Abstract
The use of stimulant drugs alone or in combination with amyl nitrites (stimulant/nitrites) has been associated with higher rates of risky sexual behavior and predictive of HIV infection among men who have sex with men. However, the temporal pattern of stimulant/nitrite use pre- and post-seroconversion has not been well established. This study assessed changes in stimulant/nitrite use and risky sexual behavior among seroconverting MSM over time. Data were collected in the Baltimore-Washington, DC; Pittsburgh; Chicago; and Los Angeles sites of the Multicenter AIDS Cohort Study (MACS), a longitudinal study of the natural history of HIV infection among MSM. We used propensity scores to select 1044 MSM from 7087 MACS participants composed of 348 seroconverting, 348 seronegative, and 348 seroprevalent participants matched on demographics, recruitment cohort, and study visits. We centered up to four-years of semi-annual data around the seroconversion visit of the seroconverting case within each matched group of participants. Mixed effects regressions estimated the effects of serostatus, recruitment cohort, and time on self-reported stimulant/nitrite use, numbers of male intercourse partners, and numbers of unprotected receptive anal intercourse (URAI) partners. Covariates included demographics, binge drinking, and marijuana use. Seroconverters had the highest odds of stimulant/inhaled nitrite use (AOR 10.3, CI 4.8–22.0), incident rates of intercourse (IRR 1.6, CI 1.3–2.1), and URAI partners (IRR 5.1, CI 3.5–7.3). All participants decreased drug use and sexual risk behavior over time. However, the decreases were largest for seroconverters who nevertheless maintained the highest rates of stimulant/nitrite use and sexual risk. Cohort-related effects were associated with sharp reductions in stimulant/nitrite use and URAI in the early 1990s that rebounded considerably within the first decade of the 2000s. Although all participants decreased risky sexual behavior and stimulant and/or nitrite use over time, seroconverters had the largest decreases. There was no evidence for abrupt or substantial increases in drug use or risky sex post-seroconversion. However, there was substantial variation at the individual level, with the factors underlying this variation not well understood and worth further study. Moreover, stimulant/nitrite use and risky sexual behavior appear to have been strongly influenced by contextual historical and socio-cultural effects. The manner in which contextual factors influence individual behavior is also not well understood and also warrants further study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Numerous studies have shown that, relative to the general population, men who have sex with men (MSM) disproportionately use an array of drugs; in particular, drugs in the stimulant class (e.g., cocaine, methamphetamine) and inhaled amyl nitrites (“poppers,” referred to hereafter as nitrites) [1,2,3,4,5,6,7,8]. The reasons for elevated rates of substance use, including alcohol use, among MSM are thought to be related to a confluence of factors including stigma, higher rates of depression, and social isolation [9]. Use of stimulants and amyl nitrites, often in combination with other drugs such as club drugs or drugs for treating erectile dysfunction, has been strongly associated with increased risk for HIV infection as use often occurs in the context of riskier and more frequent sex with higher numbers of partners [10,11,12,13]. These drugs are used to heighten sexual desire, enhance sexual experience, prolong sexual activity, and facilitate meeting partners for sex [14, 15]. An additional problematic aspect of the drug-sex link among MSM is evidenced by the fact that stimulant and nitrite use are prevalent among a subset of HIV+ MSM who continue to engage in relatively high-rates of unprotected sex with multiple partners and who, in association with their substance use, are less likely to adhere to their HIV medication regimens [7, 12, 16,17,18,19].
Although the connections between stimulants, nitrites, sexual behavior, and risk for HIV infection are well established, the temporal patterns pre- and post-HIV seroconversion are less clear [20]. A good deal of the ambiguity is owing to methodological limitations in studies conducted to date: reliance on cross-sectional data; samples that include only substance users; lack of comparison groups composed of long-term HIV− and HIV+ seroprevalent MSM; and assessing substance use and sexual risk behaviors either prior to or following but not across HIV seroconversion [7, 17, 21,22,23,24,25]. Consequently, the extent to which the use of stimulants, nitrites, and risky sexual behaviors that precedes and contributes to the risk for HIV infection increases, decreases, or is maintained post-infection has not been determined.
Using data obtained for the Multicenter AIDS Cohort Study (MACS), we examined the patterns of stimulant and nitrite use as well as sexual behavior pre-and post-HIV seroconversion and comparatively analyzed temporal changes by HIV serostatus for MSM who seroconverted during the study, were seroprevalent (HIV+) at enrollment, or who remained seronegative (HIV−). These three groups were selected to allow for comparative assessment of substance use and sexual behavior among participants shortly before and after seroconversion with participants whose HIV status was unchanged over the same time. Secondarily, we examined whether recruitment cohort affected substance use and sexual behavior given research that indicates different patterns of these behaviors for more recent (post 2000) compared with cohorts of MSM coming of age in the 1980s and 1990s [3, 24, 26].
Method
This secondary analysis used the P24 release of the MACS public data set (https://statepi.jhsph.edu/macs/pdt.html). MACS is an ongoing longitudinal study of the course of HIV infection and treatment among MSM. Participating MACS centers are in Baltimore-Washington, DC; Chicago; Los Angeles; and Pittsburgh. MACS began in 1984 and has enrolled over 7000 MSM to date, most of whom were recruited in 1 of 3 cohorts: 1984-1985 (n = 4954); 1987–1990 (n = 668), and 2001–2003 (n = 1350). The last recruitment in 2001–2003 focused on enrolling younger racial/ethnic minority individuals. Data are collected semi-annually and include self-reported information on past six-month sexual behavior and substance use. Eligibility criteria include: being male, ages 18–70, having had sex with another man in the prior 5 years, and not having been diagnosed with AIDS [27]. Further detail on MACS rationale, eligibility criteria, data collection and instrumentation, and current status are available at http://www.statepi.jhsph.edu/macs/macs.html.
