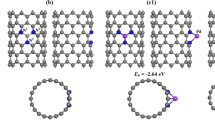
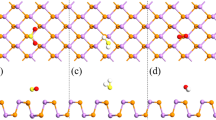
Abstract
Detecting the species and concentrations of SF6 decomposed products is crucial for on-line monitoring the running status of SF6-insulated equipment. TiO2-based gas sensing material shows a great potential in SF6 decomposed products detection. In order to improve the sensitivity and selectivity of TiO2-based gas sensing material, Pd atom modified TiO2 (Pd–TiO2) was proposed to analyze its adsorption properties to three characteristic decomposition products of SF6: SO2, SOF2, and SO2F2. The results show that Pd–TiO2 possesses strong adsorption property to these gas molecules because of the high chemical activity of the doped Pd atom. Density of states, differential charge density, and molecular orbits are studied to analyze the gas sensing mechanism of Pd–TiO2 to the gas molecules. It is found that gas molecules adsorption raises an increase of conductivity to different extents, which can be applied to identify the species and concentrations of SF6 decomposed products under electric discharge. Therefore, Pd–TiO2 can be a promising gas sensing material using in SO2, SOF2 and SO2F2 detection with high selectivity and sensitivity. This plays an important role in the detection of SF6 decomposition gas.













Similar content being viewed by others
References
Basiuk, V.A., Henao-Holguin, L.V.: Dispersion-corrected density functional theory calculations of meso-tetraphenylporphine–C-60 complex by using DMol3 module. J. Comput. Theor. Nanosci. 11(7), 1609–1615 (2014). https://doi.org/10.1166/jctn.2014.3539
Brandbyge, M., Mozos, J.L., Ordejon, P., Taylor, J., Stokbro, K.: Density-functional method for nonequilibrium electron transport. Phys. Rev. B 65(16), 17 (2002). https://doi.org/10.1103/physrevb.65.165401
Çakır, D., Gülseren, O.: Adsorption of Pt and bimetallic PtAu clusters on the partially reduced rutile (110) TiO2 surface: a first-principles study. J. Phys. Chem. C 116(9), 5735–5746 (2012)
Cui, H., Zhang, X.X., Zhang, J., Tang, J.: Adsorption behaviour of SF6 decomposed species onto Pd4-decorated single-walled CNT: a DFT study. Mol. Phys. 116(13), 1749–1755 (2018). https://doi.org/10.1080/00268976.2018.1451930
Cui, H., Chen, D.C., Zhang, Y., Zhang, X.X.: Dissolved gas analysis in transformer oil using Pd catalyst decorated MoSe2 monolayer: a first-principles theory. Sustain. Mater. Technol. 20, 8 (2019a). https://doi.org/10.1016/j.susmat.2019.e00094
Cui, H., Zhang, X.X., Chen, D.C., Tang, J.: Pt and Pd decorated CNT as a workable media for SOF2 sensing: a DFT study. Appl. Surf. Sci. 471, 335–341 (2019b). https://doi.org/10.1016/j.apsusc.2018.12.016
Cui, H., Zhang, X.X., Zhang, G.Z., Tang, J.: Pd-doped MoS2 monolayer: a promising candidate for DGA in transformer oil based on DFT method. Appl. Surf. Sci. 470, 1035–1042 (2019c). https://doi.org/10.1016/j.apsusc.2018.11.230
Dong, X.C., Zhang, X.X., Cui, H., Zhang, J.: A first principle simulation of competitive adsorption of SF6 decomposition components on nitrogen-doped anatase TiO2 (101) surface. Appl. Surf. Sci. 422, 331–338 (2017). https://doi.org/10.1016/j.apsusc.2017.06.039
