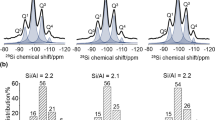
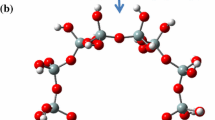
Abstract
Anthropogenic CO2 emissions are the major drivers of global warming and climate change. The adsorption of CO2 is one of the strategies to mitigate these emissions. In this sense, different materials have been used as adsorbents as is the case of zeolites. In this work,experimental infrared and X-ray analyses together with DFT (Density Functional Theory)theoretical calculations were employed to study the adsorption of CO2 on mordenite zeolites (Na-MOR). Ion exchange followed by chemical analysis showed that the Na+ of Na-MOR was not completely substituted under the experimental conditions employed. The XRD (X-ray Diffraction) analysis indicated that Na+ exchanged for Li+ did not affect the crystallinity of the materials. The infrared results showed that CO2 interacted linearly with the sodium and lithium cations present in the main channel of the mordenite framework. Sodium in dry samples favored the formation of carbonates and hydroxycarbonate species, as did the presence of low water content in the LixNa-MOR. The DFT periodical calculation of dehydrated mordenite models led to CO2 adsorption energies in the range of − 31.6 to − 34.2 kJ mol−1, with the highest adsorption energy found for sodium mordenite.The asymmetric stretching ν3 (CO2) vibration mode resulted intense and presented a shift which indicated a higher CO2 interaction of Na-MOR. The presence of some water molecules in the mordenite framework caused a decrease in the adsorption energy.











Similar content being viewed by others
References
Bachmann, T.M.: Considering environmental costs of greenhouse gas emissions for setting a CO2 tax: a review. Sci. Total Environ. 720, 137524 (2020)
Nakao, S.I., Yogo, K., Goto, K., Kai, T., Yamada, H.: Advanced CO2 capture technologies absorption adsorption and membrane separation methods. Springer International Publishing, Cham (2019)
Pelletier, C., Rogaume, Y., Dieckhoff, L., Bardeau, G., Pons, M., Dufour, A.: Effect of combustion technology and biogenic CO2 impact factor on global warming potential of wood-to-heat chains. Appl. Energy 235, 1381–13881 (2019)
Alonso, A., Moral-Vico, J., Markeb, A., Busquets-Fité, M., Komilis, D., Puntes, V., Sánchez, A., Font, X.: Critical review of existing nanomaterial adsorbents to capture carbon dioxide and methane. Sci. Total Environ. 595, 51–62 (2017)
Hussin, F., Aroua, M.: Recent trends in the development of adsorption technologies for carbon dioxide capture: a brief literature and patent reviews (2014–2018). J. Clean. Prod. 253, 119707 (2020)
Cabana, N., Serra, R., Boix, A., Bolcatto, P.: Geometries and interaction energies of toluene on CsxNa1–xMordenite. Basic Solid State Phys. (2020). https://doi.org/10.1002/pssb.202000131
Serra, R., Miró, E., Sapag, M.K., Boix, A.: Adsorption and diffusion of toluene on Na and Cs mordenites for hydrocarbon traps. Microporous Mesoporous Mater. 138, 102–109 (2011)
Serra, R., Miró, E., Bolcatto, P., Boix, A.: Experimental and theoretical studies about the adsorption of toluene on ZSM5 and mordenite zeolites modified with Cs. Microporous Mesoporous Mater. 147, 17–29 (2012)
Serra, R., Aspromonte, A., Miró, E., Boix, A.: Hydrocarbon adsorption and NOx-SCR on (Cs, Co) mordenite. Appl. Catal. B 166, 592–602 (2015)
Sels, B. F., Kustov, L. M. (eds.) Zeolites and zeolite-like materials, Elsevier (2016). Zeolyst International Available from: http://www.zeolyst.com.