All MACS participants provided informed consent and the study protocol was reviewed and approved by each participating site’s respective IRBs. As this study used the de-identified public use data set, we applied for and received determinations of exemption from the University of Illinois at Chicago and California State University at Northridge IRBs.
Participants
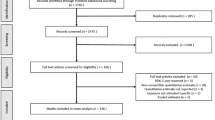
We selected the analytic sample from 7087 study participants across multiple steps using multiple selection criteria at each step (Fig. 1). Our goal was to retrospectively create matched “triads” with each composed of one seroconverting participant matched to one HIV+ and one HIV− participant. We first selected participants with available demographic and HIV status information (n = 6988) who were recruited in cohorts 1–3 (n = 6969) and had completed at least 4 interviews. Seroconverting participants had the additional selection criteria of having at least 1 interview within 2 years prior to seroconversion. The resulting subsample (n = 5773) was composed of: 464 of the 712 participants that seroconverted after study enrollment; 2372 that were HIV+ at enrollment; and 2937 that remained HIV− over the follow-up period.
We stratified this subsample by cohort and applied propensity score matching with calipers set at 0.25 s.d. of the propensity score to select the closest matching HIV+ and HIV− comparison cases for each seroconverting case [28]. Multiple iterations of the matching algorithm for each cohort were used to identify the best-matching HIV+ and HIV− cases after taking into account available interviews for matched cases [29]. Stratifying the propensity scoring by cohort allowed us to match seroconverting participants with HIV+ and HIV− participants in the same cohort, thereby maximizing the possibility matched participants would have corresponding visits and interview data. Participant attributes used to calculate propensity scores were: age at study entry, race/ethnicity, education level, first and last study visits, and total number of visits completed. Propensity scoring using these attributes resulted in matches for 391 (84.3%) of the 464 seroconverters who met study inclusion criteria, representing 54.9% of the 712 seroconverters in the data set.
In the final selection step, we removed seroconverters and their matched controls if any of the selected controls had fewer than two visits within the 2 years prior and 2 years following the indexed seroconversion study visit for the matched seroconverting case or if we could not match a seroconverter with both an HIV− and HIV+ control. This step was complicated by the fact that between April 1992 and April 1996 (visits 16–25), questions about amphetamine and MDA use were dropped from the MACS questionnaire and later re-added. However, for the affected study visits, preliminary analyses showed substantially suppressed estimates of stimulant/nitrite use. To minimize loss of cases, we set the variable for stimulant/nitrite use to missing where questions on stimulants and MDA use were not asked (143 of 7189 total visits or 2.0%) and re-estimated the number of available visits excluding those with missing substance use data. Participants were retained only if they continued to meet inclusion criteria (e.g., 2+ interviews, matching HIV+ and HIV− cases for each seroconverting case) resulting in the deletion of 129 additional cases. Bivariate analyses comparing the deleted with retained cases found no significant differences for any of the modeled independent or dependent variables except cohort where a significantly (Chi square (2) LR = 34.4, p < 0.001) higher proportion of deleted cases (21.1%, n = 27) were from the second recruitment cohort compared with retained cases (4.4% n = 45). The final analytic sample was composed of 1044 participants: 348 seroconverters individually matched to 348 HIV+ and 348 HIV− participants.
Measures
Demographics
Because the de-identified MACS data set contains only birth year, we approximated age at each visit by subtracting birth year from visit year, accounting for semi-annual visits by adding .5 years to every other visit. Race/ethnicity was dummy coded into four dichotomous variables: White, Black/AA, Hispanic/Latino, and Other. Highest year of education reported at baseline was also dummy coded into four dichotomous variables: less than high school, high school graduate, some college, or college graduate.
Study Visit
Study visit data were selected based on the first HIV-positive visit of the seroconverting case within each triad, centered between (up to) 4 visits immediately preceding and (up to) four immediately following seroconversion. Thus, each participant contributed up to nine interviews spanning 2 years (4 visits) prior and 2 years (4 visits) post the indexed seroconversion visit. All available study visits within the time-frame established by the seroconverting case were selected for the matched HIV+ and HIV− cases. For instance, if a participant seroconverted at MACS study visit 10, the preceding (6 through 9) and subsequent (11 through 14) visits were selected for analysis for this participant as well as for the matched seronegative and seroprevalent participants. All available study visits within waves 6–14 were selected for the three matched cases and then centered around the seroconversion visit, which was set as time 5. The 4 pre-seroconversion visits were set as times 1 through 4, and the 4 post-seroconversion visits set as times 6 through 9. If a visit within the time frame was unavailable, it was set to missing. Participants within each triad could contribute a minimum of two and a maximum of nine data points for the analyses. In the multivariable regression models, because we were interested in how a positive HIV test changed stimulant use and sexual risk behavior, time was represented by a series of dichotomous indicators. The 1044 matched cases averaged 6.4 interviews (s.d. = 1.8, range = 2–9). The interquartile range of visit year for the indexed seroconversion visit was: cohort 1—1985 to 1988; cohort 2—1989 to 1991; and cohort 3—2005 to 2008.
Serostatus
For the MACS, HIV serostatus is assessed biannually at each study visit for all HIV− participants [30]. Participants were classified as a seroconverter if they tested negative for HIV at enrollment but tested positive at a later visit; the seroconversion visit was indicated by the first visit with a positive test. Participants were classified as seroprevalent HIV+ if they were HIV+ at enrollment or seroprevalent HIV− if they were HIV− at enrollment with no subsequent positive HIV test. Serostatus was the primary independent variable.
