Abstract
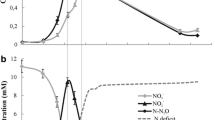
Until recently, it has not been generally known that some bacteria can contain the gene inventory for both denitrification and dissimilatory nitrate (NO3 −)/nitrite (NO2 −) reduction to ammonium (NH4 +) (DNRA). Detailed studies of these microorganisms could shed light on the differentiating environmental drivers of both processes without interference of organism-specific variation. Genome analysis of Bacillus azotoformans LMG 9581T shows a remarkable redundancy of dissimilatory nitrogen reduction, with multiple copies of each denitrification gene as well as DNRA genes nrfAH, but a reduced capacity for nitrogen assimilation, with no nas operon nor amtB gene. Here, we explored nitrogen assimilation in detail using growth experiments in media with different organic and inorganic nitrogen sources at different concentrations. Monitoring of growth, NO3 − NO2 −, NH4 + concentration and N2O production revealed that B. azotoformans LMG 9581T could not grow with NH4 + as sole nitrogen source and confirmed the hypothesis of reduced nitrogen assimilation pathways. However, NH4 + could be assimilated and contributed up to 50% of biomass if yeast extract was also provided. NH4 + also had a significant but concentration-dependent influence on growth rate. The mechanisms behind these observations remain to be resolved but hypotheses for this deficiency in nitrogen assimilation are discussed. In addition, in all growth conditions tested a denitrification phenotype was observed, with all supplied NO3 − converted to nitrous oxide (N2O).




Similar content being viewed by others
References
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Aziz RK, Bartels D, Best AA et al (2008) The RAST server: rapid annotations using subsystems technology. BMC Genomics 9:75
Baethgen W, Alley M (1989) A manual colorimetric procedure for measuring ammonium nitrogen in soil and plant Kjeldahl digests. Commun Soil Sci Plant Anal 20:961–969
Belitsky BR, Sonenshein AL (2004) Modulation of activity of Bacillus subtilis regulatory proteins GltC and TnrA by glutamate dehydrogenase. J Bacteriol 186:3399–3407
Belitsky BR, Wray LV, Fisher SH, Bohannon DE, Sonenshein AL (2000) Role of TnrA in nitrogen source-dependent repression of Bacillus subtilis glutamate synthase gene expression. J Bacteriol 182:5939–5947
Bender R, Janssen K, Resnick A, Blumenberg M, Foor F, Magasanik B (1977) Biochemical parameters of glutamine synthetase from Klebsiella aerogenes. J Bacteriol 129:1001–1009
Bleakley BH, Tiedje JM (1982) Nitrous oxide production by organisms other than nitrifiers or denitrifiers. Appl Environ Microbiol 44:1342–1348
Brenchley JE, Prival MJ, Magasanik B (1973) Regulation of the synthesis of enzymes responsible for glutamate formation in Klebsiella aerogenes. J Biol Chem 248:6122–6128
Bronk DA, Glibert PM, Ward BB (1994) Nitrogen uptake, dissolved organic nitrogen release, and new production. Science 265:1843–1846
Canfield DE, Glazer AN, Falkowski PG (2010) The evolution and future of earth’s nitrogen cycle. Science 330:192–196. doi:10.1126/science.1186120
Castignetti D, Hollocher TC (1984) Heterotrophic nitrification among denitrifiers. Appl Environ Microbiol 47:620–623
Cataldo D, Maroon M, Schrader L, Youngs V (1975) Rapid colorimetric determination of nitrate in plant tissue by nitration of salicylic acid. Commun Soil Sci Plant Anal 6:71–80
Chen J, Zhang Y, Du G-C, Hua Z-Z, Zhu Y (2007) Biodegradation of polyvinyl alcohol by a mixed microbial culture. Enzym Microb Technol 40:1686–1691
Cornell S, Randell A, Jickells T (1995) Atmospheric inputs of dissolved organic nitrogen to the oceans. Nature 376:243–246
Coutts G, Thomas G, Blakey D, Merrick M (2002) Membrane sequestration of the signal transduction protein GlnK by the ammonium transporter AmtB. EMBO J 21:536–545
Dandekar T, Snel B, Huynen M, Bork P (1998) Conservation of gene order: a fingerprint of proteins that physically interact. Trends Biochem Sci 23:324–328
Deshpande KL, Kane JF (1980) Glutamate synthase from Bacillus subtilis: in vitro reconstitution of an active amidotransferase. Biochem Biophys Res Commun 93:308–314
Deuel TF, Stadtman E (1970) Some kinetic properties of Bacillus subtilis glutamine synthetase. J Biol Chem 245:5206–5213
Durand A, Merrick M (2006) In vitro analysis of the Escherichia coli AmtB-GlnK complex reveals a stoichiometric interaction and sensitivity to ATP and 2-oxoglutarate. J Biol Chem 281:29558–29567
