Abstract
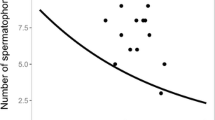
Neoseiulus californicus is an important biological control agent of small arthropod pests, widely used in preventive releases at low prey densities. Therefore, it is important to study reproductive investment of this species, both females and males, under environmental pressure of limited prey availability. Laboratory experiments were performed to investigate N. californicus male reproductive investment after short-time fasting or long-term starvation. When fasted for 36 h, males are still able to complete a single mating with no significant change in reproductive parameters of its mate. But the total mating duration became 24% longer, especially timing for the male to fill the first spermatheca of its mate increased by 35%. If daily food availability was less than 20% of demand, males were still able to mate until the end of life, but their fertility reduced after ca. the seventh mating. Mating of some males at the end of their lives are invalid, with their mates failing to oviposit. Overall, we summarized that reproductive investment demand for a single mating was low in N. californicus males, but its priority in energy allocation was high. When starved, males tried to guarantee their mating capability, but their fertility decreased gradually.








Similar content being viewed by others
References
Amano H, Chant DA (1979) Mating behaviour and reproductive mechanisms of two species of predacious mites, Phytoseiulus persimilis Athias-Henriot and Amblyseius andersoni (Chant) (Acarina: Phytoseiidae). Acarologia 20(2):196–213. https://doi.org/10.1007/978-94-011-7918-8_1
Arnqvist G, Nilsson T (2000) The evolution of polyandry: multiple mating and female fitness in insects. Anim Behav 60(2):145–164. https://doi.org/10.1006/anbe.2000.1446
Avila FW, Sirot LK, LaFlamme BA, Rubinstein CD, Wolfner MF (2011) Insect seminal fluid proteins: identification and function. Annu Rev Entomol 56:21–40. https://doi.org/10.1146/annurev-ento-120709-144823
Barber A, Campbell CAM, Crane H, Lilley R, Tregidga E (2003) Biocontrol of two-spotted spider mite Tetranychus urticae on dwarf hops by the phytoseiid mites Phytoseiulus persimilis and Neoseiulus californicus. Biocontrol Sci Tech 13(3):275–284. https://doi.org/10.1080/0958315031000110300
Bateman AJ (1948) Intra-sexual selection in Drosophila. Heredity 2(3):349–368. https://doi.org/10.1038/hdy.1948.21
Calow P (1983) Energetics of reproduction and its evolutionary implications. Biol J Lin Soc 20(2):153–165. https://doi.org/10.1111/j.1095-8312.1983.tb00359.x
Calow P (1985) Adaptive aspects of energy allocation. Fish energetics. Springer, Dordrecht, pp 13–31
Crean AJ, Bonduriansky R (2014) What is a paternal effect? Trends Ecol Evol 29(10):554–559. https://doi.org/10.1016/j.tree.2014.07.009
Daly M (1978) The cost of mating. Am Nat 112(986):771–774. https://doi.org/10.1086/283319
Dehghani-Tafti H, Golpayegani AZ, Saboori A, Krey KL (2015) The effect of mating experience, age and territoriality on the male mating competition in Phytoseiulus persimilis and Neoseiulus californicus (Acari: Phytoseiidae). Persian J Acarol. https://doi.org/10.22073/pja.v4i1.10196
Escudero L, Ferragut F (2005) Life-history of predatory mites Neoseiulus californicus and Phytoseiulus persimilis (Acari: Phytoseiidae) on four spider mite species as prey, with special reference to Tetranychus evansi (Acari: Tetranychidae). Biol Control 32(3):378–384. https://doi.org/10.1016/j.biocontrol.2004.12.010
Fowler K, Partridge L (1989) A cost of mating in female fruitflies. Nature 338(6218):760–761. https://doi.org/10.1038/338760a0