Cohort
To capture potential socio-cultural contextual/historical effects, we included MACS recruitment cohort as a predictor variable. Cohort was represented by dummy-coded variables representing the first, second, and third recruitment cohorts.
Binge Drinking
MACS participants are asked: “On days when you drank any alcoholic beverages, how many drinks do you usually have altogether?” and “How often [have] you had a drink containing alcohol?” Binge drinking was defined as reporting having 5 or more drinks on a weekly or more frequent basis in the past 6 months. Binge drinking was included as a dichotomous model predictor as research has found it is associated with both riskier sexual behavior and HIV infection [31,32,33,34,35].
Marijuana Use
MACs participants are asked if they have used any of a series of drugs even once in the past 6 months. We included marijuana use in our models to assess its association with stimulant and nitrite use and sexual behavior. Excepting alcohol, marijuana was the most commonly used drug by participants (Table 1). Marijuana use was categorized dichotomously as any use in the past 6 months (no/yes) and included as predictor in the multivariable models.
Stimulants and Nitrites
Use of stimulants and/or inhaled nitrites were measured in a single dichotomous variable (no/yes) based on any self-reported use of two or more of four drugs at any frequency of use in the past 6 months: amphetamine/methamphetamine/speed, any form of cocaine, MDA (i.e., “Ecstasy,” “X,” “Molly”), and inhaled nitrites. We selected these drugs based on studies that have found concurrent use of two or more of these drugs associated with more frequent sexual risk behaviors and predictive of HIV seroconversion. [7, 12, 24, 25, 36,37,38] The dichotomous variable stimulant/nitrite use was assessed as a binary outcome in the multivariable models.
Number of Male Sex and URAI Partners
Number of male sex partners was defined as the number of men with whom a participant reported having sexual intercourse of any kind (i.e., oral or anal, receptive or Insertive) in the past 6 months. Variables representing each measure were assessed as outcomes on a count level in separate negative binomial models.
Analyses
Analyses were conducted using Stata 14.1 [39]. Descriptive bivariate analyses used data from the enrollment visit. We used likelihood ratio Chi square tests or F-tests to compare participants by serostatus on demographics, stimulants, nitrites, and other substance use, number of available interviews, and number of male intercourse and unprotected receptive anal intercourse (URAI) partners. Multivariable analyses were conducted using two-level, mixed effects regressions with interview occasions (level 1) nested within participant (level 2). Models included random intercepts representing subject-specific effects for each dependent variable. Each model assessed changes in the dependent variable with respect to time referenced to the first HIV+ interview of the seroconverting participants (time 5). Additionally, to examine the associations between sexual behavior and stimulant use over time, the regression models with number of male sex partners and number of male URAI partners as the dependent variables, stimulant and/or nitrite use in the prior 6 months became a predictor. Stimulant and nitrite use was modeled using the logit-link function and binomial distribution whereas, the models regressing number of male and URAI sex partners used the log-link function and negative binomial distribution.
Modeling proceeded sequentially by first estimating a main-effects only model. Interaction terms for time by cohort and time by serostatus were then added one at a time to the main-effects model and assessed for significance using post-estimation, likelihood ratio Chi square tests and the BIC statistic to compare each interaction-term model with the main-effects model. We estimated the preliminary models using maximum likelihood estimation whereas final models used restricted maximum likelihood estimation. All models were estimated assuming unstructured covariances, adaptive quadrature with 30 integration points, and robust standard errors to provide less biased (i.e., more conservative) estimates of the standard errors and associated p-values and to control for residual heteroscedasticity. [40, 41]
Results
Sample Characteristics
Table 1 presents bivariate results by serostatus. Reflecting that the large majority of MACS participants were recruited for the initial cohort (1984–1985), there were 921 (88.2%) participants from the first cohort; 45 (4.3%) from the second; and 78 (7.5%) from the third. Participants were predominantly white (83.5%), averaged 33.0 years of age at enrollment, and had an education level of some college or higher (87.3%). Participants averaged 24 visits (s.d. = 15.3) across study participation and 6.4 (s.d. = 1.8) visits over the sampled range. None of these covariates were significantly different by serostatus indicating the propensity score matching produced demographically balanced groups.
Consistent with prior study findings, however, there were large and statistically significant differences by HIV serostatus on most of the substance use measures as well as numbers of sex partners [10, 17, 42]. Except for binge drinking, substance use was more prevalent for participants who seroconverted or were HIV+ compared with HIV− participants. For instance, 22.4% of MACS participants who seroconverted and 20.7% of HIV+ participants reported using amphetamine/methamphetamine in the past six months at time 5 (the indexed seroconversion interview) compared with 12.6% of HIV− participants (LR Chi square (df=2) = 13.1, p = 0.001). Participants who seroconverted or were HIV+ also had significantly higher rates than HIV− participants on all three dependent variables: use of any 2+ stimulants in the past 6 months (LR Chi square (df=2) = 40.9, p < 0.001); mean number of male sexual intercourse partners (F(df=2, 1040) = 7.58, p < 0.001) and mean number of URAI partners (F(df=2, 1040) = 7.18, p < 0.001).
Multivariable Models
Table 2 shows the results for the multivariable mixed-effects regressions. Likelihood ratio tests of the comparisons of the fixed effects only models to the mixed effects models that included the random intercept terms were all significant, indicating the models with random intercepts fit the data better than the fixed-effects only models (stimulant/nitrite use: LR Chi square(df=1) = 2051.63, p < 0.001; number of male sex partners: LR Chi square(df= 1) = 4587.38, p < .001; number of URAI partners: LR Chi square(df= 1) = 1612.54, p < 0.001). Consequently, we used the mixed effects models when testing the addition of interaction terms representing cohort and serostatus by time to the main-effects only models.
