Abstract
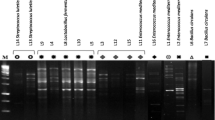
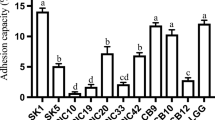
In the present study, eight strains of Enterococcus spp. were isolated from Turkish and Iranian white cheese test pieces. Enterococcus spp. strains were identified as Enterococcus faecium (6 strains) and E. faecalis (2) with 16S rDNA sequence analysis. All Enterococcus spp. strains showed susceptibility to the most of antibiotics tested in this investigation. The amount of produced acid (0.59–1.17%), hydrogen peroxide (0.65–3.91 µg/ml), and exopolysaccharide (252–362 mg/L) of these strains were determined. These strains possess the ability to inhibit Escherichia coli ATCC 35,218, Salmonella enteritidis ATCC 13,076, and Salmonella typhimurium MU 80. E. faecium RI53 and RI 42 strains were determined as the most resistant to acid (1.86 and 1.56 OD, respectively) and also exhibited high percentage of aggregation (54.1 and 51.7%, respectively). E. faecium RI 42 exhibited a higher growth viability in gastric and intestinal juice. E. faecium RI 53 and RI 42 are determined as optimal potential probiotic candidates for utilization in cheese preparations.
Similar content being viewed by others
References
Caggia C, De Angelis M, Pitino I, Pino A, Randazzo CL (2015) Probiotic features of Lactobacillus strains isolated from Ragusano and Pecorino Siciliano cheeses. Food Microbiol 50:109–117
Cebrián R, Baños A, Valdivia E, Pérez-Pulido R, Martínez-Bueno M, Maqueda M (2012) Characterization of functional, safety, and probiotic properties of Enterococcus faecalis UGRA10, a new AS-48-producer strain. Food Microbiol 30:59–67
Clinical and Laboratory Standards Institute (CLSI), (2012). Performance standards for antimicrobial susceptibility tenting: twenty-second informational supplement M100-S22, CLSI, Wayne, PA, USA
Demirci M, Gündüz H (1994) Süt Teknolojisi El Kitabı. Hasad Yayıncılık, p.184
Dos Santos KM, Vieira ADS, Salles HO, da Silva OJ, Rocha CRC, de Fátima BM, Bruno LM, de Melo Franco BDG, Todorov SD (2015) Safety, beneficial and technological properties of Enterococcus faecium isolated from Brazilian cheeses. Braz J Microbiol 46:237–249
DuBois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colometric method for determination of sugars and related substances. Anal Chem 28:350–356
FAO/WHO (2002) Guidelines for the evaluation of probiotics in food. food and health agricultural organisation of the united nations d world health organisation. Working group report
Favaro L, Basaglia M, Casella S, Hue I, Dousset X, de Melo G, Franco BD, Todorov SD (2014) Bacteriocinogenic potential and safety evaluation of non-starter Enterococcus faecium strains isolated from home made white brine cheese. Food Microbiol 38:228–239
Foulquié Moreno MR, Sarantinopoulos P, Tsakalidou E, De Vuyst L (2006) The role and application of Enterococci in food and health. Int J Food Microbiol 106:1–24
Franz CM, Muscholl-Silberhorn AB, Yousif NM, Vancanneyt M, Swings J, Holzapfel WH (2001) Incidence of virulence factors and antibiotic resistance among Enterococci isolated from food. Appl Environ Microbiol 67:4385–4389
Franz CMAP, Huch M, Abriouel H, Holzapfel W, Gálvez A (2011) Enterococci as probiotics and their implications in food safety. Int J Food Microbiol 151:125–140
García-Solache M, Rice LB (2019) The Enterococcus: A model of adaptability to its environment. Clin Microbiol Rev 32:e00058-e118
Garriga M, Pascual M, Monfort JM, Hugas M (1998) Selection of lactobacilli for chicken probiotic adjuncts. J Appl Microbiol 84:125–132
Ghahremani E, Mardani M, Rezapour S (2015) Phenotypic and genotypic characterization of lactic acid bacteria from traditional cheese in Khorramabad city of Iran with probiotic potential. Appl Biochem Biotechnol 175:2516–2527
Giraffa G (2002) Enterococci from foods. FEMS Microbiol Rev 26:163–171
Giraffa G (2003) Functionality of enterococci in dairy products. Int J Food Microbiol 88:215–222
Gomes BC, Esteves CT, Palazzo ICV, Darini ALC, Felis GE, Sechi LA, Franco BDGM, De Martinis ECP (2008) Prevalence and characterization of Enterococcus spp. isolated from Brazilian foods. Food Microbiol 25:668–675