Fox C (1993) Multiple mating, lifetime fecundity and female mortality of the bruchid beetle, Callosobruchus maculatus (Coleoptera: Bruchidae). Funct Ecol. https://doi.org/10.2307/2389888
Gomez-Diaz C, Benton R (2013) The joy of sex pheromones. EMBO Rep 14(10):874–883. https://doi.org/10.1038/embor.2013.140
Gotoh T, Tsuchiya A (2008) Effect of multiple mating on reproduction and longevity of the phytoseiid mite Neoseiulus californicus. Exp Appl Acarol 44(3):185–197. https://doi.org/10.1007/s10493-008-9143-0
Gotoh T, Yamaguchi K, Mori K (2004) Effect of temperature on life history of the predatory mite Amblyseius (Neoseiulus) californicus (Acari: Phytoseiidae). Exp Appl Acarol 32(1):15–30
Gotoh T, Tsuchiya A, Kitashima Y (2006) Influence of prey on developmental performance, reproduction and prey consumption of Neoseiulus californicus (Acari: Phytoseiidae). Exp Appl Acarol 40(3):189–204. https://doi.org/10.1007/s10493-006-9032-3
Hamilton WD (1967) Extraordinary sex ratios. Science 156:477–488. https://doi.org/10.1126/science.156.3774.477
Heifetz Y, Lung O, Frongillo EA Jr, Wolfner MF (2000) The Drosophila seminal fluid protein acp26aa stimulates release of oocytes by the ovary. Curr Biol 10(2):99–102. https://doi.org/10.1016/S0960-9822(00)00288-8
Jacot A, Scheuber H, Brinkhof MW (2004) Costs of an induced immune response on sexual display and longevity in field crickets. Evolution 58(10):2280–2286. https://doi.org/10.1554/03-660
Janssen A, Sabelis MW (1992) Phytoseiid life-histories, local predator-prey dynamics, and strategies for control of tetranychid mites. Exp Appl Acarol 14:233–250. https://doi.org/10.1007/BF01200566
Jervis MA, Ellers J, Harvey JA (2008) Resource acquisition, allocation, and utilization in parasitoid reproductive strategies. Annu Rev Entomol 53:361–385. https://doi.org/10.1146/annurev.ento.53.103106.093433
Jiang XH, Lv JL, Wang ED, Xu XN (2019) Spermatophore producing process and sperm transfer in Phytoseiulus persimilis. Exp Appl Acarol 77(1):11–25. https://doi.org/10.1007/s10493-018-0333-0
Karlsson B (1998) Nuptial gifts, resource budgets, and reproductive output in a polyandrous butterfly. Ecology 79(8):2931–2940. https://doi.org/10.1890/0012-9658
Kozłowski J, Wiegert RG (1986) Optimal allocation of energy to growth and reproduction. Theor Popul Biol 29(1):16–37. https://doi.org/10.1016/0040-5809(86)90003-1
Kuştutan O, Cakmak I (2009) Development, fecundity, and prey consumption of Neoseiulus californicus (Mcgregor) fed Tetranychus cinnabarinus Boisduval. Turk J Agric for 33(1):19–28. https://doi.org/10.3906/tar-0806-39
Li GY, Zhang ZQ (2018) Does size matter? Fecundity and longevity of spider mites (Tetranychus urticae) in relation to mating and food availability. Syst Appl Acarol 23(9):1796–1808. https://doi.org/10.11158/saa.23.9.6
Li XB, Zhu R, Yi TC, Guo JJ, Jin DC (2022) Tolerance and functional response of Neoseiulus californicus (McGregaor) to starvation stress. Syst Appl Acarol 27(1):94–106. https://doi.org/10.11158/saa.27.1.10
Lv JL, Zhang BH, Jiang XH, Wang ED, Xu XN (2019) Quantitative impact of mating duration on reproduction and offspring sex ratio of Phytoseiulus persimilis (Acari: Phytoseiidae). J Integr Agric 18(4):884–892. https://doi.org/10.1016/S2095-3119(18)61974-4
Nguyen TT, Amano H (2009) Mating duration and egg production of the predaceous mite Neoseiulus californicus (Acari: Phytoseiidae) vary with temperature. J Asia Pac Entomol 12(4):297–299. https://doi.org/10.1016/j.aspen.2009.06.003
