Abstract
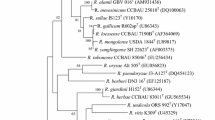
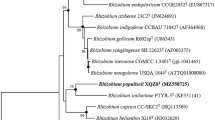
Two novel, Gram-negative, motile, rod-shaped, aerobic bacterial strains, MH17T and RD15, were isolated from the sterilized root and rhizosphere soil of rice, respectively. Phylogenetic analysis based on 16S rRNA gene sequences showed that the similarity between strains MH17T and RD15 was 100%. The isolates exhibit high sequence similarities to Rhizobium oryzae CGMCC 1.7048T (98.7%) and Rhizobium petrolearium SL-1T (97.0% and 97.1%), which supports that they belong to a novel species in the genus Rhizobium. Strains MH17T and RD15 exhibited growth at 15–45 °C, pH 5.0–11.0, 0–2.0% sodium chloride (w/v). Sequence analysis of housekeeping genes gyrB, recA, atpD, ropB, gltA showed that these two novel strains had less than 94% similarity with the known species, indicating the distinct position of MH17T and RD15 in the genus Rhizobium. The major cellular fatty acids were identified as summed feature 8 (C18:1 ω7c and/or C18:1 ω6c). Type strain MH17T had 87.5% DNA–DNA relatedness with RD15 by using the initial renaturation rate method. Based on draft genome sequences, strain MH17T showed 30.1% DNA–DNA hybridization values to R. oryzae CGMCC 1.7048T, the closely related strain, which supported that MH17T represents a novel species in the genus Rhizobium. Average nucleotide identity (ANI) between strains MH17T and RD15 were 97.8%, and strain MH17T showed 82.2% ANI value with R. oryzae CGMCC 1.7048T. The DNA G+C content was 60.4 mol% (Tm). Based on physiological, biochemical characteristic, genotypic data, strains MH17T and RD15 are concluded to represent a new species within the genus Rhizobium, for which the name Rhizobium rhizosphaerae sp. nov. is proposed. The type strain is MH17T (=ACCC 19963T = KCTC 52414T).


Similar content being viewed by others
References
De Ley J (1970) Reexamination of the association between melting point, buoyant density, and chemical base composition of deoxyribonucleic acid. J Bacteriol 101:738–754
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376
Frank B (1889) Ueber die Pilzsymbiose der Leguminosen. Ber DtschBot Ges 7:332–346 (in German)
Gu T, Sun LN, Zhang J, Sui XH, Li SP (2014) Rhizobium flavum sp. nov., a triazophos-degrading bacterium isolated from soil under the long-term application of triazophos. Int J Syst Evol Microbiol 64:2017–2022
Jordan DC (1984) Genus I. Rhizobium Frank 1889, 338AL. In: Krieg NR, Holt JG (eds) Bergey’s manual of systematic bacteriology, vol 1. Williams & Wilkins, Baltimore, pp 235–242
Kim OS, Cho YJ, Lee K, Yoon SH, Kim M, Na H, Park SC, Jeon YS, Lee JH, Yi H, Won S, Chun J (2012) Introducing EzTaxon-e: a prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species. Int J Syst Evol Microbiol 62:716–721
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1–6
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematics. Wiley, Chichester, pp 115–175
Lin SY, Hung MH, Hameed A, Liu YC, Hsu YH, Wen CZ, Arun AB, Busse HJ, Glaeser SP, Kämpfer P, Young CC (2015) Rhizobium capsici sp. nov., isolated from root tumor of a green bell pepper (Capsicum annuum var. grossum) plant. Antonie Van Leeuwenhoek 107:773–784
Marmur J, Doty P (1962) Determination of the base composition of deoxyribonucleic acid from its thermal denaturation temperature. J Mol Biol 5:109–118
Martens M, Delaere M, Coopman R, De Vos P, Gillis M, Willems A (2007) Multilocus sequence analysis of Ensifer and related taxa. Int J Syst Evol Microbiol 57:489–503
Martens M, Dawyndt P, Coopman R, Gillis M, De Vos P, Willems A (2008) Advantages of multilocus sequence analysis for taxonomic studies: a case study using 10 housekeeping genes in the genus Ensifer (including former Sinorhizobium). Int J Syst Evol Microbiol 58:200–214
Meier-Kolthoff JP, Auch AF, Klenk HP, Göker M (2012) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform 14:1–14
Rzhetsky A, Nei M (1992) A simple method for estimating and testing minimum evolution trees. Mol Biol Evol 9(5):945–967
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Sasser M (1990) Identification of bacteria by gas chromatography of cellular fatty acids, MIDI Technical Note 101. MIDI Inc, Newark, DE
Sheu S, Huang H, Young C, Chen W (2015) Rhizobium alvei sp. nov., isolated from a freshwater river. Int J Syst Evol Microbiol 65:472–478
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTALW: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Tighe SW, de Lajudie P, Dipietro K, Lindström K, Nick G, Jarvis BD (2000) Analysis of cellular fatty acids and phenotypic relationships of Agrobacterium, Bradyrhizobium, Mesorhizobium, Rhizobium and Sinorhizobium species using the Sherlock microbial identification system. Int J Syst Evol Microbiol 50:787–801
Turdahon M, Osman G, Hamdun M, Yusuf K, Abdurehim Z, Abaydulla G, Abdukerim M, Fang C, Rahman E (2013) Rhizobium tarimense sp. nov., isolated from soil in the ancient Khiyik River. Int J Syst Evol Microbiol 63:2424–2429
Vincent JM (1970) The cultivation, isolation and maintenance of rhizobia. In: Vincent JM (ed) A manual for the practical study of the root-nodule bacteria. Blackwell Scientific, Oxford, pp 1–13
Young JM, Kuykendall LD, Martínez-Romero E, Kerr A, Sawada H et al (2001) A revision of Rhizobium Frank 1889, with an emended description of the genus, and the inclusion of all species of Agrobacterium Conn 1942 and Allorhizobium undicola de Lajudie, 1998 as new combinations: Rhizobium radiobacter, R. rhizogenes, R. rubi, R. undicola and R. vitis. Int J Syst Evol Microbiol 51:89–103
Zhang X, Sun L, Ma X, Sui XH, Jiang R (2011) Rhizobium pseudoryzae sp. nov., isolated from the rhizosphere of rice. Int J Syst Evol Microbiol 61:2425–2429
Zhang X, Tang X, Ali Sheirdil R, Sun L, Ma X (2014) Rhizobium rhizoryzae sp. nov., isolated from rice roots. Int J Syst Evol Microbiol 64:1373–1377
Zhang X, Gao J, Cao Y, Sheirdil R, Wang X, Zhang L (2015) Isolation and proposal novel rice promoting endophytic bacteria, Rhizobium oryzicola sp. nov. Int J Syst Evol Microbiol 65:2931–2936
Acknowledgements
This work was supported by the Grants from the Chinese Ministry of Science and Technology (2013AA102802) and National Natural Science Foundation of China (316700005; 41271273).
Author information
Authors and Affiliations
Corresponding author
Additional information
The GenBank/EMBL/DDBJ accession numbers for the 16S rRNA, gyrB, recA, atpD, ropB, gltA gene sequences of strain MH17T are KX129902, KX434874, KX434876, KY321909, KY321911, KY321915, respectively.
Rights and permissions
About this article
Cite this article
Zhao, JJ., Zhang, J., Zhang, RJ. et al. Rhizobium rhizosphaerae sp. nov., a novel species isolated from rice rhizosphere. Antonie van Leeuwenhoek 110, 651–656 (2017). https://doi.org/10.1007/s10482-017-0831-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-017-0831-9