Torres, F.J., Vitillo, J.G., Civalleri, B., Ricchiardi, G., Zecchina, A.: Interaction of H2 with alkali-metal-exchanged zeolites:a quantum mechanical study. J. Phys. Chem. C 111, 2505–2513 (2007)
Siriwardane, R.V., Shen, M., Fisher, E.P.: Adsorption of CO2 on zeolites at moderate temperatures. Energ. Fuel. 19, 1153–1159 (2005)
Stevens, R., Siriwardane, R., Jr., Logan, J.: In situ fourier transform infrared (FTIR) investigation of CO2 adsorption onto zeolite materials. Energy Fuel 22, 3070–3079 (2008)
Villarreal, A., Garbarino, G., Riani, P., Finocchio, E., Bosio, B., Ramírez, J., Busca, G.: Adsorption and separation of CO2 from N2-rich gas on zeolites: Na-X faujasite vs Na-mordenite. J. CO2 Util. 19, 266–275 (2017)
Bonelli, B., Civalleri, B., Fubini, B., Ugliengo, P., Areán, C., Garrone, E.: Experimental and quantum chemical studies on the adsorption of carbon dioxide on alkali-metal-exchanged ZSM-5 zeolites. J. Phys. Chem. B 104, 10978–10988 (2000)
Larin, A.V.: Role of cation size for hydrogen carbonate stabilization and modification of the zeolite CO2 interaction energy: computational analysis in alkali Y zeolites. Microporous Mesoporous Mater. 228, 182–195 (2016)
Walton, K., Abney, M., Douglas Levan, M.: CO2 adsorption in Y and X zeolites modified by alkali metal cation exchange. Microporous and Mesoporous Mater. 91, 78–84 (2006)
Maurin, G., Bell, R., Devautour, S., Hennand, F., Giuntini, J.: Modelling the effect of hydration in zeolite Na+-mordenite. J. Phys. Chem. B. 108, 3739–3745 (2004)
Vilhena, F.S., Serra, R., Boix, A., Ferreira, G.B., Carneiro, J.W.M.: DFT study of Li+ and Na+ positions in mordenites and hydration stability. Comput. Theor. Chem. 1091, 115–121 (2016)
Barthomeuf, D.: Basic zeolites characterization and uses in adsorption and catalysis. Catal. Rev. Sci. Eng. 38(4), 521–612 (1996)
Pham, T., Lobo, R.: Adsorption equilibria of CO2 and small hydrocarbons in AEI-, CHA-, STT-, and RRO-type siliceous zeolites. Microporous Mesoporous Mater. 236, 100–108 (2016)
Zeolyst International (2019) Available from: https://www.zeolyst.com
Lippens, B.C., Linsen, B.G., de Boer, J.H.: Studies on pore systems in catalysts. J. Catal. 3, 32–37 (1964)
Holzapfel, M., Haak, C., Ott, A.: Lithium-ion conductors of the system LiCo12xFexO2, preparation and structural investigation. J. Solid State Chem. 156, 470–479 (2001)
Coelho, A.: TOPAS-Academic v5. Coehlo Software, Brisbane, Australia (2012)
Rietveld, H.M.: A profile refinement method for nuclear and magnetic structures. J. Appl. Cryst. 2, 65–71 (1969)
Klug, H.P., Alexander, L.E.: X-ray difraction procedures for polycrystalline and amorphous material. Wiley-Interscience, New York (1974)
IZA: Database of Zeolite structure (2019). http://www.iza-structure.org/database/.