Nguyen TT, Amano H (2010) Temperature at immature and adult stages differentially affects mating duration and egg production of Neoseiulus californicus females mated once (Acari: Phytoseiidae). J Asia Pac Entomol 13(1):65–68. https://doi.org/10.1016/j.aspen.2009.10.002
Nguyen DT, Vangansbeke D, Lu X, De Clercq P (2013) Development and reproduction of the predatory mite Amblyseius swirskii on artificial diets. Biocontrol 58:369–377. https://doi.org/10.1007/s10526-012-9502-y
Penn D, Potts WK (1998) Chemical signals and parasite-mediated sexual selection. Trends Ecol Evol 13(10):391–396. https://doi.org/10.1016/S0169-5347(98)01473-6
Pianka ER, Parker WS (1975) Age-specific reproductive tactics. Am Nat 109(968):453–464. https://doi.org/10.1086/283013
Rasmy AH, Hussein HE (1996) Effect of mating on egg production in two species of predatory mites, Agistemus exsertus Gonzalez and Phytoseiulus persimilis Athias-Henriot. Anz. Schädlingskde., Pflanzenschutz, Umweltschutz 69:88–89 (1996). https://doi.org/10.1007/BF01908437
Richner H, Christe P, Oppliger A (1995) Paternal investment affects prevalence of malaria. Proc Natl Acad Sci USA 92(3):1192–1194. https://doi.org/10.1073/pnas.92.4.1192
Ridley M (1988) Mating frequency and fecundity in insects. Biol Rev 63(4):509–549. https://doi.org/10.1111/j.1469-185x.1988.tb00669.x
Ridley M (1990) The control and frequency of mating in insects. Funct Ecol. https://doi.org/10.2307/2389655
Saber S (2013) Predation, oviposition and conversion rates of the predacious mite, Neoseiulus californicus (Mcgregor) consuming different densities of Tetranychus urticae koch, Bemisia tabaci (Genn.) and Thrips tabaci lind. Arch Phytopathol Plant Prot 46(17):2146–2152. https://doi.org/10.1080/03235408.2013.787700
Schausberger P, Patiño-Ruiz JD, Osakabe M, Murata Y, Sugimoto N, Uesugi R, Walzer A (2016) Ultimate drivers and proximate correlates of polyandry in predatory mites. PLoS ONE 11(4):e0154355. https://doi.org/10.1007/978-981-287-952-3_20
Schausberger P, Walzer A, Murata Y, Osakabe M (2017) Low level of polyandry constrains phenotypic plasticity of male body size in mites. PLoS ONE 12(11):e0188924. https://doi.org/10.1371/journal.pone.0188924
Schrempf A, Von Wyschetzki K, Klein A, Schrader L, Oettler J, Heinze J (2015) Mating with an allopatric male triggers immune response and decreases longevity of ant queens. Mol Ecol 24(14):3618–3627. https://doi.org/10.1111/mec.13267
Schulten GGM, Vanarendonk RCM, Russell VM, Roorda FA (1978) Copulation, egg production and sex-ratio in Phytoseiulus persimilis and Amblyseius biben (Acari: Phytoseiidae). Entomol Exp Appl 24:145–153
Schwenke RA, Lazzaro BP, Wolfne MF (2016) Reproduction–immunity trade-offs in insects. Ann Rev Entomol 61:239–256. https://doi.org/10.1146/annurev-ento-010715-023924
Scott AM, Zhang Z, Jia L, Li K, Zhang Q, Dexheimer T, Ellsworth E, Ren J, Chung-Davidson YW, Zu Y (2019) Spermine in semen of male sea lamprey acts as a sex pheromone. PLoS Biol 17(7):e3000332. https://doi.org/10.1371/journal.pbio.3000332
Smith JM (1977) Parental investment: a prospective analysis. Anim Behav. https://doi.org/10.1016/0003-3472(77)90062-8
Strodl MA, Schausberger P (2012) Social familiarity modulates group living and foraging behaviour of juvenile predatory mites. Naturwissenschaften 99:303–311. https://doi.org/10.1007/s00114-012-0903-7
Thornhill R (1976) Sexual selection and paternal investment in insects. Am Nat 110(971):153–163. https://doi.org/10.1086/283055
Toyoshima S (2008) Effect of starving treatment on gravid females of Neoseiulus californicus (Acari: Phytoseiidae). Int Symp Mites Whitefly 1:185–185. https://doi.org/10.1007/s10493-008-9211-5
