Abstract
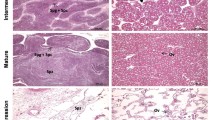
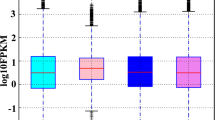
Sex regulation of bivalves is a complex process, and there has also been little research on their sex-determining genes. To better understand the sex determination/differentiation of bivalves, we analyzed the gonadal proteomics of Hyriopsis cumingii. We identified 673 peptides and 76 proteins. The 35 differentially expressed proteins identified included 19 upregulated and 16 downregulated proteins. FEM-1B protein associated with sex determination showed female-specific expression; the function of fem-1b gene was explored. The full-length cDNA of fem-1b cloned by RACE was 2105 bp. Quantitative PCR (qPCR) was performed on male and female tissues and early developmental stages; the results showed that fem-1b was expressed in both male and female tissues, and its expression level in gonads was significantly higher in females than in males (P < 0.01). In early embryonic development, the fem-1b mRNA expression increased significantly on the third day after fertilization (cleavage stage). Results of in situ hybridization showed a strong signal on oocyte membranes and no signal in male gonads. Our results indicate that fem-1b plays a certain role in sex determination and oocyte development of H. cumingii. This study provides valuable resources for understanding the mechanism and pathway of sex determination/differentiation of H. cumingii.






Similar content being viewed by others
Data availability
All data and materials as well as software application support their published claims and comply with field standards.
References
Arnaud-Haond S, Goyard E, Vonau V, Herbaut C, Prou J, Saulnier D (2007) Pearl formation: persistence of the graft during the entire process of biomineralization. Mar Biotechnol 9:113–116. https://doi.org/10.1007/s10126-006-6033-5
Barnes TM, Hodgkin J (1996) The tra-3 sex determination gene of Caenorhabditis elegans encodes a member of the calpain regulatory protease family. EMBO J 15:4477–4484
Cao LQ, Kenchington E, Zouros E (2004) Differential segregation patterns of sperm mitochondria in embryos of the blue mussel (Mytilus edulis). Genetics 166:883–894. https://doi.org/10.1534/genetics.166.2.883
Castelli MA, Whiteley SL, Georges A, Holleley CE (2020) Cellular calcium and redox regulation: the mediator of vertebrate environmental sex determination? Biol Rev 95:680–695. https://doi.org/10.1111/brv.12582
Chan SL, Yee KSY, Tan KML, Yu VC (2000) The Caenorhabditis elegans sex determination protein FEM-1 is a CED-3 substrate that associates with CED-4 and mediates apoptosis in mammalian cells. J Biol Chem 275:17925–17928. https://doi.org/10.1074/jbc.c000146200
Chen Y, Wang YY, Wang GL, He FS, Li JL (2008) Full-length cDNA cloning of M and F-type COII genes and expression in different age freshwater mussel Hyriopsis cumingii. J Fish China 42:29–38. https://doi.org/10.11964/jfc.20161210644
Chen JT, Zhou AG, Xie SL, Wang C, Lv ZJ, Zou JX (2016) Comparative proteomic identification of mature and immature sperm in the catfish Cranoglanis bouderius. PLoS One 11:e0151254. https://doi.org/10.1371/journal.pone.0151254
Doniach T, Hodgkin J (1984) A sex-determining gene, fem-1, required for both male and hermaphrodite development in Caenorhabditis elegans. Dev Biol 106:223–235. https://doi.org/10.1016/0012-1606(84)90077-0
Fabioux C, Huvet A, Souchu LP, Pennec LM, Pouvreau S (2005) Temperature and photoperiod drive Crassostrea gigas reproductive internal clock. Aquaculture 250:458–470. https://doi.org/10.1016/j.aquaculture.2005.02.038