Baerlocher, Ch., Lynne, B., McCusker, D.H., Olson, D.H.: Atlas of zeolite framework types, 6th edn., pp. 218–219. Elsevier, Amsterdam (2007)
Cejka, J., Corma, A., Zones, S., Weinheim, J.: Zeolites and catalysis: Synthesis reactions and applications. John Wiley and Sons, USA (2010)
Blochl, P.E., Forst, C.J., Schimpl, J.: Projector augmented wave method: ab initio molecular dynamics with full wave functions. Bull. Mater. Sci. 26, 33–41 (2003)
Perdew, J.P., Burke, K., Ernzerhof, M.: Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996)
VandeVondele, J., Hutter, J.: Gaussian basis sets for accurate calculations on molecular systems in gas and condensed phases. J. Chem. Phys. 127, 114105–114109 (2007)
Grimme, S.: Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006)
Breck, D.: Zeolite molecular sieves, pp. 530–536. John Wiley and Sons, USA (1974)
Geobaldo, F., Lamberti, C., Ricchiardi, G., Bordiga, S., Zecchina, A., Palomino, G., Areán, C.: N2 adsorption at 77 K on H-mordenite and alkali-metal-exchanged mordenites: an IR study. J. Phys. Chem. 99, 11167–11177 (1995)
Treacy, M.M.J., Higgins, J.B.: Collection of simulated XRD powder patterns for zeolites. Elsevier, Amsterdam (2007)
Boix, A., García Fierro, J.L.: XPS analysis of platinum and/or cobalt-loaded zeolites relevant for SCR reaction. Surf. Interface Anal. 27, 1107–1113 (1999)
Karge, H., Weitkamp, J. 2003 “Molecular sieves, characterization I” Springer
Garrone, E., Bonelli, B., Lamberti, C., Civalleri, B., Rocchia, M., Roy, P., Otero, C.: Coupling of framework modes and adsorbate vibrations for CO2 molecularly adsorbed on alkali ZSM-5 zeolites: Mid- and far-infrared spectroscopy and ab initio modeling. J. ChemPhys 117, 10274–10282 (2002)
Martra, G., Coluccia, S., Davit, P., Gianotti, E., Marchese, L., Tsuji, H., Hattori, H.: Acidic and basic sites in NaX and NaY faujasites investigated by NH3, CO2 and CO molecular probes. Res. Chem. Intermed. 25, 77–93 (1999)
Martra, G., Ocule, R., Marchese, L., Centi, G., Coluccia, S.: Alkali and alkaline-earth exchanged faujasites: strength of lewis base and acid centres and cation site occupancy in Na- and BaY and Na- and BaX zeolites. Catal. Today 73, 83–93 (2002)
Maurin, G., Llewellyn, P.L., Bell, R.: Adsorption mechanism of carbon dioxide in faujasites: grand canonical Monte Carlo simulations and microcalorimetry measurements. J. Phys. Chem. B 109, 16084–16091 (2005)
Wong-Ng, W., Kaduk, J.A., Huang, Q., Espinal, L., Li, L., Burress, J.W.: Investigation of NaY Zeolite with adsorbed CO2 by neutron powder diffraction. Microporous and Mesoporous Mater. 172, 95–104 (2013)
Joos, L., Swisher, J.A., Smit, B.: Molecular simulation study of the competitive adsorption of H2O and CO2 in zeolite 13X. Langmuir 29, 15936–15942 (2013)
Pulido, A., Delgado, M.R., Bludsky, O., Rubes, M., Nachtigall, P., Areán, C.O.: Combined DFT/CC and IR spectroscopic studies on carbon dioxide adsorption on the zeolite H-FER. Energy Environ. Sci. 2, 1187–1195 (2009)
Chu, C.T.W., Chang, C.D.: Isomorphous substitution in zeolite frameworks. Acidity of surface hydroxyls in [B]-, [Fe]-, [Ga]-, and [AI]-ZSM-5. J. Phys. Chem. 89, 1569–1571 (1985)
Acknowledgements
We would like to thank Agencia Nacional de Promoción Científica y Tecnología (ANPCyT) and CONICET for the financial support of this study. This research was supported by a grant from the Brazilian agency CAPES (PNPD program) and by the Bilateral Cooperation Project FAPERJ-CONICET (Grant Number E-26/110.041/2014).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Serra, R.M., de Vilhena, F.S., Gutierrez, L.B. et al. Experimental and theoretical investigation of the Na+ → Li+ cation exchange in mordenite and its effect on CO2 adsorption properties. Adsorption 27, 891–907 (2021). https://doi.org/10.1007/s10450-020-00288-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-020-00288-3