Stimulant/Nitrite Use
Parameter estimates represented as odds ratios, confidence intervals, and significance levels are shown in Table 2. The residual intraclass correlation of 0.75 (95% CI 0.72–0.78) indicated a large amount of the variation in stimulant/nitrite use is accounted for at the individual level, providing further evidence that using a multilevel framework was warranted. Comparisons of the models that included interaction terms for cohort by time (LR Chi square(df=16) = 49.13, p < 0.001) and serostatus by time (LR Chi square(df=16) = 117.20, p < 0.001) to the main-effects only model yielded significant results. Consequently, as shown in the second half of Table 2, we retained these interactions in the final model. Figure 2 depicts estimations of the predicted marginal mean probability of stimulant/nitrite use by serostatus, time and cohort based on final model results.
Demographics related to lower odds of 2+ stimulant use were: age (AOR 0.96, p < 0.01) and being African = American (AOR 0.26, p < 0.01; White = reference category). Recruitment cohorts 2 (AOR 0.13, p < 0.001) and 3 (AOR 0.15, p < 0.01) also had significantly lower odds of 6-month stimulants/nitrite use relative to the initial MACS cohort. The main effect of time indicated that stimulant/nitrite use declined in the subsequent 2 years, reaching the lowest point at the two-years following the midpoint (AOR 0.29, p < 0.001). Factors related to increased odds of using 2+ stimulants included: binge drinking (AOR 2.21, p < 0.001), and any use of marijuana (AOR 8.89, p < 0.001). Participants who seroconverted (AOR 10.24, p < 0.001) or were HIV+ throughout the study (AOR 3.42, p < 0.01) were more likely to use stimulants/nitrites than participants who remained HIV−.
The significant interaction effects obtained indicated that across serostatus and cohort, stimulant/nitrite use was relatively constant in the first 2 years assessed and not significantly different from the mid-point interview. However, in contrast to the reference group participants in the first cohort, third cohort participants did not show consistent declines in stimulant/nitrite use with statistically significant increased odds of use at the 6-month (AOR 4.00, p < 0.05), 18-month (AOR 5.05, p < 0.01), and two-year interviews (AOR 5.73, p < 0.01) relative to the indexed seroconversion interview. The significant serostatus by time interaction was also due to post-seroconversion differences. Relative to HIV− participants, seroconverting MACS participants had larger reductions at 1-year (AOR 0.41, p < 0.05), 18-months (AOR 0.30, p < 0.01), and 2-years post-seroconversion (AOR 0.30, p < 0.05). Hence, seroconverting participants had the highest rates of stimulant/nitrite use across time, their use declined more sharply following the seroconversion visit than HIV+ participants relative to the reference group of HIV− participants
Number of Male Sex Partners
Model estimates for number of male sex partners are reported as incident rate ratios (IRR) reflecting that this dependent variable was assessed using counts of partners per unit time (6 months). MACS participants who seroconverted had a higher number of male intercourse partners relative to HIV− participants (IRR 1.62, p < 0.001). Binge drinking (IRR 1.24, p < 0.001), any marijuana use (IRR 1.28, p < 0.001), and stimulant/nitrite use (IRR 1.56, p < 0.001) were all significantly associated with having higher numbers of male sex partners. Conversely, significantly lower numbers of male sex partners were reported by participants who were Black/African-American (IRR .57, p < 0.001). Whereas the number of male sex partners increased by 17% prior to the mid-point visit (IRR 1.17, p < 0.05), the IRRs trended lower at subsequent visits (e.g., 2 years IRR 0.78, p < 0.01).
The interactions between cohort (LR Chi square(df=16) = 49.13. p = 0.001) and serostatus (LR Chi square(df=16) = 117.20, p < 0.001) by time were again statistically significant and retained in the final model (Table 2 and Fig. 3). The significant cohort by time interaction effects were exclusively due to participants in the third cohort who, relative to the first cohort, had lower numbers of male sex partners at two-years (IRR 0.55, p < 0.01) and 18 months (IRR 0.61, p < 0.05) prior to the index visit but higher numbers (IRR 1.61, p < 0.01) at 6 months index visit. The significant serostatus by time interaction effects were due primarily to seroconverting participants having the largest decreases in the number of male sex partners relative to HIV− participants over the 2 years post-seroconversion: (6-months, IRR .69, p < 0.001; 1-year, IRR .61, p < 0.001; 18-months, IRR 0.62, p < 0.001; 2-years, IRR .53, p < 0.001).
Number of URAI Partners
Results for number of URAI partners are also reported as IRRs. Main effects associated with an increased number of URAI partners were: marijuana use (IRR 1.39, p < 0.001), stimulant/nitrite use (IRR 2.20, p < 0.001), seroconverting (IRR 5.07, p < 0.001) or being HIV+ (IRR 3.00, p < 0.001) or being in the third cohort (IRR 1.41, p < 0.01). Conversely, age (IRR 0.95, p < 0.001), being black/African American (IRR 0.58, p < 0.05) or in the other racial/ethnic category (IRR 0.20, p < 0.01), and having a HS/GED or lower education level (IRR 0.62, p < 0.05) were all associated with a lower number of URAI partners.
As with the prior two models, interaction terms for both cohort (LR Chi square(df=14) = 136.43, p < 0.001) and serostatus (LR Chi square(df=14) = 47.99, p < 0.001) by time were also statistically significant and retained in the model. The cohort by time interaction effect (Fig. 4) was due to three very different temporal patterns for each cohort: participants in the first cohort showed very large drops in the rates of URAI partners regardless of serostatus whereas cohort 2 had persistently low (near zero) URAI partners over nine interview periods assessed. Cohort 3, however, showed a striking and increasing rebound in the number of URAI partners, reversing the trends seen with cohorts 1 and 2. The serostatus by time interaction effect is attributable to seroconverters reporting larger relative decreases in the number of URAI partners at every interview following the indexed seroconversion interview (6-months, IRR .52, p < 0.01; 1-year, IRR 0.50, p < 0.01; 18-months, IRR 0.30, p < 0.001; 2-years, IRR 0.29, p < 0.001).