Fabregat-Santiago, F., Garcia-Belmonte, G., Bisquert, J., Zaban, A., Salvador, P.: Decoupling of transport, charge storage, and interfacial charge transfer in the nanocrystalline TiO2/electrolyte system by impedance methods. J. Phys. Chem. B 106(2), 334–339 (2002)
Fehsenfeld, F.C.: Electron attachment to SF6. J. Chem. Phys. 53(5), 2000 (1970). https://doi.org/10.1063/1.1674280
Grimme, S.: Accurate description of van der Waals complexes by density functional theory including empirical corrections. J. Comput. Chem. 25(12), 1463–1473 (2004). https://doi.org/10.1002/jcc.20078
Grimme, S.: Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27(15), 1787–1799 (2006). https://doi.org/10.1002/jcc.20495
Guo, H., Zhao, C., Zheng, Q., Lan, Z., Prezhdo, O.V., Saidi, W.A., Zhao, J.: Superatom molecular orbital as an interfacial charge separation state. J. Phys. Chem. Lett (2018). https://doi.org/10.1021/acs.jpclett.8b01302
Jain, A., Hautier, G., Ong, S.P., Moore, C.J., Fischer, C.C., Persson, K.A., Ceder, G.: Formation enthalpies by mixing GGA and GGA plus U calculations. Phys. Rev. B 84(4), 10 (2011). https://doi.org/10.1103/physrevb.84.045115
Kline, L.E., Davies, D.K., Chen, C.L., Chantry, P.J.: Dielectric-properties for SF6 and SF6 mixtures predicted from basic data. J. Appl. Phys. 50(11), 6789–6796 (1979). https://doi.org/10.1063/1.325814
Kolokoltsev, Y., Amelines-Sarria, O., Gromovoy, T.Y., Basiuk, V.A.: Interaction of meso-tetraphenylporphines with C-60 fullerene: comparison of several density functional theory functionals implemented in DMol3 module. J. Comput. Theor. Nanosci. 7(6), 1095–1103 (2010). https://doi.org/10.1166/jctn.2010.1457
Maiss, M., Brenninkmeijer, C.A.M.: Atmospheric SF6: trends, sources, and prospects. Environ. Sci. Technol. 32(20), 3077–3086 (1998). https://doi.org/10.1021/es9802807
Methfessel, M., Paxton, A.T.: High-precision sampling for Brillouin-zone integration in metals. Phys. Rev. B 40(6), 3616–3621 (1989). https://doi.org/10.1103/physrevb.40.3616
Monkhorst, H.J., Pack, J.D.: Special points for Brillouin-zone integrations. Phys. Rev. B 13(12), 5188–5192 (1976). https://doi.org/10.1103/physrevb.13.5188
Ozgonenel, O., Font, A., Ilhan, S.: IEEE: electrostatic analysis of SF6 gas insulated distribution transformer. In: 2016 National Conference on Electrical, Electronics and Biomedical Engineering. IEEE, New York (2016)
Ozgonenel, O., Thomas, D.: Modeling and simulation of 2.5 MVA SF6-gas insulated transformer. Turk. J. Electr. Eng. Comput. Sci. 25(4), 3475–3485 (2017). https://doi.org/10.3906/elk-1603-108
Perdew, J.P.: Density-functional approximation for the correlation-energy of the inhomogeneous electron-gas. Phys. Rev. B 33(12), 8822–8824 (1986). https://doi.org/10.1103/physrevb.33.8822
Rangan, S., Ruggieri, C., Bartynski, R.A., Martinez, J.I., Flores, F., Ortega, J.: Adsorption geometry and energy level alignment at the PTCDA/TiO2 (110) interface. J. Phys. Chem. B (2017). https://doi.org/10.1021/acs.jpcb.7b04227
Schierbaum, K.D., Xu, W.X.: The electronic structure of intrinsic defects at TiO2 (110) surfaces: an ab initio molecular orbital study. Int. J. Quantum Chem. 57(6), 1121–1129 (1996)
Solomon, J.S., Baun, W.L.: Molecular orbital effects on the Ti LMV auger spectra of TiO and TiO2. Surf. Sci. 51(1), 228–236 (1976)
Steeve, C., Horia, M.: Enhanced adsorption energy of Au1 and O2 on the stoichiometric TiO2 (110) surface by coadsorption with other molecules. J. Chem. Phys. 128(4), 7896 (2008)