Trivers RL (1974) Parent-offspring conflict. Integr Comp Biol 14:249–264. https://doi.org/10.1093/icb/14.1.249
Ullah MS, Sugimoto R, Kongchuensin M, Konvipasruang P, Gotoh T (2017) Copulation duration, sperm transfer and reproduction of the two closely related phytoseiid mites, Noseiulus womersleyi and Neoseiulus longispinosus (Acari: Phytoseiidae). Exp Appl Acarol 71:47–61. https://doi.org/10.1007/s10493-016-0101-y
Walzer A, Schausberger P (1999a) Cannibalism and interspecific predation in the phytoseiid mites Phytoseiulus persimilis and Neoseiulus californicus: predation rates and effects on reproduction and juvenile development. Biocontrol 43(4):457–468. https://doi.org/10.1023/A:1009980401662
Walzer A, Schausberger P (1999b) Predation preferences and discrimination between con-and heterospecific prey by the phytoseiid mites Phytoseiulus persimilis and Neoseiulus californicus. Biocontrol 43(4):469–478. https://doi.org/10.1023/A:1009974918500
Walzer A, Schausberger P (2014) Canalization of body size matters for lifetime reproductive success of male predatory mites (Acari: Phytoseiidae). Biol J Lin Soc 111:889–899. https://doi.org/10.1111/bij.12235
Walzer A, Schausberger P (2015) Interdependent effects of male and female body size plasticity on mating behaviour of predatory mites. Anim Behav 100:96–105. https://doi.org/10.1016/j.anbehav.2014.11.017
Walzer A, Castagnoli M, Simoni S, Liguori M, Palevsky E, Schausberger P (2007) Intraspecific variation in humidity susceptibility of the predatory mite Neoseiulus californicus: survival, development and reproduction. Biol Control 41:42–52. https://doi.org/10.1016/j.biocontrol.2006.11.012
Webberley KM, Hurst GD, Buszko J, Majerus ME (2002) Lack of parasite-mediated sexual selection in a ladybird/sexually transmitted disease system. Anim Behav 63:131–141. https://doi.org/10.1006/anbe.2001.1877
Wedell N (1993) Mating effort or paternal investment? Incorporation rate and cost of male donations in the wartbiter. Behav Ecol Sociobiol 32(4):239–246. https://doi.org/10.1007/bf00166513
Wessels FJ, Jordan DC, Hahn DA (2010) Allocation from capital and income sources to reproduction shift from first to second clutch in the flesh fly, Sarcophaga crassipalpis. J Insect Physiol 56(9):1269–1274. https://doi.org/10.1016/j.jinsphys.2010.03.033
Xu XN, Wang BM, Wang ED, Zhang ZQ (2013) Comments on the identity of Neoseiulus californicus sensu lato (Acari: Phytoseiidae) with a redescription of this species from southern china. Syst Appl Acarol 18(4):329–344. https://doi.org/10.11158/saa.18.4.3
Yasui Y (1997) Sperm competition and the significance of female multiple mating in the predatory mite Parasitus fimetorum. Exp Appl Acarol 21(10):651–664. https://doi.org/10.1023/A:1018456618435
Zhang X, Lv JL, Hu Y, WangB CX, Xu XN, Wang ED (2015) Prey preference and life table of Amblyseius orientalis on Bemisia tabaci and Tetranychus cinnabarinus. PLoS ONE 10(10):e0138820. https://doi.org/10.1371/journal.pone
Acknowledgements
This study was supported by the National Natural Science Foundation of China (31872028) and co-innovation project of CAAS and SDAAS ‘Key technologies on regional green agricultural development and integrated demonstration’.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There is no conflict of interest concerning the results provided in this manuscript. The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lu, Q., Lv, J., Wang, E. et al. Impact of starvation on paternal reproductive investment in Neoseiulus californicus. Exp Appl Acarol 87, 1–18 (2022). https://doi.org/10.1007/s10493-022-00717-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-022-00717-8