Discussion
This study of MACS participants examined the temporal pattern of stimulant/nitrite use among MSM over four-and-a-half-year periods centered around the time of seroconversion. Results indicate that study participants, including those who seroconverted, decreased their use of stimulants and nitrites over time. MSM who seroconverted had the highest rates of use compared with other participants, but their decrease in use over time was more pronounced. Still, even after these sharper declines, seroconverting participants continued to have the highest rate of stimulant/nitrite use relative to other participants.
These results are consistent with more circumscribed longitudinal as well as cross-sectional studies that have examined stimulant use either prior to or following seroconversion and found stimulant use to be elevated both leading up to and following seroconversion among recently HIV+ MSM [3, 20, 24, 42, 43]. Our study adds to these findings by showing that although stimulant/nitrite use among seroconverters is elevated compared with seronegative MSM as well as MSM with longer-standing HIV infections, their (seroconverters) higher rates of stimulant/nitrite use post-seroconversion reflects a decrease from even higher pre-seroconversion rates. Their relatively high rate of stimulant/nitrite use does not represent sustained or increased use as might be concluded if only post-seroconversion data are examined.
Therefore, whereas the findings don’t support a general pattern of increased stimulant/nitrite use as an immediate response to HIV seroconversion, MSM who seroconvert continue to evidence relatively higher rates of stimulant/nitrite use and riskier sex than MSM who remain seronegative or whose seroconversion was a more distal event. Moreover, despite declining use, substance use among recently seroconverted MSM remains an issue immediately post-seroconversion and should be assessed and, as needed treated particularly given it’s known negative association with medication adherence. Similarly, the rates of male sexual and URAI partners also decreased over time. As with stimulant/nitrite use, seroconverters had the highest rates of male sexual intercourse and URAI partners over time and across cohorts but sharply decreased their rates of sexual partners. They maintained or continued lowering these rates across the two-years of post-conversion interviews.
Cohort-Related Effects
However, these just-noted changes over time in stimulant/nitrite use and numbers of male sexual partners, particularly number of URAI partners, were subject to cohort-related effects. MACS recruitment of MSM in three temporally distinct cohorts provided the opportunity to examine the effects of changing and unique historical contexts such as medical advancements in HIV treatment as well as changing cultural norms that have accompanied and framed the HIV epidemic in the United States [44]. We believe the significant cohort and cohort-by-time effects reflect these macro-level changes and show how broader, contextual factors influence individual behavior. These effects are most evident for the third (2001–2003) MACS recruitment cohort. Averaging across time and serostatus, participants in the third cohort, which includes higher proportions of African American (42.3%) and Latino participants (32.1%) with lower education levels (34.6% with ≤ 12 years) and a higher mean age at seroconversion [45], had the highest rates of stimulant use even though, as shown in Fig. 2, the rates of the first cohort appear higher overall. This apparent discrepancy is because unlike participants in the first two MACS cohorts, participants in the third cohort maintained their stimulant/nitrite use over time regardless of serostatus. The unique temporal pattern for the third cohort is consistent with the findings of other studies that show an increase in stimulant use among MSM that began approximately in the late 1990s and continuing through the present [6, 10, 19, 46].
Cohort differences appear to be most pronounced for number of URAI partners. Whereas participants in the first MACS cohort showed steep and continued declines in URAI partners over time, second cohort participants maintained an extremely low rate. Conversely, cohort 3 participants had a sharp increase in number of URAI partners over time. We believe this finding captures several important contextual and historical influences: For members of the second cohort, who enrolled in 1987–1991 when contracting HIV was most likely to result in death and who had likely witnessed first-hand the death of many partners and friends, stimulant/nitrite use and sexual risk behaviors were at their lowest levels. Third cohort MACS participants were recruited 10 years later following the advent and increasing use of much more effective treatments for HIV circa the mid 1990s (i.e., highly active anti-retroviral treatment or HAART) [47]. The resultant “treatment optimism” as well as “safe-sex fatigue” along with the emerging availability of the Internet for readily finding sexual partners were all likely contributors to the rebound in stimulant/nitrite use as well as to higher rates of URAI partners reflected in the cohort and cohort by time effects [48, 49]. It is important to note, however, that despite these cohort effects, seroconverting MSM had the most elevated rates of stimulant/nitrite use and highest numbers of URAI partners over time and cohort. These findings suggest that whereas cultural context plays an important role in shaping behavior, it does not completely over-ride individual-level propensities for using drugs and engaging in risky sexual behavior.
Poly-Drug Use
While the research is clear that stimulant/nitrite use is strongly associated with risky sexual behavior among MSM regardless of HIV serostatus, binge drinking and marijuana use also appear to contribute to and are indicative of an overall risky lifestyle that involves poly-drug use. MSM who binge drink and/or use marijuana in addition to using stimulants/nitrites appear to be at highest risk for HIV infection [16, 37]. Hence, tailored prevention and intervention efforts need to take what is likely opportunistic and “wide-ranging” use of multiple drugs into account for this highest risk group of MSM regardless of HIV serostatus. Binge drinking and marijuana use could, in effect, be indicators for poly-drug use that involves stimulants/nitrites. As alcohol and marijuana are less socially-sanctioned drugs than stimulants like cocaine or methamphetamine, individuals could be more likely to admit use at screening, warranting further assessment for poly-drug use and risky sex.