Fisher SH (1999) Regulation of nitrogen metabolism in Bacillus subtilis: vive la difference! Mol Microbiol 32:223–232
Gibson T (1934) An investigation of the Bacillus Pasteurii group. J Bacteriol 28:313
Goss TJ, Perez-Matos A, Bender RA (2001) Roles of glutamate synthase, gltBD, and gltF in nitrogen metabolism of Escherichia coli and Klebsiella aerogenes. J Bacteriol 183:6607–6619
Griess P (1879) Bemerkungen zu der abhandlung der HH. Weselsky und Benedikt” ueber einige azoverbindungen”. Ber Dtsch Chem Ges 12:426–428
Gunka K, Commichau FM (2012) Control of glutamate homeostasis in Bacillus subtilis: a complex interplay between ammonium assimilation, glutamate biosynthesis and degradation. Mol Microbiol 85:213–224
Hashimoto S, Fujita M (1985) Isolation of a bacterium requiring three amino acids for polyvinyl alcohol degradation. J Ferment Technol 63:471–474
Heylen K, Keltjens J (2012) Redundancy and modularity in membrane-associated dissimilatory nitrate reduction in Bacillus. Front Microbiol 3:1–27. doi:10.3389/fmicb.2012.00371
Jansson SL, Hallam M, Bartholomew W (1955) Preferential utilization of ammonium over nitrate by micro-organisms in the decomposition of oat straw. Plant Soil 6:382–390
Kloosterman TG, Hendriksen WT, Bijlsma JJE et al (2006) Regulation of glutamine and glutamate metabolism by GlnR and GlnA in Streptococcus pneumoniae. J Biol Chem 281:25097–25109
Kormelink TG, Koenders E, Hagemeijer Y, Overmars L, Siezen RJ, de Vos WM, Francke C (2012) Comparative genome analysis of central nitrogen metabolism and its control by GlnR in the class Bacilli. BMC Genomics 13:191
Kristjansson J, Hollocher T (1980) First practical assay for soluble nitrous oxide reductase of denitrifying bacteria and a partial kinetic characterization. J Biol Chem 255:704–707
Leigh JA, Dodsworth JA (2007) Nitrogen regulation in bacteria and archaea. Annu Rev Microbiol 61:349–377
Li Y (2007) Principles and technology of fermentation engineering. Higher Education Press, Beijing
Magasanik B (1992) Regulation of nitrogen utilization. In: Jones EW, Pringle JR, Broach JR (eds). Cold spring harbor monograph archive, vol 21, pp 283–317
Magasanik B (1996) Regulation of nitrogen utilization. In: Neidhardt FC et al (eds) Escherichia coli and Salmonella: cellular and molecular biology, vol 1. ASM Press, Washington, pp 1344–1356
Mörsdorf G, Kaltwasser H (1989) Ammonium assimilation in Proteus vulgaris, Bacillus pasteurii, and Sporosarcina ureae. Arch Microbiol 152:125–131
Navarro-Gonzalvez JA, Garcia-Benayas C, Arenas J (1998) Semiautomated measurement of nitrate in biological fluids. Clin Chem 44:679–681
Overbeek R, Olson R, Pusch GD et al (2014) The SEED and the rapid annotation of microbial genomes using subsystems technology (RAST). Nucleic Acids Res 42:D206–D214
Pichinoty F, Bigliardi-Rouvier J, Mandel M, Greenway B, Méténier G, Garcia J-L (1976a) The isolation and properties of a denitrifying bacterium of the genus Flavobacterium. Antonie Van Leeuwenhoek 42:349–354
Pichinoty F, de Berjac H, Mandel M, Greenway B, Garcia JL (1976b) A new, sporulating, denitrifying, mesophilic bacterium: Bacillus azotoformans N. SP. (author’s transl). Ann Microbiol 127b:351–361
Pichinoty F, Durand M, Job C, Mandel M, Garcia JL (1978a) Morphological, physiological and taxonomic studies of Bacillus azotoformans. Can J Microbiol 24:608–617
Pichinoty F, Garcia J-L, Job C, Durand M (1978b) La dénitrification chez Bacillus licheniformis. Can J Microbiol 24:45–49
Pichinoty F, Mandel M, Garcia JL (1979) The properties of novel mesophilic denitrifying Bacillus cultures found in tropical soils. Microbiology 115:419–430
Pichinoty F, De Barjac H, Mandel M, Asselineau J (1983) Description of Bacillus azotoformans sp. nov. Int J Syst Bacteriol 33:660–662
Reitzer LJ, Magasanik B (1996) Ammonia assimilation and the biosynthesis of glutamine, glutamate, aspartate, asparagine, l-alanine, and d-alanine. Escherichia coli and Salmonella: cellular and molecular biology, 2nd edn, ASM Press, Washington, DC, pp 391–407
Richards E, Shrikhande J (1934) The preferential utilization of different forms of inorganic nitrogen in the decomposition of plant materials. Soil Sci 39:1–8
Sander R (1999) Compilation of Henry’s law constants for inorganic and organic species of potential importance in environmental chemistry. Max-Planck Institute of Chemistry, Air Chemistry Department Mainz, Germany
