Mello RA, de Andrade ES et al (2010) Levels of oxygen at reduced model for small yellow mandi (Pimelodus maculatus) in the funil hydroelectric plant. Bol do Inst Pesca 36:197–204
Figueroa E, Lee-Estevez M, Valdebenito I et al (2019) Effects of cryopreservation on mitochondrial function and sperm quality in fish. Aquaculture 511:634190. https://doi.org/10.1016/j.aquaculture.2019.06.004
Gallego V, Asturiano JF (2019) Fish sperm motility assessment as a tool for aquaculture research: a historical approach. Rev. Aquac. 11:697–724. https://doi.org/10.1111/raq.12253
Graham JE, Parks JK (1992) Effects of cryopreservation procedures on sperm membranes. Theriogenology 38:209–222. https://doi.org/10.1016/0093-691X(92)90231-F
Herranz-Jusdado JG, Gallego V, Morini M, Rozenfeld C, Pérez L, Müller T, Horváth Á, Ohta H, Asturiano JF (2019) Eels sperm cryopreservation: an overview. Theriogenology 133:210–215. https://doi.org/10.1016/j.theriogenology.2019.03.033
Judycka S, Nynca J, Ciereszko A (2019) Opportunities and challenges related to the implementation of sperm cryopreservation into breeding of salmonid fishes. Theriogenology 132:12–21. https://doi.org/10.1016/j.theriogenology.2019.03.022
Liu S, Su Y, Yi H, Liu X, Chen X, Lai H, Bi S, Zhang Y, Zhao X, Li G (2022) Ultra-low temperature cryopreservation and −80 °C storage of sperm from normal-male and pseudo-male Siniperca chuatsi. Aquaculture 553. https://doi.org/10.1016/j.aquaculture.2022.738007
Martínez-Páramo S, Horváth Á, Labbé C et al (2017) Cryobanking of aquatic species. Aquaculture 472:156–177. https://doi.org/10.1016/J.AQUACULTURE.2016.05.042
Miliorini AB, Murgas LDS, Rosa PV et al (2011) A morphological classification proposal for curimba (Prochilodus lineatus) sperm damages after cryopreservation. Aquac Res 42:177–187. https://doi.org/10.1111/j.1365-2109.2010.02575.x
Motta NC, Egger RC, Monteiro KS et al (2022) Effects of melatonin supplementation on the quality of cryopreserved sperm in the neotropical fish Prochilodus lineatus. Theriogenology 179:14–21. https://doi.org/10.1016/j.theriogenology.2021.11.012
Murgas LDS, Freitas RTF et al (2007) Cryopreservation of curimba (Prochilodus lineatus) semen after addition of different diluters, activators and cryoprotectants. Rev Bras Zootec 36:526–531
Paula DAJ, Murgas LDS, Castro TFD et al (2019) Effects of cooling rates on the quality of Prochilodus lineatus (Valenciennes, 1836) sperm. Reprod Domest Anim 54:1034–1043. https://doi.org/10.1111/RDA.13459
Pezo F, Cheuquemán C, Salinas P, Risopatrón J (2017) Freezing dog semen using −80 °C ultra-freezer: sperm function and in vivo fertility. Theriogenology 99:36–40. https://doi.org/10.1016/j.theriogenology.2017.05.007
Raspa M, Fray M, Paoletti R et al (2018) A new, simple and efficient liquid nitrogen free method to cryopreserve mouse spermatozoa at -80 °C. Theriogenology 119:52–59. https://doi.org/10.1016/j.theriogenology.2018.06.020
Sánchez R, Risopatrón J, Schulz M et al (2012) Vitrified sperm banks: the new aseptic technique for human spermatozoa allows cryopreservation at -86 °C. Andrologia 44:433–435. https://doi.org/10.1111/j.1439-0272.2012.01314.x
Sandoval-Vargas L, Silva Jiménez M, Risopatrón González J et al (2021) Oxidative stress and use of antioxidants in fish semen cryopreservation. Rev Aquac 13:365–387. https://doi.org/10.1111/raq.12479
Silva JMA, Murgas LDS, Felizardo VO, Pereira GJM, Navarro RD, Melo RA (2009) Características seminais e índices reprodutivos de curimba (Prochilodus lineatus) em diferentes períodos reprodutivos. Rev Bras Saúde e Produção Anim 10:668–677
Viveiros ATM, Maria AN, Orfão LH, et al (2008) Powder coconut water (ACP®) as extender for semen cryopreservation of Brazilian migratory fish species. In: Cybium. p 215. https://sfi-cybium.fr/en/node/876
Viveiros ATM, Nascimento AF, Orfão LH, Isaú ZA (2010) Motility and fertility of the subtropical freshwater fish streaked prochilod (Prochilodus lineatus) sperm cryopreserved in powdered coconut water. Theriogenology 74:551–556. https://doi.org/10.1016/j.theriogenology.2010.03.018
Yang H, Norris M, Winn R, Tiersch TR (2010) Evaluation of cryoprotectant and cooling rate for sperm cryopreservation in the euryhaline fish medaka Oryzias latipes. Cryobiology 61:211–219. https://doi.org/10.1016/j.cryobiol.2010.07.006
Yang S, Chen X, Fan B, et al (2021) Successful short term sperm cryopreservation in brown-marbled grouper (Epinephelus fuscoguttatus) with the utility of ultra-freezer (-80 °C). Reprod Domest Anim 1–6. https://doi.org/10.1111/rda.14072
Acknowledgements
The authors would like to express their gratitude to Ricardo José da Silva (Estação de Piscicultura da Usina Hidrelétrica da CEMIG) and Ivo Gavião Prado and Fabio Mineo Suzuki (Pisces – Consultoria e Serviços Ambientais) for providing the broodstock and team accommodation as well as Jailson M. Silva (Estação de Piscicultura da Usina Hidrelétrica da CEMIG) for assistance during animal manipulation.
Funding
This work was supported by funds from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) (grant number 88882.306651/2018-01), Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) (grant number APQ-01070-18), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (grant number 308359/2019-4).
Author information
Authors and Affiliations
Contributions
Daniella Aparecida de Jesus Paula: Lab work, methodology, investigation, project administration, resources, writing–original draft, writing–review and editing, and supervision experimental design. Naiara Melo: Lab work, writing–original draft, visualization data analysis, and draft editing. William Franco: Data analysis and writing–original draft. Pedro Uzeda: Sampling and sample analysis. Bruna Resende Chaves: Lab work, preparing all equipment, and sampling. Isaac Filipe Moreira Konig: visualization, writing–original draft, and writing–review and editing. Luis David Solis Murgas: Supervision, project administration, visualization, and writing–review and editing.
Corresponding authors
Ethics declarations
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed by the authors.
Consent for publication
All authors read and approved the final manuscript.
Competing interest
The authors declare no competing interests.
Additional information
Handling Editor: Gavin Burnell
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Jesus Paula, D.A., Melo, N., Carneiro, W.F. et al. Cryopreservation of semen in streaked prochilod (Prochilodus lineatus): use of ultra-freezer as alternative technique to liquid nitrogen. Aquacult Int 30, 2629–2639 (2022). https://doi.org/10.1007/s10499-022-00926-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-022-00926-3