Abstract
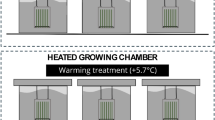
In epiphytic associations, cyanobacteria form the periphyton with phytoplanktonic algae and with aquatic macrophytes. In this study, we found homocytous and heterocytous filamentous strains of epiphytic cyanobacteria associated with submerged leaves of the aquatic fern Salvinia auriculata. Filamentous morphotypes can produce adaptive structures such as heterocysts, akinetes, and hormogonia. Based on the premise that light limitation and nutrient limitation affect the adaptive strategies of cyanobacteria epiphytic we hypothesized that the heterocysts production would be greater under nutrient scarcity and full sunlight conditions, akinetes would be predominantly produced under growth-limiting conditions, such as nutrient scarcity and shade, and hormogonia would be abundantly produced under shade. In addition, for purposes of recording, identification and assembling a collection, we carry out the isolation of cultivable cyanobacteria. We conducted an experiment in a greenhouse applying a shade cloth and Hoagland’s solution to manipulate, respectively, the light intensity and the concentration of nutrients. Both factors, light and nutrients, affected the production of adaptive structures. Heterocysts were produced in greater numbers when no nutrient was added and under full light. Akinetes were produced mainly in the treatments under shade and no nutrient addition. In its turn, hormogonia were produced in the treatments with nutrients and mainly in the shade. Accordingly, akinete differentiation was negatively correlated with that of hormogonia. We conclude that the non-addition of nutrients stimulates heterocysts production, as well as akinete production. Therefore, due to the BFN BNF performed in heterocysts, we suggest that in oligotrophic aquatic environments, cyanobacteria epiphytic on the roots of aquatic macrophytes can supply fixed nitrogen to the aquatic ecosystem. Besides, with the shading produced by macrophytes, and available nutrients, the production of hormogonia is stimulated due to positive phototaxis. At the end of the experiment, nine morphologically distinct strains were isolated and taxonomically classified, up to the level of family and genus, and will allow us to assemble a collection for future research.




Similar content being viewed by others
References
Adams DG, Duggan PS (1999) Tansley Review No. 107. Heterocyst and akinete differentiation in cyanobacteria. New Phytol 144(1):3–33. https://doi.org/10.1046/j.1469-8137.1999.00505.x
Adams DG, Duggan PS (2008) Cyanobacteria–bryophyte symbioses. J Exp Bot 59(5):1047–1058. https://doi.org/10.1093/jxb/ern005
Adams DG, Carr NG, Wilcox M (1981) The developmental biology of heterocyst and akinete formation in cyanobacteria. CRC Crit Rev Microbiol 9(1):45–100. https://doi.org/10.3109/10408418109104486
Agrawal SC (2012) Factors controlling induction of reproduction in algae—review: the text. Folia Microbiol 57(5):387–407. https://doi.org/10.1007/s12223-012-0147-0
Andrews JH, Harris RF (2000) The ecology and biogeography of microorganisms on plant surfaces. Annu Rev Phytopathol 38(1):145–180. https://doi.org/10.1146/annurev.phyto.38.1.145
Argueta C, Summers ML (2005) Characterization of a model system for the study of Nostoc punctiforme akinetes. Arch Microbiol 183(5):338–346. https://doi.org/10.1007/s00203-005-0778-5
Azevedo JL (1998) Biodiversidade microbiana e potencial biotecnológico. Ecologia Microbiana. EMBRAPA CNPMA, Jaguariúna
Azim ME, Asaeda T (2005) Periphyton structure, diversity and colonization. In: Azim ME, Verdegem MCJ, van Dam AA, Beveridge MCM (eds) Periphyton ecology exploitation and management. CABI, pp 15–33. https://doi.org/10.1079/9780851990965.0015
