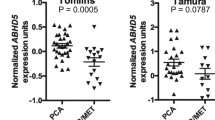
Abstract
Prostate cancer (PCa) is one of the most common cancers affecting the health of men worldwide. Castration-resistant prostate cancer (CRPC), the advanced and refractory phase of prostate cancer, has multiple mechanisms of resistance to androgen deprivation therapy (ADT) such as AR mutations, aberrant androgen synthase, and abnormal expression of AR-related genes. Based on the research of the AR pathway, new drugs for the treatment of CRPC have been developed in clinical practice, such as Abiraterone and enzalutamide. However, many areas in this pathway are still worth exploring. In this study, single-cell sequencing analysis was utilized to scrutinize significant genes in the androgen receptor (AR) pathway related to CRPC. Our analysis of single-cell sequencing combined with bulk-cell sequencing revealed a substantial downregulation of AR-regulated AFF3 in CRPC. Overexpression of AFF3 restricted the proliferation and migration of prostate cancer cells whilst also increasing their sensitivity towards enzalutamide, while knockdown of AFF3 had the opposite effect. To elucidate the mechanism of tumor inhibition by AFF3, we applied GSVA and GSEA to investigate the metabolic pathways related to AFF3 and revealed that AFF3 had an impact on fatty acids metabolism and ferroptosis through the regulation of ACSL4 protein expression. Based on correlation analysis and flow cytometry, we can speculate that AFF3 can impact the sensitivity of the CRPC cell lines to the ferroptosis inducer (RSL3) by regulating ACSL4. Therefore, our findings may provide new insights into the mechanisms of drug resistance in CRPC, and AFF3 may serve as a novel prognostic biomarker in prostate cancer.







Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
Abbreviations
- PCa:
-
Prostate cancer
- HSPC:
-
Hormone-sensitive prostate cancer
- CRPC:
-
Castration-resistant prostate cancer
- ADT:
-
Androgen deprivation therapy
- AR:
-
Androgen receptor
- RP:
-
Radical prostatectomy
- MSKCC:
-
The Memorial Sloan Kettering Cancer Center
- GSEA:
-
Gene set enrichment analysis
- GSVA:
-
Gene set variation analysis
- DHT:
-
Dihydrotestosterone
- ChIP:
-
Chromatin immunoprecipitation
- RFS:
-
Recurrence-free survival
- ROC:
-
Time-dependent receiver operating characteristic
- AUC:
-
Area under the curve
- DEGs:
-
Differential expression genes
References
Siegel RL, Miller KD, Wagle NS, Jemal A (2023) Cancer statistics, 2023. CA Cancer J Clin 73(1):17–48
Rebello RJ, Oing C, Knudsen KE, Loeb S, Johnson DC, Reiter RE et al (2021) Prostate cancer. Nat Rev Dis Primers 7(1):9
Van den Broeck T, van den Bergh RCN, Arfi N, Gross T, Moris L, Briers E et al (2019) Prognostic value of biochemical recurrence following treatment with curative intent for prostate Cancer: a systematic review. Eur Urol 75(6):967–987
Zhang Y, Fan A, Li Y, Liu Z, Yu L, Guo J et al (2023) Single-cell RNA sequencing reveals that HSD17B2 in cancer-associated fibroblasts promotes the development and progression of castration-resistant prostate cancer. Cancer Lett 566:216244
Fan A, Zhang Y, Cheng J, Li Y, Chen W (2022) A novel prognostic model for prostate cancer based on androgen biosynthetic and catabolic pathways. Front Oncol 12:950094
Hong M, Tao S, Zhang L, Diao LT, Huang X, Huang S et al (2020) RNA sequencing: new technologies and applications in cancer research. J Hematol Oncol 13(1):166
Stubbington MJT, Rozenblatt-Rosen O, Regev A, Teichmann SA (2017) Single-cell transcriptomics to explore the immune system in health and disease. Science 358(6359):58–63
Price DK, Chau CH, Till C, Goodman PJ, Leach RJ, Johnson-Pais TL et al (2016) Association of androgen metabolism gene polymorphisms with prostate cancer risk and androgen concentrations: results from the prostate Cancer Prevention Trial. Cancer 122(15):2332–2340
Kruse U, Pallasch CP, Bantscheff M, Eberhard D, Frenzel L, Ghidelli S et al (2011) Chemoproteomics-based kinome profiling and target deconvolution of clinical multi-kinase inhibitors in primary chronic lymphocytic leukemia cells. Leukemia 25(1):89–100
Ma C, Staudt LM (1996) LAF-4 encodes a lymphoid nuclear protein with transactivation potential that is homologous to AF-4, the gene fused to MLL in t(4;11) leukemias. Blood 87(2):734–745
Shi Y, Zhao Y, Zhang Y, AiErken N, Shao N, Ye R et al (2018) AFF3 upregulation mediates tamoxifen resistance in breast cancers. J Exp Clin Cancer Res 37(1):254
