Abstract
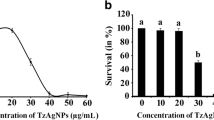
Biofilms are structured consortia of microbial cells that grow on living and non living surfaces and surround themselves with secreted polymers. Infections with bacterial biofilms have emerged as a foremost public health concern because biofilm growing cells can be highly resistant to both antibiotics and host immune defenses. Zinc oxide nanoparticles have been reported as a potential antimicrobial agent, thus, in the current study, we have evaluated the antimicrobial as well as antibiofilm activity of zinc oxide nanoparticles against the bacterium Streptococcus pneumoniae which is a significant cause of disease. Zinc oxide nanoparticles showed strong antimicrobial activity against S. pneumoniae, with an MIC value of 40 μg/ml. Biofilm inhibition of S. pneumoniae was also evaluated by performing a series of experiments such as crystal violet assay, microscopic observation, protein count, EPS secretion etc. using sub-MIC concentrations (3, 6 and 12 µg/ml) of zinc oxide nanoparticles. The results showed that the sub-MIC doses of zinc oxide nanoparticles exhibited significant anti-biofilm activity against S. pneumoniae, with maximum biofilm attenuation found at 12 μg/ml. Taken together, the results indicate that zinc oxide nanoparticles can be considered as a potential agent for the inhibition of microbial biofilms.








Similar content being viewed by others
References
Balasubramanian V, Natarajan K, Hemambika B, Ramesh N, Sumathi CS, Kottaimuthu R, Rajash KV (2010) High-density polyethylene (HDPE)-degrading potential bacteria from marine ecosystem of Gulf of Mannar, India. Lett Appl Microbiol 51(2):205–211
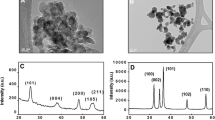
Baruah S, Rafique RF, Dutta J (2008) Visible light photo-catalysis by tailoring crystal defects in zinc oxide nanostructures. NANO 3(05):399–407
Bjarnsholt T (2013) The role of bacterial biofilms in chronic infections. Apmis 121(s136):1–58
Bogaert D, de Groot R, Hermans PWM (2004) Streptococcus pneumoniae colonization: the key to pneumococcal disease. Lancet Infect Dis 4(3):144–154
Chao Y, Marks LR, Pettigrew MM, Hakansson AP (2014) Streptococcus pneumoniae biofilm formation and dispersion during colonization and disease. Front Cell Infect Microbiol 13:4
Clinical and Laboratory Standards Institute (2005) Performance standards for antimicrobial susceptibility testing: fifteenth informational supplement M100-S15. CLSI, Wayne
Crossley KB, Jefferson KK, Archer GL, Fowler VG (2009) Staphylococci in human disease, 2nd edn. Blackwell, West Sussex
Das MC, Paul S, Gupta P, Tribedi P, Sarkar S, Manna D, Bhattacharjee S (2016) 3-Amino-4-aminoximidofurazan derivatives: small molecules possessing antimicrobial and anti-biofilm activity against Staphylococcus aureus and Pseudomonas aeruginosa. J Appl Microbiol 120(4):842–859
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Flemming HC, Wingender J (2010) The biofilm matrix. Nat Rev Microbiol 8(9):623–633
Gupta P, Sarkar S, Das B, Bhattacharjee S, Tribedi P (2016) Biofilm, pathogenesis and prevention-a journey to break the wall: a review. Arch Microbiol 198(1):1–15
Hall-Stoodley L, Stoodley P (2009) Evolving concepts in biofilm infections. Cell Microbiol 11(7):1034–1043
Johnson LR (2008) Microcolony and biofilm formation as a survival strategy for bacteria. J Theor Biol 251(1):24–34
Joo HS, Otto M (2012) Molecular basis of in vivo biofilm formation by bacterial pathogens. Chem Biol 19(12):1503–1513
Kaplan JB, Ragunath C, Ramasubbu N, Fine DH (2003) Detachment of Actinobacillus actinomycetemcomitans biofilm cells by an endogenous β-hexosaminidase activity. J Bacteriol 185(16):4693–4698
Lau PC, Lindhout T, Beveridge TJ, Dutcher JR, Lam JS (2009) Differential lipopolysaccharide core capping leads to quantitative and correlated modifications of mechanical and structural properties in Pseudomonas aeruginosa biofilms. J Bacteriol 191(21):6618–6631