Hanchi H, Mottawea W, Sebei K, Hammami R (2018) The genus Enterococcus: between probiotic potential and safety concerns-an update. Front Microbiol 9:1791
Handa S, Sharma N (2016) In vitro study of probiotic properties of Lactobacillus plantarum F22 isolated from chchang- a traditional fermented beverage of Himachal Pradesh, India. J Genet Eng Biotechnol 14:91–97
Hassanzadazar H, Ehsani A, Mardani K (2014) Antibacterial activity of Enterococcus faecium derived from Koopeh cheese against Listeria monocytogenes in probiotic ultra-filtrated cheese. Vet Res Forum 5:169–175
Hollenbeck BL, Rice LB (2012) Intrinsic and acquired resistance mechanisms in enterococcus. Virulence 15:421–433
Ilango S, Pandey R, Antony U (2016) Functional characterization and microencapsulation of probiotic bacteria from koozh. J Food Sci Technol 53:977–989
Jahansepas A, Sharifi Y, Aghazadeh M, Rezaee MA (2020) Comparative analysis of Enterococcus faecalis and Enterococcus faecium strains isolated from clinical samples and traditional cheese types in the Northwest of Iran: antimicrobial susceptibility and virulence traits. Arch Microbiol 202:765–772
Kirmaci HA, Özer BH, Akçelik M, Akçelik N (2015) Identification and characterization of lactic acid bacteria isolated from traditional Urfa cheese. Int J Dairy Technol 69:301–307
Kivanç SA, Kivanç M, Yiğit T (2016) Antibiotic susceptibility, antibacterial activity and characterisation of Enterococcus faecium strains isolated from breast milk. Exp Ther Med 12:1732–1740
Kizerwetter-Świda M, Binek M (2016) Assessment of potentially probiotic properties of Lactobacillus strains isolated from chickens. Polish J Vet Sci 19:15–20
Klayraung S, Okonogi S (2009) Antibacterial and antioxidant activities of acid and bile resistant strains of Lactobacillus fermentum isolated from miang. Braz J Microbiol 40:757–766
Koluman A, Akan LS, Çakiroğlu FP (2009) Occurrence and antimicrobial resistance of enterococci in retail foods. Food Control 20:281–283
Kozak K, Charbonneau D, Sanozky-Dawes R, Klaenhammer T (2015) Characterization of bacterial isolates from the microbiota of mothers’ breast milk and their infants. Gut Microbes 6:341–351
Looijesteijn PJ, Trapet L, De Vries E, Abee T, Hugenholtz J (2001) Physiological function of exopolysaccharides produced by Lactococcus lactis. Int J Food Microbiol 64:71–80
Marshall VM, Rawson HL (1999) Effects of exopolysaccharide-producing strains of thermophilic lactic acid bacteria on the texture of stirred yoghurt. Int J Food Sci Technol 34:137–143
Mater DD, Langella P, Corthier G, Flores MJ (2008) A probiotic Lactobacillus strain can acquire vancomycin resistance during digestive transit in mice. J Mol Microbiol Biotechnol 14:123–127
Mozzi F, Vaningelgem F, Hébert ME, der Meulen RV, Moreno MRF, De Valdez GF, De Vuyst L (2006) Diversity of heteropolysaccharide-producing lactic acid bacterium strains and their biopolymers. Appl Environ Microbiol 72:4431–4435
O’Hanlon DE, Moench TR, Cone RA (2011) In vaginal fluid, bacteria associated with bacterial vaginosis can be suppressed with lactic acid but not hydrogen peroxide. BMC Infect Dis 11:200
Onal Darilmaz D (2013) Relationship between gastrointestinal tolerance and exopolysaccharide production of propionibacteria strains under different pH and bile conditions. Int J Dairy Technol 66:194–201
Pieniz S, Andreazza R, Okeke BC, Camargo FAO, Brandelli A (2015) Antimicrobial and antioxidant activities of Enterococcus species isolated from meat and dairy products. Braz J Biol 75:923–931
Rao KP, Chennappa G, Suraj U, Nagaraja H, Raj APC, Sreenivasa MY (2015) Probiotic potential of Lactobacillus strains isolated from sorghum-based traditional fermented food. Probiotics Antimicrob Proteins 7:146–156
Rasouli Pirouzian H, Hesari J, Farajnia S, Moghaddam M, Ghiassifar S (2009) Isolation and identification of dominant strains of Enterococci in traditional Lighvan cheese. Food Technol 19:13–24
Reinheimer JA, Demkow MR, Condioti MC (1990) Inhibition of coliform bacteria by lactic cultures. Aust J Dairy Technol 45:5–9