Limitations
Matching participants by visit and demographics across serostatus and selecting only those participants with more than two interviews reduced the pool of eligible participants and could have limited study generalizability as could the limited geographical coverage of MACS generally. Substance use and sexual behaviors over the prior six months were collected via self-report and can be subject to over- or, more likely, under-reporting particularly for sanctioned drugs like cocaine and methamphetamine [50, 51]. We used single item measures of substance use with unknown psychometric properties and we did not have measures of the motivation for substance use. Most of the MSM in this study were long-term MACS participants and were interviewed and tested repeatedly every 6 months over many years. Participation in the study could have increased participants’ awareness of HIV-risk factors on a regular basis thereby affecting their rates of substance use as well as sexual behavior contributing to the decreases over time that we found for both. The relatively small numbers of participants in cohorts two and three means that cohort effects for these participants, especially for URAI partners where the rates of having any such partners were relatively low, should be interpreted with caution. Recent studies suggest different trends in stimulant use among young MSM, meaning that the findings for the MACS recruitment cohorts might no longer apply to the current generation of MSM [46]. The finding of distinct temporal patterns of stimulant use and URAI partners by cohort underscores the importance of macro-level and historical factors and the need to obtain comparable data from more recent cohorts of MSM.
Conclusions and Recommendations
This study contributes to the literature on stimulant use and sexual behavior of MSM by taking advantage of the unique opportunity afforded by the MACS to compare changes over time prior to and following HIV seroconversion. Our findings suggest that among MSM, both risky sexual behavior and stimulant/nitrite use tend to decrease following an HIV diagnosis. These findings are encouraging as they suggest HIV+ MSM, in general, do act to reduce their chances of spreading HIV. However, the findings also show sexual behavior and drug use are based on individual factors such as age and race/ethnicity as well as macro-level, social norms and historical events which can act to increase an individual’s level of behavior. This finding means that community-level interventions aimed at changing social and cultural norms could be at least as important as individual-level interventions in reducing the still high HIV infection rate among MSM; both are clearly needed.
Relatedly, it is interesting to note that research has found some individuals maintain or even increase stimulant use after being diagnosed with HIV and that the intra-individual pattern of stimulant use can vary episodically over time including substitution of one stimulant (cocaine) for another (methamphetamine) [52, 53]. We also found substantial inter- and intra-individual variation in stimulant/nitrite use as evidenced by the variation in intercepts and residuals in the statistical models. The motivations behind, or causes of increased or sustained use remain unclear as we focused on average behaviors in this study and did not try to further determine or characterize variation at the individual level [54]. Yet because of the individual as well as public health concerns represented, we believe further researching the small subgroup of MSM who might maintain or increase substance use and high rates of risky sexual behaviors post-seroconversion is warranted. Such study will help us better understand the reasons why this minority’s high-risk behavior runs contrary to the majority, and how to more effectively engage them in health promotion and HIV prevention/risk-reduction interventions. At the same time, we should also note that MSM whose substance use puts them at high risk are indeed in the minority; identifying the resiliencies of most MSM who are NOT high-risk substance users might also provide important information for shaping more successful individual and community-based interventions [25].
References
Gorbach PM, Weiss RE, Jeffries R, Javanbakht M, Drumright LN, Little SJ. Behaviors of recently HIV-infected men who have sex with men in the year post-diagnosis: effects of drug use and partner types. J Acquir Immune Defic Syndr. 2011;56(2):176–82.
Gonzales R, Mooney L, Rawson RA. The methamphetamine problem in the United States. Annu Rev Public Health. 2010;31:385–98.
Halkitis PN, Levy MD, Solomon TM. Temporal relations between methamphetamine use and HIV seroconversion in gay, bisexual, and other men who have sex with men. J Health Psychol. 2016;21(1):93–9.
Halkitis PN, Pollock JA, Pappas MK, Dayton A, Moeller RW, Siconolfi D, et al. Substance use in the MSM population of New York City during the era of HIV/AIDS. Subst Use Misuse. 2011;46(2–3):264–73.
Goldstein ND, Burstyn I, LeVasseur MT, Welles SL. Drug use among men by sexual behaviour, race and ethnicity: prevalence estimates from a nationally representative US sample. Int J Drug Policy. 2016;36:148–50.
Halkitis PN, Parsons JT, Stirratt MJ. A double epidemic: crystal methamphetamine drug use in relation to HIV transmission among gay men. J Homosex. 2001;41(2):17–34.
Ostrow DG, Plankey MW, Cox C, Li X, Shoptaw S, Jacobson LP, et al. Specific sex drug combinations contribute to the majority of recent HIV seroconversions among MSM in the MACS. J Acquir Immune Defic Syndr. 2009;51(3):349–55.
Marshal MP, Friedman MS, Stall R, King KM, Miles J, Gold MA, et al. Sexual orientation and adolescent substance use: a meta-analysis and methodological review. Addiction. 2008;103(4):546–56.
Halkitis PN, Kapadia F, Bub KL, Barton S, Moreira AD, Stults CB. A longitudinal investigation of syndemic conditions among young gay, bisexual, and other MSM: the P18 cohort study. AIDS Behav. 2015;19(6):970–80.
Shoptaw S, Reback CJ. Methamphetamine use and infectious disease-related behaviors in men who have sex with men: implications for interventions. Addiction. 2007;102(Suppl. 1):130–5.
Fisher DG, Reynolds GL, Ware MR, Napper LE. Methamphetamine and Viagra use: relationship to sexual risk behaviors. Arch Sex Behav. 2011;40(2):273–9.