Azizan AA, Radzi R, Omar WMW, Convey P, Merican FMMS (2020) First records of morphological diversity and ecology of periphytic cyanobacteria from Tukun River, Penang Forest Reserve, Malaysia. Trop Life Sci Res 31(1):85–105. https://doi.org/10.21315/tlsr2020.31.1.6
Bergman B, Rai AN, Rasmussen U (2007) Cyanobacterial associations. In: Elmerich C, Newton WE (eds) Associative and endophytic nitrogen-fixing bacteria and cyanobacterial associations. Springer, Dordrecht, pp 257–301. https://doi.org/10.1007/1-4020-3546-2_12
Bicudo CEM, Menezes M (2017) Cyanobacteria. In: Gêneros de algas de águas continentais do Brasil: chave para identificação e descrições, 3ª ed. São Carlos: RiMa, pp 33–41
Buikema WJ, Haselkorn R (1991) Characterization of a gene controlling heterocyst differentiation in the cyanobacterium Anabaena 7120. Genes Dev 5(2):321–330. https://doi.org/10.1101/gad.5.2.321
Camargo VM, Ferragut C (2014) Estrutura da comunidade de algas perifíticas em Eleocharis acutangula (Roxb.) Schult (Cyperaceae) em reservatório tropical raso, São Paulo, SP. Brasil Hoehnea 41:31–40. https://doi.org/10.1590/S2236-89062014000100003
Canini A, Caiola MG (1993) Characterization of gonidial zone of Cycas revoluta coralloid roots by means of microelectrodes. FEMS Microbiol Lett 109(1):75–79. https://doi.org/10.1111/j.1574-6968.1993.tb06146.x
Canini A, Grilli Caiola M, Mascini M (1990) Ammonium content, nitrogenase activity and heterocyst frequency within the leaf cavities of Azolla filiculoides Lam. FEMS Microbiol Lett 71(1–2):205–210. https://doi.org/10.1111/j.1574-6968.1990.tb03823.x
Carrapiço F (2017) The Azolla–Anabaena–bacteria association: a case of symbiotic abduction? In: Grube M, Seckbach J, Muggia L (eds) Algal and cyanobacteria symbioses. World Scientific (Europe), pp 329–345. https://doi.org/10.1142/9781786340580_0010
Cmiech HA, Reynolds CS, Leedale GF (1984) Seasonal periodicity, heterocyst differentiation and sporulation of planktonic Cyanophyceae in a shallow lake, with special reference to Anabaena solitaria. Brit Phycol J 19(3):245–257. https://doi.org/10.1080/00071618400650271
Coelho FF, Lopes FS, Sperber CF (2005) Persistence strategy of Salvinia auriculata Aublet in temporary ponds of Southern Pantanal, Brazil. Aquat Bot 81(4):343–352. https://doi.org/10.1016/j.aquabot.2005.02.001
Dıez B, Ininbergs K (2014) Ecological importance of cyanobacteria. In: Sharma NK, Rai AK, Stal LJ (eds) Cyanobacteria: an economic perspective. Wiley, Chichester. https://doi.org/10.1002/9781118402238
Evans HJ, Burris RH (1992) Highlights in biological nitrogen fixation during the last 50 years. Biological Nitrogen Fixation, 1–42
Fay P (1988). Viability of akinetes of the planktonic cyanobacterium Anabaena circinalis. Proceedings of the Royal society of London. Series B. Biological sciences, 234(1276), 283–301. https://doi.org/10.1098/rspb.1988.0049
Fleming JP, Dibble ED (2015) Ecological mechanisms of invasion success in aquatic macrophytes. Hydrobiologia 746(1):23–37. https://doi.org/10.1007/s10750-014-2026-y
Gantar M, Kerby NW, Rowell P (1993) Colonization of wheat (Triticum vulgare L.) by N2-fixing cyanobacteria: III. The role of a hormogonia-promoting factor. New Phytol 124(3):505–513. https://doi.org/10.1111/j.1469-8137.1993.tb03842.x
Gonzalez A, Riley KW, Harwood TV, Zuniga EG, Risser DD (2019) A tripartite, hierarchical sigma factor cascade promotes hormogonium development in the filamentous cyanobacterium Nostoc punctiforme. Msphere 4(3):e00231-e319. https://doi.org/10.1128/mSphere.00231-19