Ribeiro SC, Coelho MC, Todorov SD, Franco BDGM, Dapkevicius MLE, Silva CCG (2013) Technological properties of bacteriocin-producing lactic acid bacteria isolated from Pico cheese an artisanal cow’s milk. J Appl Microbiol 116:573–585
Ruas-Madiedo P, Hugenholtz J, Zoon P (2002) An overview of the functionality of exopolysaccharides produced by lactic acid bacteria. Int Dairy J 12:163–171
Ruas-Madiedo P, Moreno JA, Salazar N, Delgado S, Mayo B, Margolles A, de Los Reyes-Gavilán CG (2007) Screening of exopolysaccharide-producing Lactobacillus and Bifidobacterium strains isolated from the human intestinal microbiota. Appl Environ Microbiol 73:4385–4388
Saavedra L, Taranto MP, Sesma F, Font de Valdez G (2003) Homemade traditional cheeses for the isolation of probiotic Enterococcus faecium strains. Int J Food Microbiol 88:241–245
Sahoo TK, Jena PK, Nagar N, Patel AK, Seshadri S (2015) In vitro evaluation of probiotic properties of lactic acid bacteria from the gut of Labeo rohita and Catla catla. Probiotics Antimicrob Proteins 7:126–136
Shim YH, Lee SJ, Lee JW (2016) Antimicrobial activity of lactobacillus strains against uropathogens. Pediatr Int 58:1009–1013
Skrzypczak K, Gustaw W, Waśko A (2015) Health-promoting properties exhibited by Lactobacillus helveticus strains. Acta Biochim Pol 62:713–720
Sönmez Ş, Darilmaz DO, Beyatli Y (2018) Determination of the relationship between oxalate degradation and exopolysaccharide production by different Lactobacillus probiotic strains. Int J Dairy Technol 71:741–752
Sun W, Liu J, Xu H, Li W, Zhang J (2015) L-Lactic acid fermentation by Enterococcus faecium: a new isolate from bovine rumen. Biotechnol Lett 37:1379–1383
Svec P, Devriese LA (2009) Genus I. Enterococcus (ex Thiercelin and Jouhaud, Schleifer and Kilpper-Balz 1984, 32VP). In: De Vos P, Garrity GM, Jones D, Krieg NR, Ludwig W, Rainey FA, Schleifer KH, Whitman WB (eds) Bergey’s manual of systematic microbiology, the Firmicutes, 2nd edn. Springer, New York, pp. 594–607
Topcuoglu S, Gursoy T, Ovalı F, Serce O, Karatekin G (2015) A new risk factor for neonatal vancomycin resistant Enterococcus colonisation: bacterial probiotics. J Matern Fetal Neonatal Med 28:1491–1494
Tulumoglu S, Yuksekdag ZN, Beyatli Y, Simsek O, Cinar B, Yasar E (2013) Probiotic properties of Lactobacilli species isolated from children’s feces. Anaerobe 24:36–42
Valladares RB, Graves C, Wright K, Gardner CL, Lorca GL, Gonzalez CF (2015) H2O2 production rate in Lactobacillus johnsonii is modulated via the interplay of a heterodimeric flavin oxidoreductase with a soluble 28 Kd PAS domain containing protein. Front Microbiol 6:716
Valyshev AV (2014) Antimicrobial compounds of enterococci. Zh Mikrobiol Epidemiol Immunobiol 5:119–126
Yoo MY, Kim YJ, Hwang HJ (2008) Properties and safety aspects of Enterococcus faecium strains isolated from Chungkukjang, a fermented soy product. LWT-Food Sci Technol 41:925–933
Yuksekdag ZN, Sahin N, Aslim B (2014) In vitro evaluation of the suitability potential probiotic of lactobacilli isolates from the gastrointestinal tract of chicken. Eur Food Res Technol 239:313–320
Acknowledgements
The authors sincerely thank Gazi University, Faculty of Science, Department of Biology for the financial support.
Author information
Authors and Affiliations
Contributions
YB and ZY conceived the study and analyzed the data. RH, DÖD and ZY performed molecular and microbiologic analyses. DÖD wrote the first draft of the report and all authors revised and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Consent for publication
All authors gave their consent to participate in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hajikhani, R., Onal Darilmaz, D., Yuksekdag, Z. et al. Assessment of some metabolic activities and potential probiotic properties of eight Enterococcus bacteria isolated from white cheese microbiota. Antonie van Leeuwenhoek 114, 1259–1274 (2021). https://doi.org/10.1007/s10482-021-01599-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-021-01599-3