Patterson TL, Semple SJ, Zians JK, Strathdee SA. Methamphetamine-using HIV-positive men who have sex with men: correlates of polydrug use. J Urban Health. 2005;82(1 Suppl 1):i120–6.
Menza TW, Hughes JP, Celum CL, Golden MR. Prediction of HIV acquisition among men who have sex with men. Sex Transm Dis. 2009;36(9):547–55.
Lyons T, Chandra G, Goldstein J, Ostrow DG. Breaking the bond between stimulant use and risky sex: a qualitative study. Subst Abus. 2010;31(4):224–30.
Clatts MC, Welle DL, Goldsamt LA. Reconceptualizing the interaction of drug and sexual risk among MSM speed users: notes toward an ethno-epidemiology. AIDS Behav. 2001;5(2):115–30.
McCarty-Caplan D, Jantz I, Swartz J. MSM and drug use: a latent class analysis of drug use and related sexual risk behaviors. AIDS Behav. 2014;18(7):1339–51.
Plankey MW, Ostrow DG, Stall R, Cox C, Li X, Peck JA, et al. The relationship between methamphetamine and popper use and risk of HIV seroconversion in the Multicenter AIDS Cohort Study. J Acquir Immune Defic Syndr. 2007;45(1):85–92.
Reback CJ, Larkins S, Shoptaw S. Methamphetamine abuse as a barrier to HIV medication adherence among gay and bisexual men. AIDS Care. 2003;15(6):775–85.
Colfax G, Shoptaw S. The methamphetamine epidemic: implications for HIV prevention and treatment. Curr HIV/AIDS Rep. 2005;2(4):194–9.
Fisher DG, Reynolds GL, Napper LE. Use of crystal methamphetamine, Viagra, and sexual behavior. Curr Opin Infect Dis. 2010;23(1):53–6.
Shoptaw S, Reback CJ. Associations between methamphetamine use and HIV among men who have sex with men: a model for guiding public policy. J Urban Health. 2006;83(6):1151–7.
Rudy ET, Shoptaw S, Lazzar M, Bolan RK, Tilekar SD, Kerndt PR. Methamphetamine use and other club drug use differ in relation to HIV status and risk behavior among gay and bisexual men. Sex Transm Dis. 2009;36(11):693–5.
Shoptaw S, Reback CJ, Freese TE. Patient characteristics, HIV serostatus, and risk behaviors among gay and bisexual males seeking treatment for methamphetamine abuse and dependence in Los Angeles. J Addict Dis. 2002;21(1):91–105.
Colfax G, Coates TJ, Husnik MJ, Huang Y, Buchbinder S, Koblin B, Chesney M, Vittinghoff E, The EXPLORE Study Team. Longitudinal patterns of methamphetamine, popper (amyl nitrite), and cocaine use and high-risk sexual behavior among a cohort of San Francisco men who have sex with men. J Urban Health. 2005;82(1):162–70.
Lim SH, Ostrow D, Stall R, Chmiel J, Herrick A, Shoptaw S, et al. Changes in stimulant drug use over time in the MACS: evidence for resilience against stimulant drug use among men who have sex with men. AIDS Behav. 2012;16(1):151–8.
Montoya JL, Cattie J, Morgan E, Woods SP, Cherner M, Moore DJ, et al. The impact of age, HIV serostatus and seroconversion on methamphetamine use. Am J Drug Alcohol Abuse. 2016;42(2):168–77.
Kaslow RA, Ostrow DG, Detels R, Phair JP, Polk BF, Rinaldo CR Jr. The Multicenter AIDS Cohort Study: rationale, organization, and selected characteristics of the participants. Am J Epidemiol. 1987;126(2):310–8.
Austin PC. Optimal caliper widths for propensity-score matching when estimating differences in means and differences in proportions in observational studies. Pharm Stat. 2011;10(2):150–61.
Stuart EA, Rubin DB. Matching with multiple control groups with adjustment for group differences. J Educ Behav Stat. 2008;33:279–306.
Constantine NT. Serologic tests for the retroviruses: approaching a decade of evolution. AIDS. 1993;7(1):1–13.
Sander PM, Cole SR, Stall RD, Jacobson LP, Eron JJ, Napravnik S, et al. Joint effects of alcohol consumption and high-risk sexual behavior on HIV seroconversion among men who have sex with men. AIDS. 2013;27(5):815–23.
Mansergh G, Flores S, Koblin B, Hudson S, McKirnan D, Colfax GN. Alcohol and drug use in the context of anal sex and other factors associated with sexually transmitted infections: results from a multi-city study of high-risk men who have sex with men in the USA. Sex Transm Infect. 2008;84(6):509–11.
Woolf SE, Maisto SA. Alcohol use and risk of HIV infection among men who have sex with men. AIDS Behav. 2009;13(4):757–82.
Fritz K, Morojele N, Kalichman S. Alcohol: the forgotten drug in HIV/AIDS. Lancet. 2010;376(9739):398–400.
Kahler CW, Wray TB, Pantalone DW, Kruis RD, Mastroleo NR, Monti PM, et al. Daily associations between alcohol use and unprotected anal sex among heavy drinking HIV-positive men who have sex with men. AIDS Behav. 2015;19(3):422–30.
Shoptaw S, Peck J, Reback CJ, Rotheram-Fuller E. Psychiatric and substance dependence comorbidities, sexually transmitted diseases, and risk behaviors among methamphetamine-dependent gay and bisexual men seeking outpatient drug abuse treatment. J Psychoactive Drugs. 2003;35(Suppl 1):161–8.
Ramo DE, Grov C, Delucchi K, Kelly BC, Parsons JT. Typology of club drug use among young adults recruited using time-space sampling. Drug Alcohol Depend. 2010;107(2–3):119–27.