Farlora R, Araya-Garay J, Gallardo-Escárate C (2014) Discovery of sex-related genes through high-throughput transcriptome sequencing from the salmon louse Caligus rogercresseyi. Mar Genomics 15:85–93. https://doi.org/10.1016/j.margen.2014.02.005
Ge CM, Lu WQ, Chen AQ (2017) Quantitative proteomic reveals the dynamic of protein profile during final oocyte maturation in zebrafish. Biochem Biophys Res Commun 490:657–663. https://doi.org/10.1016/j.bbrc.2017.06.093
Gilder AS, Chen YB, Iii RJJ, Maher JF (2013) Fem1b promotes ubiquitylation and suppresses transcriptional activity of Gli1. Biochem Biophys Res Commun 3:431–436. https://doi.org/10.1016/j.bbrc.2013.09.090
Guo X, Hedgecock D, Hershberger WK, Cooper K, Allen K, JR A (1998) Genetic determinants of protandric sex in the pacific oyster, Crassostrea gigas Thunberg. Evolution 52:394–402. https://doi.org/10.1111/j.1558-5646.1998.tb01640.x
He Y, Fang J, Xue LY, Wu JJ, Dawar FU, Mei J (2017) Potential contributions of heatshock proteins and related genes in sexual differentiation in yellow catfish (Pelteobagrus fulvidraco). Fish Physiol Biochem 43:465–475. https://doi.org/10.1007/s10695-016-0303-6
Hedrick PW, Hedgecock D (2010) Sex determination: genetic models for oysters. J Hered 101:602–611. https://doi.org/10.1093/jhered/esq065
Hu BQ, Xiao JY, Yi PP, Hu CX, Zhu MX, Yin SY, Wen CG, Wu JL (2019) Cloning and characteristic of MMP1 gene from Hyriopsis cumingii and collagen hydrolytic activity of its recombinant protein. Gene 693:92–100. https://doi.org/10.1016/j.gene.2018.12.087
Kang YS, Xie HB, Zhao CT (2019) Ankrd45 is a novel ankyrin repeat protein required for cell proliferation. Genes 10:462. https://doi.org/10.3390/genes10060462
Kimble J, Edgar L, Hirsh D (1984) Specification of male development in Caenorhabditis elegans: the fem genes. Developmental Biology 105:234–239. https://doi.org/10.1016/0012-1606(84)90279-3
Kohno S, Katsu Y, Urushitani H, Ohta Y, Lguchi T, Guillette LJ Jr (2010) Potential contributions of heat shock proteins to temperature-dependent sex determination in the American alligator. Sex Development 4:73–87. https://doi.org/10.1159/000260374
Kuwabara PE (2007) A complex solution to a sexual dilemma. Dev Cell 13:0–8. https://doi.org/10.1016/j.devcel.2007.06.004
Li YP, Zhang LL, Sun Y, Ma XL, Wang J, Li RJ, Zhang MW, Wang S, Hu XL, Bao ZM (2016) Transcriptome sequencing and comparative analysis of ovary and testis identifies potential key sex-related genes and pathways in scallop Patinopecten yessoensis. Mar Biotechnol 18:453–465. https://doi.org/10.1007/s10126-016-9706-8
Luo L, Zhang Q, Kong X, Huang HQ, Ke CH (2017) Differential effects of bisphenol a toxicity on oyster (Crassostrea angulata) gonads as revealed by label-free quantitative proteomics. Chemosphere 176:305–314. https://doi.org/10.1016/j.chemosphere.2017.02.146
Ma KY, Liu ZQ, Lin JY, Li JL, Qiu GF (2016) Molecular characterization of a novel ovary-specific gene fem-1 homolog from the oriental river prawn, Macrobrachium nipponense. Gene 575:244–252. https://doi.org/10.1016/j.gene.2015.08.070
Mosavi LK, Cammett TJ, Desrosiers DC, Peng ZY (2004) The ankyrin repeat as molecular architecture for protein recognition. Protein Sci 13:1435–1448. https://doi.org/10.1110/ps.03554604
Oyhenart J, Benichou S, Raich N (2005) Putative Homeodomain transcription factor 1 interacts with the feminization factor homolog Fem1b in male germ cells. Biol Reprod 72:780–787. https://doi.org/10.1095/biolreprod.104.035964
Pan BB, Li JL, Bai ZY (2010) Histological study on ovarian development and oogenesis of Hyriopsis cumingii cultured in pond. Journal of Shanghai Ocean University 04:23–27. https://doi.org/10.3724/SP.J.1231.2010.06781