Schmidt E, Schmidt F (1974) Glutamate dehydrogenase. Methods Enzym Anal 3:216–227
Schmidt CS, Richardson DJ, Baggs EM (2011) Constraining the conditions conducive to dissimilatory nitrate reduction to ammonium in temperate arable soils. Soil Biol Biochem 43:1607–1611
Schreier HJ, Brown SW, Hirschi KD, Nomellini JF, Sonenshein AL (1989) Regulation of Bacillus subtilis glutamine synthetase gene expression by the product of the glnR gene. J Mol Biol 210:51–63
Smith JS, Hillier A, Lees G, Jago G (1975) The nature of the stimulation of the growth of Streptococcus lactis by yeast extract. J Dairy Res 42:123–138
Sonenshein AL (2007) Control of key metabolic intersections in Bacillus subtilis. Nat Rev Microbiol 5:917–927
Soupene E, He L, Yan D, Kustu S (1998) Ammonia acquisition in enteric bacteria: physiological role of the ammonium/methylammonium transport B (AmtB) protein. Proc Natl Acad Sci USA 95:7030–7034
Stanier R, Palleroni N, Doudoroff M (1966) The aerobic pseudomonads a taxonomic study. J Gen Microbiol 43:159–271
Stewart G, Mann A, Fentem P (1980) Enzymes of glutamate formation: glutamate dehydrogenase, glutamine synthetase and glutamate synthase. Biochem Plants 5:271–327
Strohm TO, Griffin B, Zumft WG, Schink B (2007) Growth yields in bacterial denitrification and nitrate ammonification. Appl Environ Microbiol 73:1420–1424
Suharti Strampraad MJ, Schroder I, de Vries S (2001) A novel copper A containing menaquinol NO reductase from Bacillus azotoformans. Biochemistry 40:2632–2639
Thomas G, Coutts G, Merrick M (2000) The glnKamtB operon: a conserved gene pair in prokaryotes. Trends Genet 16:11–14
Tiedje JM, Sexstone AJ, Myrold DD, Robinson JA (1982) Denitrification: ecological niches, competition and survival. Antonie Van Leeuwenhoek 48:569–583
Van den Berg EM, Van Dongen U, Abbas B, Van Loosdrecht MC (2015) Enrichment of DNRA bacteria in a continuous culture. ISME J 9:2153–2161
Van den Heuvel R, Curti B, Vanoni M, Mattevi A (2004) Glutamate synthase: a fascinating pathway from l-glutamine to l-glutamate. Cell Mol Life Sci 61:669–681
Van Heeswijk WC, Westerhoff HV, Boogerd FC (2013) Nitrogen assimilation in Escherichia coli: putting molecular data into a systems perspective. Microbiol Mol Biol Rev 77:628–695
Vanoni M, Curti B (1999) Glutamate synthase: a complex iron-sulfur flavoprotein. Cell Mol Life Sci 55:617–638
Wheeler PA, Kirchman DL (1986) Utilization of inorganic and organic nitrogen by bacteria in marine systems. Limnol Oceanogr 31:998–1009
Wray L, Atkinson MR, Fisher SH (1994) The nitrogen-regulated Bacillus subtilis nrgAB operon encodes a membrane protein and a protein highly similar to the Escherichia coli glnB-encoded PII protein. J Bacteriol 176:108–114
Yoon S, Sanford RA, Loeffler FE (2015a) Nitrite control over dissimilatory nitrate/nitrite reduction pathways in Shewanella loihica strain PV-4. Appl Environ Microbiol 81:3510–3517
Yoon SH, Cruz-Garcia C, Sanford RA, Ritalahti KM, Löffler FE (2015b) Denitrification versus respiratory ammonification: environmental controls of two competing dissimilatory NO3 −/NO2 − reduction pathways in Shewanella loihica strain PV-4. ISME J 9:1–12
Zalieckas JM, Wray LV, Fisher SH (2006) Cross-regulation of the Bacillus subtilis glnRA and tnrA genes provides evidence for DNA binding site discrimination by GlnR and TnrA. J Bacteriol 188:2578–2585
Zdobnov EM, Apweiler R (2001) InterProScan–an integration platform for the signature-recognition methods in InterPro. Bioinformatics 17:847–848
Acknowledgements
We thank Kim Heylen for input on experimental design and helpful discussion of results. We thank Rob Van Spanning for advice of experimental design and data analysis. We thank Robin Cornelis for her experimental assistance in NH4 + concentration tests. We thank Bart Hoste, Evie De Brandt and Bram Vekeman for technical assistance with the spectrophotometer, colorimetrics and gas chromatography. YS was funded by the Chinese Scholarship Council (File number 201206330054) and the Special Research Fund (BOF) from Ghent University (grant 01SC2713 and GOA project 01G01911).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Not applicable.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sun, Y., De Vos, P. & Willems, A. Nitrogen assimilation in denitrifier Bacillus azotoformans LMG 9581T . Antonie van Leeuwenhoek 110, 1613–1626 (2017). https://doi.org/10.1007/s10482-017-0911-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-017-0911-x