Tang, J., Liu, F., Zhang, X.X., Meng, Q.H., Zhou, J.B.: Partial discharge recognition through an analysis of SF6 decomposition products. Part 1: decomposition characteristics of SF6 under four different partial discharges. IEEE Trans. Dielectr. Electr. Insul. 19(1), 29–36 (2012). https://doi.org/10.1109/tdei.2012.6148499
Tang, J., Zeng, F.P., Pan, J.Y., Zhang, X.X., Yao, Q., He, J.J., Hou, X.Z.: Correlation analysis between formation process of SF6 decomposed components and partial discharge qualities. IEEE Trans. Dielectr. Electr. Insul. 20(3), 864–875 (2013). https://doi.org/10.1109/tdei.2013.6518956
Wang, Y., Gui, Y.G., Ji, C., Tang, C., Zhou, Q., Li, J., Zhang, X.X.: Adsorption of SF6 decomposition components on Pt3–TiO2 (101) surface: a DFT study. Appl. Surf. Sci. 459, 242–248 (2018). https://doi.org/10.1016/j.apsusc.2018.07.219
Zhang, X.X., Zhang, J.B., Jia, Y.C., Xiao, P., Tang, J.: TiO2 nanotube array sensor for detecting the SF6 decomposition product SO2. Sensors 12(3), 3302–3313 (2012). https://doi.org/10.3390/s120303302
Zhang, X.X., Chen, Q.C., Hu, W.H., Zhang, J.B.: A DFT study of SF6 decomposed gas adsorption on an anatase (101) surface. Appl. Surf. Sci. 286, 47–53 (2013a). https://doi.org/10.1016/j.apsusc.2013.09.005
Zhang, X.X., Tie, J., Zhang, J.B.: A Pt-doped TiO2 nanotube arrays sensor for detecting SF6 decomposition products. Sensors 13(11), 14764–14776 (2013b). https://doi.org/10.3390/s131114764
Zhang, X.X., Tie, J., Chen, Q.C., Xiao, P., Zhou, M.: Pt-doped TiO2-based sensors for detecting SF6 decomposition components. IEEE Trans. Dielectr. Electr. Insul. 22(3), 1559–1566 (2015). https://doi.org/10.1109/tdei.2014.004256
Zhang, X., Gui, Y., Xiao, H., Zhang, Y.: Analysis of adsorption properties of typical partial discharge gases on Ni-SWCNTs using density functional theory. Appl. Surf. Sci. 379, 47–54 (2016)
Zhang, X.X., Zhang, J., Dong, X.C., Cui, H.: A DFT calculation of fluoride-doped TiO2 nanotubes for detecting SF6 decomposition components. Sensors 17(8), 14 (2017). https://doi.org/10.3390/s17081907
Zhang, X.X., Zhang, J., Cui, H.: Adsorption mechanism of SF6 decomposition components onto N, F-co-doped TiO2: a DFT study. J. Fluor. Chem. 213, 18–23 (2018). https://doi.org/10.1016/j.jfluchem.2018.05.014
Zhao, Y., Schultz, N.E., Truhlar, D.G.: Design of density functionals by combining the method of constraint satisfaction with parametrization for thermochemistry, thermochemical kinetics, and noncovalent interactions. J. Chem. Theory Comput. 2(2), 364–382 (2006). https://doi.org/10.1021/ct0502763
Acknowledgements
This work is supported by The National Key R&D Program of China (Grant Nos. 2017YFB0902700, 2017YBF0902702), The Chongqing Research Program of Basic Research and Frontier Technology (No. cstc2018jcyjAX0068) and The Fundamental Research Funds for the Central Universities (Grant No. SWU118030).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no competing financial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, H., Gui, Y., Wei, H. et al. DFT study of SF6 decomposed products on Pd–TiO2: gas sensing mechanism study. Adsorption 25, 1643–1653 (2019). https://doi.org/10.1007/s10450-019-00150-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-019-00150-1