Herdman M, Rippka R (1988) [22] Cellular differentiation: Hormogonia and baeocytes. Methods Enzymol 167:232–242. https://doi.org/10.1016/0076-6879(88)67025-X
Herdman M, Janvier M, Rippka R, Stanier RY (1979) Genome size of cyanobacteria. Microbiology 111(1):73–85. https://doi.org/10.1099/00221287-111-1-73
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Circular. California agricultural experiment station, 347(2nd edit)
Julien MH, Hill MP, Tipping PW (2009) Salvinia molesta DS Mitchell (Salviniaceae). Weed biological control with arthropods in the tropics. Cambridge University Press, Cambridge, pp 378–407. https://doi.org/10.1111/epp.12428
Julien MH, Center TD, Tipping PW (2002) Floating fern (Salvinia). Biological control of invasive plants in the Eastern United States, 17–32
Kaplan-Levy RN, Hadas O, Summers ML, Ru¨cker J, Sukenik A (2010) Akinetes - dormant cells of cyanobacteria. In: Lubzens E, Cerda J, Clark MS (eds) Topics in Current Genetics. Springer-Verlag: Berlin, Heidelberg
Komárek J, Kaštovský J, Mareš J, Johansen JR (2014) Taxonomic classification of cyanoprokaryotes (cyanobacterial genera) 2014, using a polyphyletic approach. Preslia 86:295–335
Komarzewska K, Glogowska B (2005) Blooming of Aphanizomenon flos-aquae in the urban pond. Oceanol Hydrobiol Stud 34(3):105
Lang JD, Haselkorn ROBERT (1991) A vector for analysis of promoters in the cyanobacterium Anabaena sp. strain PCC 7120. J Bacteriol 173(8):2729–2731. https://doi.org/10.1128/jb.173.8.2729-2731.1991
Lazaroff N (1973) Photomorphogenesis and nostocacean development. Botanical monographs
Maldener I, Summers ML, Sukenik A (2014) Cellular differentiation in filamentous cyanobacteria. Cell Biol Cyanobacteria 15:263–291. https://doi.org/10.21775/9781908230508
Meeks JC, Elhai J (2002) Regulation of cellular differentiation in filamentous cyanobacteria in free-living and plant-associated symbiotic growth states. Microbiol Mol Biol Rev 66(1):94–121. https://doi.org/10.1128/MMBR.66.1.94-121.2002
Meeks JC, Wycoff KL, Chapman JS, Enderlin CS (1983) Regulation of expression of nitrate and dinitrogen assimilation by Anabaena species. Appl Environ Microbiol 45(4):1351–1359. https://doi.org/10.1128/aem.45.4.1351-1359.1983
Meeks JC, Enderlin CS, Joseph CM, Chapman JS, Lollar MWL (1985) Fixation of [13 N] N 2 and transfer of fixed nitrogen in the Anthoceros-Nostoc symbiotic association. Planta 164(3):406–414. https://doi.org/10.1007/BF00402954
Meeks JC, Steinberg NA, Enderlin CS, Joseph CM, Peters GA (1987) Azolla-Anabaena relationship: XIII. Fixation of [13N] N2. Plant Physiol 84(3):883–886. https://doi.org/10.1104/pp.84.3.883
Meeks JC, Campbell EL, Summers ML, Wong FC (2002) Cellular differentiation in the cyanobacterium Nostoc punctiforme. Arch Microbiol 178(6):395–403. https://doi.org/10.1007/s00203-002-0476-5
Mohamed ZA, Al- Shehri AM (2010) Differential responses of epiphytic and planktonic toxic cyanobacteria to allelopathic substances of the submerged macrophyte Stratiotes aloides. Int Rev Hydrobiol 95(3):224–234. https://doi.org/10.1002/iroh.200911219
Moore D, McGregor GB, Shaw G (2004) Morphological changes during akinete germination in Cylindrospermopsis raciborskii (nostocales, cyanobacteria) 1. J Phycol 40(6):1098–1105. https://doi.org/10.1111/j.1529-8817.2004.04050.x
Pereira AL, Carrapiço F (2007) Histochemistry of simple hairs from the foliar cavities of Azolla filiculoides. Plant Biosyst 141(3):323–328. https://doi.org/10.1080/11263500701627588
Pimenta LL (2020) Efeito da limitação de luz e nutrientes na alocação para estruturas adaptativas em cianobactérias associadas epifiticamente às raízes de Salvinia auriculata. Universidade Federal de Lavras, Dissertação de mestrado