Lee JH, Kim YG, Cho MH, Lee J (2014) ZnO nanoparticles inhibit Pseudomonas aeruginosa biofilm formation and virulence factor production. Microbiol Res 169(12):888–896
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Markman JL, Rekechenetskiy A, Holler E, Ljubimova JY (2013) Nanomedicine therapeutic approaches to overcome cancer drug resistance. Adv Drug Deliv Rev 65(13):1866–1879
Mukherjee K, Tribedi P, Mukhopadhyay B, Sil AK (2013) Antibacterial activity of long chain fatty alcohols against Mycobacteria. FEMS Microbiol Lett 338(2):177–183
Ojha AK, Baughn AD, Sambandan D, Hsu T, Trivelli X, Guererdal Y, Alahari A, Kremer L, Jacobs WR, Hattful GF (2008) Growth of Mycobacterium tuberculosis biofilms containing free mycolic acids and harbouring drug-tolerant bacteria. Mol Microbiol 69(1):164–174
Oldak E, Trafny EA (2005) Secretion of proteases by Pseudomonas aeruginosa biofilms exposed to Ciprofloxacin. Antimicrob Agents Chemother 49(8):3281–3288
Ortqvist Å, Hedlund J, Kalin M (2005) Streptococcus pneumoniae: epidemiology, risk factors, and clinical features. Semin Respir Crit Care Med 26(6):563–574
Pati R, Mehta RK, Mohanty S, Padhi A, Sengupta M, Vaseeharan B, Goswami C, Sonawane A (2014) Topical application of zinc oxide nanoparticles reduces bacterial skin infection in mice and exhibits antibacterial activity by inducing oxidative stress response and cell membrane disintegration in macrophages. Nanomed Nanotechnol Biol Med 10(6):1195–1208
Salem W, Leitner DR, Zingl FG, Schratter G, Prassl R, Goessler W, Reidl J, Schild S (2015) Antibacterial activity of silver and zinc nanoparticles against Vibrio cholerae and enterotoxic Escherichia coli. Int J Med Microbiol 305(1):85–95
Thaya R, Malaikozhundan B, Vijayakumar S, Sivakamavalli J, Jeyasekar R, Shanthi S, Sonawane A (2016) Chitosan coated Ag/ZnO nanocomposite and their antibiofilm, antifungal and cytotoxic effects on murine macrophages. Microb Pathog 100:124–132
Tribedi P, Sil AK (2013) Low-density polyethylene degradation by Pseudomonas sp. AKS2 biofilm. Environ Sci Pollut R 20(6):4146–4153
Tribedi P, Sil AK (2014) Cell surface hydrophobicity: a key component in the degradation of polyethylene succinate by Pseudomonas sp. AKS2. J Appl Microbiol 116(2):295–303
Tribedi P, Gupta AD, Sil AK (2015) Adaptation of Pseudomonas sp. AKS2 in biofilm on low-density polyethylene surface: an effective strategy for efficient survival and polymer degradation. Bioresour Bioprocess 2(1):14
Vasudevan R (2014) Biofilms: microbial cities of scientific significance. J Microbiol Exp 1(3):00014
Vijayakumar S, Vinoj G, Malaikozhundan B, Shanthi S, Vaseeharan B (2015) Plectranthus amboinicus leaf extract mediated synthesis of zinc oxide nanoparticles and its control of methicillin resistant Staphylococcus aureus biofilm and blood sucking mosquito larvae. Spectrochim Acta Part A Mol Biomol Spectrosc 137:886–891
Vijayakumar S, Malaikozhundan B, Shanthi S, Vaseeharan B, Thajuddin N (2017) Control of biofilm forming clinically important bacteria by green synthesized ZnO nanoparticles and its ecotoxicity on Ceriodaphnia cornuta. Microb Pathog 107:88–97
Acknowledgements
The authors would like to express sincere gratitude to the learned reviewers for their valuable comments raised during the course of review for the betterment of the quality of the current research work.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Authors declare that they do not have any conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bhattacharyya, P., Agarwal, B., Goswami, M. et al. Zinc oxide nanoparticle inhibits the biofilm formation of Streptococcus pneumoniae . Antonie van Leeuwenhoek 111, 89–99 (2018). https://doi.org/10.1007/s10482-017-0930-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-017-0930-7