Stahl EA, Raychaudhuri S, Remmers EF, Xie G, Eyre S, Thomson BP et al (2010) Genome-wide association study meta-analysis identifies seven new rheumatoid arthritis risk loci. Nat Genet 42(6):508–514
Bowes J, Ho P, Flynn E, Ali F, Marzo-Ortega H, Coates LC et al (2012) Comprehensive assessment of rheumatoid arthritis susceptibility loci in a large psoriatic arthritis cohort. Ann Rheum Dis 71(8):1350–1354
Tsukumo SI, Subramani PG, Seija N, Tabata M, Maekawa Y, Mori Y et al (2022) AFF3, a susceptibility factor for autoimmune diseases, is a molecular facilitator of immunoglobulin class switch recombination. Sci Adv 8(34):eabq0008
Chen X, Li J, Kang R, Klionsky DJ, Tang D (2021) Ferroptosis: machinery and regulation. Autophagy 17(9):2054–2081
Wei X, Yi X, Zhu XH, Jiang DS (2020) Posttranslational modifications in Ferroptosis. Oxid Med Cell Longev 2020:8832043
Zhang HL, Hu BX, Li ZL, Du T, Shan JL, Ye ZP et al (2022) PKCbetaII phosphorylates ACSL4 to amplify lipid peroxidation to induce ferroptosis. Nat Cell Biol 24(1):88–98
Yang Y, Liu T, Hu C, Xia H, Liu W, Chen J et al (2021) Ferroptosis inducer erastin downregulates androgen receptor and its splice variants in castration–resistant prostate cancer. Oncol Rep 45(4):1–1
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA et al (2012) The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2(5):401–404
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W et al (2015) Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43(7):e47
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA et al (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A 102(43):15545–15550
Hanzelmann S, Castelo R, Guinney J (2013) GSVA: gene set variation analysis for microarray and RNA-seq data. BMC Bioinformatics 14:7
Zeng D, Ye Z, Shen R, Yu G, Wu J, Xiong Y et al (2021) IOBR: Multi-omics Immuno-Oncology Biological Research to Decode Tumor Microenvironment and signatures. Front Immunol 12:687975
Beltran H, Beer TM, Carducci MA, de Bono J, Gleave M, Hussain M et al (2011) New therapies for castration-resistant prostate cancer: efficacy and safety. Eur Urol 60(2):279–290
Schweizer MT, Yu EY (2015) Persistent androgen receptor addiction in castration-resistant prostate cancer. J Hematol Oncol 8:128
Castellanos E, Feld E, Horn L (2017) Driven by mutations: the Predictive Value of Mutation Subtype in EGFR-Mutated Non-small Cell Lung Cancer. J Thorac Oncol 12(4):612–623
Li B, Severson E, Pignon JC, Zhao H, Li T, Novak J et al (2016) Comprehensive analyses of tumor immunity: implications for cancer immunotherapy. Genome Biol 17(1):174
Diaz-Montero CM, Rini BI, Finke JH (2020) The immunology of renal cell carcinoma. Nat Rev Nephrol 16(12):721–735
Tang D, Chen X, Kang R, Kroemer G (2021) Ferroptosis: molecular mechanisms and health implications. Cell Res 31(2):107–125
Bayir H, Dixon SJ, Tyurina YY, Kellum JA, Kagan VE (2023) Ferroptotic mechanisms and therapeutic targeting of iron metabolism and lipid peroxidation in the kidney. Nat Rev Nephrol 19(5):315–336
Jiang X, Stockwell BR, Conrad M (2021) Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol 22(4):266–282
Hou J, Jiang C, Wen X, Li C, Xiong S, Yue T et al (2022) ACSL4 as a potential target and biomarker for Anticancer: from Molecular mechanisms to clinical therapeutics. Front Pharmacol 13:949863
Jia B, Li J, Song Y, Luo C (2023) ACSL4-mediated ferroptosis and its potential role in central nervous system diseases and injuries. Int J Mol Sci 24(12):10021
Ross RK, Coetzee GA, Pearce CL, Reichardt JK, Bretsky P, Kolonel LN et al (1999) Androgen metabolism and prostate cancer: establishing a model of genetic susceptibility. Eur Urol 35(5–6):355–361
Aus G, Abbou CC, Bolla M, Heidenreich A, Schmid HP, van Poppel H et al (2005) EAU guidelines on prostate cancer. Eur Urol 48(4):546–551
Scher HI, Buchanan G, Gerald W, Butler LM, Tilley WD (2004) Targeting the androgen receptor: improving outcomes for castration-resistant prostate cancer. Endocr Relat Cancer 11(3):459–476
Mohler JL, Gregory CW, Ford OH 3rd, Kim D, Weaver CM, Petrusz P et al (2004) The androgen axis in recurrent prostate cancer. Clin Cancer Res 10(2):440–448
Titus MA, Schell MJ, Lih FB, Tomer KB, Mohler JL (2005) Testosterone and dihydrotestosterone tissue levels in recurrent prostate cancer. Clin Cancer Res 11(13):4653–4657