Postius C, Neuschaefer-Rube O, Haid V, Böger P (2001) N2-fixation and complementary chromatic adaptation in non-heterocystous cyanobacteria from Lake Constance. FEMS Microbiol Ecol 37(2):117–125. https://doi.org/10.1111/j.1574-6941.2001.tb00859.x
Raymond J, Siefert JL, Staples CR, Blankenship RE (2004) The natural history of nitrogen fixation. Mol Biol Evol 21(3):541–554. https://doi.org/10.1093/molbev/msh047
Reed SC, Cleveland CC, Townsend AR (2011) Functional ecology of free-living nitrogen fixation: a contemporary perspective. Annu Rev Ecol Evol Syst 42:489–512. https://doi.org/10.1146/annurev-ecolsys-102710-145034
Rippka R, Deruelles J, Waterbury JB, Herdman M, Stanier RY (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. Microbiology 111(1):1–61. https://doi.org/10.1099/00221287-111-1-1
Room PM (1983) Falling apart’as a lifestyle: the rhizome architecture and population growth of Salvinia molesta. J Ecol. https://doi.org/10.2307/2259719
Silvester WB, Parsons R, Watt PW (1996) Direct measurement of release and assimilation of ammonia in the Gunnera-Nostoc symbiosis. New Phytol 132(4):617–625. https://doi.org/10.1111/j.1469-8137.1996.tb01880.x
Sukenik A, Beardall J, Hadas O (2007) Photosynthetic characterization of developing and mature akinetes of Aphanizomenon ovalisporum (Cyanoprokaryota) 1. J Phycol 43(4):780–788. https://doi.org/10.1111/j.1529-8817.2007.00374.x
Sukenik A, Kaplan-Levy RN, Viner-Mozzini Y, Quesada A, Hadas O (2013) Potassium deficiency triggers the development of dormant cells (akinetes) in Aphanizomenon ovalisporum (Nostocales, Cyanoprokaryota). J Phycol 49(3):580–587. https://doi.org/10.1111/jpy.12069
Tandeau de Marsac N, Houmard J (1993) Adaptation of cyanobacteria to environmental stimuli: new steps towards molecular mechanisms. FEMS Microbiol Rev 10(1–2):119–189. https://doi.org/10.1111/j.1574-6968.1993.tb05866.x
Thompson AW, Zehr JP (2013) Cellular interactions: lessons from the nitrogen-fixing cyanobacteria. J Phycol 49(6):1024–1035. https://doi.org/10.1111/jpy.12117
Wolk CP, Ernst A, Elhai J (1994) Heterocyst metabolism and development. In: Bryant DA (ed) The molecular biology of cyanobacteria. Springer, Dordrecht, pp 769–823. https://doi.org/10.1007/978-94-011-0227-8_27
Acknowledgements
The authors are grateful to Antônio Galvão do Nascimento, for his support and guidance in the early stages of our studies on the “cyanobacteria field”; to Mirian Silva Braz, for assisting in statistical analyses; and to the Plant Anatomy Laboratory and to the professors Whasley Ferreira Duarte and Victor Satler Pylro (Federal University of Lavras), for providing the required research equipment and materials. This study was developed during the M. Sc. program of the first author. Moreover, we are grateful for the scholarship granted by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) to MGMVV (PNPD-1638006).
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Not applicable.
Availability of data and material
All of our data will be made available for consultation, if required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Handling Editor: Ted Harris.
Rights and permissions
About this article
Cite this article
Pimenta, L.L., Lima, G.P., Biondi, M. et al. Epiphytic cyanobacterial strains in the roots of Salvinia auriculata and the effect of light and nutrients on the production of heterocyst, akinete and hormogonia. Aquat Ecol 56, 543–553 (2022). https://doi.org/10.1007/s10452-021-09922-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-021-09922-w