Pavel GT, Claudia VL, Raúl LH, Ana MI (2019) A natural antisense transcript of the fem-1 gene was found expressed in female gonads during the characterization, expression profile, and cellular localization of the fem-1 gene in pacific white shrimp penaeus vannamei. Gene 706:19–31. https://doi.org/10.1016/j.gene.2019.04.066
Rahman NMA, Fu H, Qiao H, Jin S, Bai H, Zhang W, Jiang FW, Liang G, Sun S, Gong Y (2016) Molecular cloning and expression analysis of Fem1b from oriental river prawn Macrobrachium nipponense. Genet Mol Res 15:2. https://doi.org/10.4238/gmr.15027950
Shi H, Hao YJ, Chen B, Si FL, Wang P, He ZB (2013) Cloning and expression analysis of fem-1 gene from the oriental migratory locust, Locusta migratoria manilensis (Orthoptera: Locustidae). Acta Entomol Sin 56:729–737
Shi JW, Hong YJ, Sheng JQ, Peng K, Wang JH (2015) De novo transcriptome sequencing to identify the sex-determination genes in Hyriopsis schlegelii. Biosci Biotechnol Biochem 79:1257–1265. https://doi.org/10.1080/09168451.2015.1025690
Shi Y, Liu WG, He MX (2018) Proteome and transcriptome analysis of ovary, intersex gonads, and testis reveals potential key sex reversal/differentiation genes and mechanism in scallop Chlamys nobilis. Mar Biotechnol 20:220–245. https://doi.org/10.1007/s10126-018-9800-1
Siegfried KR, Nüsslein-Volhard C (2008) Germ line control of female sex determination in zebrafish. Dev Biol 324:277–287. https://doi.org/10.1016/j.ydbio.2008.09.025
Song CW, Cui ZX, Hui M, Liu Y, Li YD (2015) Molecular characterization and expression profile of three Fem-1 genes in Eriocheir sinensis provide a new insight into crab sex-determining mechanism. Comp Biochem Physiol B Biochem Mol Biol 189:6–14. https://doi.org/10.1074/jbc.c000146200
Spence AM, Coulson A, Hodgkin J (1990) The product of fem-1, a nematode sex-determining gene, contains a motif found in cell cycle control proteins and receptors for cell-cell interactions. Cell 60:981–990. https://doi.org/10.1016/0092-8674(90)90346-g
Starostina NG, Lim JM, Schvarzstein M, Wells L, Spence AM, Kipreos ET (2007) A CUL-2 ubiquitin ligase containing three FEM proteins degrades TRA-1 to regulate C. elegans sex determination. Dev Cell 13:127–139. https://doi.org/10.1016/j.devcel.2007.05.008
Stefania G, Barbara L, Grazia B, Maura B, Daniela P, Tonina R, Sergio U, Roberto A, Maria FA (2016) Proteomic dataset of Paracentrotus lividus gonads of different sexes and at different maturation stages. Data in brief 8:824–827 https://doi.org/10.1016/j.jprot.2016.05.035
Subauste MC, Sansom OJ, Porecha N, Raich N, Du LQ, Maher JF (2010) Fem1b, a proapoptotic protein, mediates proteasome inhibitor-induced apoptosis of human colon cancer cells. Mol Carcinog 49(2):105–113. https://doi.org/10.1002/mc.20594
Tan KML, Chan SL, Tan KO, Yu VC (2001) The Caenorhabditis elegans sex-determining protein FEM-2 and its human homologue, hFEM-2, are Ca2+/calmodulin-dependent protein kinase phosphatases that promote apoptosis. The Jouranl of Biological Chemistry 276:44193–44202. https://doi.org/10.1074/jbc.m105880200
Teaniniuraitemoana V, Huvet A, Levy P, Klopp C, Lhuillier E, Gaertner-Mazouni N, Gueguen Y, Moullac GL (2014) Gonad transcriptome analysis of pearl oyster Pinctada margaritifera: identification of potential sex differentiation and sex determining genes. BMC Genomics 15:491. https://doi.org/10.1186/1471-2164-15-491
Teaniniuraitemoana V, Leprêtre M, Levy P, Vanaa V, Parrad S, Mazouni GN, Gueguen Y, Huvet A, Moullac LG (2016) Effect of temperature, food availability, and estradiol injection on gametogenesis and gender in the pearl oyster, Pinctada margaritifera. J Exp Zool A Ecol Genet Physiol 325:13–24. https://doi.org/10.1002/jez.1992
Ventura-Holman T, Lu DY, Si XH, Izevbigie EB, Maher JF (2003) The Fem1c genes: conserved members of the Fem1 gene family in vertebrates. Gene 314:133–139. https://doi.org/10.1016/s0378-1119(03)00712-1