Locke JA, Guns ES, Lubik AA, Adomat HH, Hendy SC, Wood CA et al (2008) Androgen levels increase by intratumoral de novo steroidogenesis during progression of castration-resistant prostate cancer. Cancer Res 68(15):6407–6415
Locke JA, Nelson CC, Adomat HH, Hendy SC, Gleave ME, Guns ES (2009) Steroidogenesis inhibitors alter but do not eliminate androgen synthesis mechanisms during progression to castration-resistance in LNCaP prostate xenografts. J Steroid Biochem Mol Biol 115(3–5):126–136
Tan YF, Zhang Y, Ge SY, Zhong F, Sun CY, Xia GW (2022) AR-regulated ZIC5 contributes to the aggressiveness of prostate cancer. Cell Death Discov 8(1):393
Baslan T, Hicks J (2017) Unravelling biology and shifting paradigms in cancer with single-cell sequencing. Nat Rev Cancer 17(9):557–569
Shen MM, Abate-Shen C (2010) Molecular genetics of prostate cancer: new prospects for old challenges. Genes Dev 24(18):1967–2000
Tan RJ, Gibbons LJ, Potter C, Hyrich KL, Morgan AW, Wilson AG et al (2010) Investigation of rheumatoid arthritis susceptibility genes identifies association of AFF3 and CD226 variants with response to anti-tumour necrosis factor treatment. Ann Rheum Dis 69(6):1029–1035
Risbridger GP, Davis ID, Birrell SN, Tilley WD (2010) Breast and prostate cancer: more similar than different. Nat Rev Cancer 10(3):205–212
Sikora MJ, Cordero KE, Larios JM, Johnson MD, Lippman ME, Rae JM (2009) The androgen metabolite 5alpha-androstane-3beta,17beta-diol (3betaAdiol) induces breast cancer growth via estrogen receptor: implications for aromatase inhibitor resistance. Breast Cancer Res Treat 115(2):289–296
Giunchi F, Fiorentino M, Loda M (2019) The Metabolic Landscape of prostate Cancer. Eur Urol Oncol 2(1):28–36
Swinnen JV, Van Veldhoven PP, Esquenet M, Heyns W, Verhoeven G (1996) Androgens markedly stimulate the accumulation of neutral lipids in the human prostatic adenocarcinoma cell line LNCaP. Endocrinology 137(10):4468–4474
Zaidi N, Lupien L, Kuemmerle NB, Kinlaw WB, Swinnen JV, Smans K (2013) Lipogenesis and lipolysis: the pathways exploited by the cancer cells to acquire fatty acids. Prog Lipid Res 52(4):585–589
Cai C, Chen S, Ng P, Bubley GJ, Nelson PS, Mostaghel EA et al (2011) Intratumoral de novo steroid synthesis activates androgen receptor in castration-resistant prostate cancer and is upregulated by treatment with CYP17A1 inhibitors. Cancer Res 71(20):6503–6513
Lv Z, Wang J, Wang X, Mo M, Tang G, Xu H et al (2021) Identifying a ferroptosis-related gene signature for Predicting biochemical recurrence of prostate Cancer. Front Cell Dev Biol 9:666025
Maccarinelli F, Coltrini D, Mussi S, Bugatti M, Turati M, Chiodelli P et al (2023) Iron supplementation enhances RSL3-induced ferroptosis to treat naive and prevent castration-resistant prostate cancer. Cell Death Discov 9(1):81
Nassar ZD, Mah CY, Dehairs J, Burvenich IJ, Irani S, Centenera MM et al (2020) Human DECR1 is an androgen-repressed survival factor that regulates PUFA oxidation to protect prostate tumor cells from ferroptosis. Elife 9:e54166
Wang ME, Chen J, Lu Y, Bawcom AR, Wu J, Ou J et al (2023) RB1-deficient prostate tumor growth and metastasis are vulnerable to ferroptosis induction via the E2F/ACSL4 axis. J Clin Invest. https://doi.org/10.1172/JCI166647
Acknowledgements
None.
Author information
Authors and Affiliations
Contributions
WC and ZM performed the study concept and design; AF and YL performed the development of methodology and writing, review, and revision of the paper; AF, YZ, and YL provided acquisition, analysis, and interpretation of data, and statistical analysis; WM, MC and WP provided technical and material support. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This study was approved by the Ethics Committee of Zhongshan Hospital, Fudan University (Approval No: B2020-351R) and written informed consent was obtained from all patients.
Consent to participate
All animal experiments and protocols were reviewed and approved by the Animal Care and Use Committee of Shanghai University.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fan, A., Li, Y., Zhang, Y. et al. Loss of AR-regulated AFF3 contributes to prostate cancer progression and reduces ferroptosis sensitivity by downregulating ACSL4 based on single-cell sequencing analysis. Apoptosis (2024). https://doi.org/10.1007/s10495-024-01941-w
Accepted:
Published:
DOI: https://doi.org/10.1007/s10495-024-01941-w