Semple SJ, Strathdee SA, Zians J, Patterson TL. Sexual risk behavior associated with co-administration of methamphetamine and other drugs in a sample of HIV-positive men who have sex with men. Am J Addict. 2009;18(1):65–72.
StataCorp. Stata 14.2 for Mac. College Station, TX: StataCorp; 2017.
Rabe-Hesketh S, Skrondal A. Multilevel and longitudinal modeling using Stata Volume II: categorical responses, counts, and survival. 3rd ed. College Station, TX: Stata Press; 2012.
West BT, Welch KB, Galecki AT. Linear mixed models: a practical guide using statistical software. 2nd ed. Boca Raton: Taylor & Francis Group; 2015.
Garofalo R, Mustanski BS, McKirnan DJ, Herrick A, Donenberg GR. Methamphetamine and young men who have sex with men: understanding patterns and correlates of use and association with HIV-related sexual risk. Arch Pediatr Adolesc Med. 2007;161:591–6.
Ellis RJ, Childers ME, Cherner M, Lazzaretto D, Letendre S, Grant I, The HIV Neurobehavioral Research Center Group. Increased human immunodeficiency virus loads in active methamphetamine users are explained by reduced effectiveness of antiretroviral therapy. J Infect Dis. 2003;188:1820–6.
Mor Z, Dan M. The HIV epidemic among men who have sex with men-behaviour beats science. EMBO Rep. 2012;13(11):948–53.
Becker JT, Kingsley LA, Molsberry S, Reynolds S, Aronow A, Levine AJ, et al. Cohort profile: recruitment cohorts in the neuropsychological substudy of the Multicenter AIDS Cohort Study. Int J Epidemiol. 2015;44(5):1506–16.
Reback CJ, Fletcher JB, Shoptaw S, Grella CE. Methamphetamine and other substance use trends among street-recruited men who have sex with men, from 2008 to 2011. Drug Alcohol Depend. 2013;133(1):262–5.
Brennan DJ, Welles SL, Miner MH, Ross MW, Rosser BR, Positive Connections T. HIV treatment optimism and unsafe anal intercourse among HIV-positive men who have sex with men: findings from the positive connections study. AIDS Educ Prev. 2010;22(2):126–37.
Rowniak S. Safe sex fatigue, treatment optimism, and serosorting: new challenges to HIV prevention among men who have sex with men. J Assoc Nurses AIDS Care. 2009;20(1):31–8.
Ostrow DG, Fox KJ, Chmiel JS, Silvestre A, Visscher BR, Vanable PA, Jacobson LP, Strathdee SA. Attitudes towards highly antiretroviral therapy are associated with sexual risk taking among HIV-infected and uninfected homosexual men. AIDS. 2002;16:775–80.
Fendrich M, Mackesy-Amiti ME, Johnson TP. Validity of self-reported substance use in men who have sex with men: comparisons with a general population sample. Ann Epidemiol. 2008;18(10):752–9.
Mackesy-Amiti ME, Fendrich M, Johnson TP. Prevalence of recent illicit substance use and reporting bias among MSM and other urban males. Addict Behav. 2008;33(8):1055–60.
Melendez-Torres GJ, Bourne A. Illicit drug use and its association with sexual risk behaviour among MSM: more questions than answers? Curr Opin Infect Dis. 2016;29(1):58–63.
Pantalone DW, Bimbi DS, Holder CA, Golub SA, Parsons JT. Consistency and change in sexual minority men’s club drug use in New York City, 2002–2007. Am J Public Health. 2010;100(10):1892–5.
Melendez-Torres GJ, Hickson F, Reid D, Weatherburn P, Bonell C. Nested event-level case-control study of drug use and sexual outcomes in multipartner encounters reported by men who have sex with men. AIDS Behav. 2016;20(3):646–54.
Acknowledgements
Data in this manuscript were collected by the Multicenter AIDS Cohort Study (MACS) and their participants. MACS (Principal Investigators): Johns Hopkins University Bloomberg School of Public Health (Joseph Margolick), U01-AI35042; Northwestern University (Steven Wolinsky), U01-AI35039; University of California, Los Angeles (Roger Detels, Oto Martinez-Maza), U01-AI35040; University of Pittsburg (Charles Rinaldo), U01-AI35041; the Center for Analysis and Management of MACS, Johns Hopkins University Bloomberg School of Public Health (Lisa Jacobson, Gypsyamber D’Souza), UM1-AI35043. The MACS is funded primarily by the National Institute of Allergy and Infectious Diseases (NIAID), with additional co-funding from the National Cancer Institute (NCI), the National Institute on Drug Abuse (NIDA), and the National Institute of Mental Health (NIMH). Targeted supplemental funding for specific projects was also provided by the National Heart, Lung, and Blood Institute (NHLBI), and the National Institute on Deafness and Communication Disorders (NIDCD). MACS data collection is also supported by UL1-TR001079 (JHU ICTR) from the National Center for Advancing Translational Sciences (NCATS) a component of the National Institutes of Health (NIH), and NIH Roadmap for Medical Research. The contents of this publication are solely the responsibility of the authors and do not represent the official views of the National Institutes of Health (NIH), Johns Hopkins ICTR, or NCATS. The MACS website is located at http://aidscohortstudy.org/.
Funding
None to report.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None to report
Ethical Approval
This article does not contain any studies with human participants performed by any of the authors.
Rights and permissions
About this article
Cite this article
Swartz, J.A., McCarty-Caplan, D. A Study of the Longitudinal Patterns of Stimulant and Amyl Nitrite Use and Sexual Behavior Pre- and Post-HIV Seroconversion Among MSM. AIDS Behav 22, 1395–1409 (2018). https://doi.org/10.1007/s10461-017-2008-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10461-017-2008-y