Wang H, Bai ZY, Li JL, Wang JJ (2007) A primary study on the morphological changes of Hyriopsis cumingii embryos in nurturing pouch of outer gill. J Shanghai Fish Univ 03:219–223. https://doi.org/10.3969/j.issn.1004-7271
Wang YY, Wu CD, Guo PF, Wang GL, Li JL (2018) Molecular characterization and expression of the feminization-1c (fem-1c) in the freshwater mussel (Hyriopsis cumingii). Aquaculture and Fisheries 3:6–13. https://doi.org/10.1016/j.aaf.2017.10.005
Wilkins MR, Pasquali C, Appel RD, Ou K, Golaz O, Sanchez JC, Yan JX, Gooley AA, Hughes G, Humphery-Smith L (1996) From proteins to proteomes: large scale protein identification by two-dimensional electrophoresis and amino acid analysis. Nat Biotechnol 14:61–65. https://doi.org/10.1038/nbt0196-61
Xi W, Nishita D, Ya-Ping H, Sandy MP, Cory AS, Michael MS (2018) Mouse Fem1b interacts with the Nkx3.1 homeoprotein and is required for proper male secondary sexual development. Dev Dyn 237:2963–2972. https://doi.org/10.1002/dvdy.21694
Zarkower D, Hodgkin J (1992) Molecular analysis of the C. elegans sex-determining gene tra-1: a gene encoding two zinc finger proteins. Cell 70:237–249. https://doi.org/10.1016/0092-8674(92)90099-x
Zhang Y, Zhao HF, Wang J, Ge JP, Li Y, Gu JK, Li P, Feng Y, Yang MJ (2013) Structural insight into Caenorhabditis elegans sex-determining protein FEM-2. J Biol Chem 288:22058–22066. https://doi.org/10.1074/jbc.m113.464339
Zhang N, Xu F, Guo X (2014) Genomic analysis of the pacific oyster (Crassostrea gigas) reveals possible conservation of vertebrate sex determination in a Mollusc. G3 & 58 Genesgenetics 4:2207–2217. https://doi.org/10.1534/g3.114.013904
Zhou ZY, Li Q, Yu H, Kong LF (2018) Cloning and expression analysis of Fem-1 gene of Pacific oyster (Crassostrea gigas). Periodical of Ocean University of China 48(6):45–54. https://doi.org/10.16441/j.cnki.hdxb.20170186
Zhou LX, Liu X, Ye BQ, Liu Y, Tan SP, Ma KY, Qiu GF (2020) Molecular characterization of ovary-specific gene Mrfem-1 and siRNA-mediated regulation on targeting Mrfem-1 in the giant freshwater prawn, Macrobrachium rosenbergii. Gene 754:144891. https://doi.org/10.1016/j.gene.2020.144891
Acknowledgments
We would like to thank the native English speaking scientists of Elixigen Company (Huntington Beach, California) for editing our manuscript.
Code availability
Not applicable.
Funding
This study was supported by the National Key R&D Program of China (grant number 2018YFD0901406) and the National Natural Science Foundation of China (grant number 31772835).
Author information
Authors and Affiliations
Contributions
Y.-Y. W. conceived and designed the experiments; S.-H. D. completed the sample collection. Y.-Y. W. and S.-S. D. performed the experiments; Y.-Y. W. and X.-Y. C. analyzed the data; Y.-Y. W. wrote the paper. G.-L. W. and J.-L.L. provided feedback on discussion and results. All authors have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
All handling of H. cumingii were conducted in accordance with guidelines on the care and use of animals for scientific purposes set up by the Institutional Animal Care and Use Committee (IACUC) of Shanghai Ocean University, Shanghai, China.
Consent to participate
All the authors agree to participate in this experiment.
Consent for publication
All the authors of this article agree to the publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, YY., Duan, SH., Dong, SS. et al. Comparative proteomic study on fem-1b in female and male gonads in Hyriopsis cumingii. Aquacult Int 29, 1–18 (2021). https://doi.org/10.1007/s10499-020-00605-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-020-00605-1