Abstract
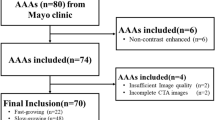
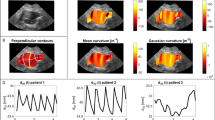
Abdominal aortic aneurysms (AAA) are localized, commonly occurring aortic dilations. Following rupture only immediate treatment can prevent morbidity and mortality. AAA maximal diameter and growth are the current metrics to evaluate the associated risk and plan intervention. Although these criteria alone lack patient specificity, predicting their evolution would improve clinical decision. If the disease is known to be associated with altered morphology and blood flow, intraluminal thrombus deposit and clinical symptoms, the growth mechanisms are yet to be fully understood. In this retrospective longitudinal study of 138 scans, morphological analysis and blood flow simulations for 32 patients with clinically diagnosed AAAs and several follow-up CT-scans, are performed and compared to 9 control subjects. Several metrics stratify patients between healthy, low and high risk groups. Local correlations between hemodynamic metrics and AAA growth are also explored but due to their high inter-patient variability, do not explain AAA heterogeneous growth. Finally, high-risk predictors trained with successively clinical, morphological, hemodynamic and all data, and their link to the AAA evolution are built from supervise learning. Predictive performance is high for morphological, hemodynamic and all data, in contrast to clinical data. The morphology-based predictor exhibits an interesting effort-predictability tradeoff to be validated for clinical translation.








Similar content being viewed by others
Notes
Approval \(\#12.153\) from the research ethics committee of the University of Montreal Health Centre (CHUM).
References
Antiga, L. Patient-specific modeling of geometry and blood flow in large arteries. Politecnico di Milano, 2002.
Antiga, L., B. Ene-Iordache, and A. Remuzzi. Computational geometry for patient-specific reconstruction and meshing of blood vessels from MR and CT angiography. IEEE Trans. Med. Imaging 22:674–684, 2003.
Antiga, L. and D. A. Steinman. Robust and objective decomposition and mapping of bifurcating vessels. IEEE Trans. Med. Imaging 23:704–713, 2004.
Arzani, A., G.-Y. Suh, R. L. Dalman, and S. C. Shadden. A longitudinal comparison of hemodynamics and intraluminal thrombus deposition in abdominal aortic aneurysms. Am. J. Physiol. 307:H1786–H1795, 2014.
Basciano, C., C. Kleinstreuer, S. Hyun, and E. A. Finol. A relation between near-wall particle-hemodynamics and onset of thrombus formation in abdominal aortic aneurysms. Ann. Biomed. Eng. 39:2010–2026, 2011.
Bertoglio, C., A. Caiazzo, Y. Bazilevs, M. Braack, M. Esmaily, V. Gravemeier, A. L. Marsden, O. Pironneau, I. E. Vignon-Clementel, and W. A. Wall. Benchmark problems for numerical treatment of backflow at open boundaries. International Journal for Numerical Methods in Biomedical Engineering , 2018.
Bhagavan, D., P. D. Achille, and J. D. Humphrey. Strongly coupled morphological features of aortic aneurysms Drive intraluminal thrombus. Sci. Rep. pp. 1–18, 2018.
Boyd, A. J., D. C. Kuhn, R. J. Lozowy, and G. P. Kulbisky. Low wall shear stress predominates at sites of abdominal aortic aneurysm rupture. J. Vasc. Surg. 63:1613–1619, 2016.
Brady, A. R., Thompson, S. G., Fowkes, F. G. R., Greenhalgh, R. M., and Powell, J. T. Abdominal aortic aneurysm expansion: risk factors and time intervals for surveillance. Circulation, 110(1), 16-21, 2004.
Breunig, M. M., H.-P. Kriegel, R. T. Ng, and J. Sander. LOF: Identifying Density-Based Local Outliers. Proceedings of the 2000 Acm Sigmod International Conference on Management of Data, 2000.
Brewster, D. C., J. L. Cronenwett, J. W. Hallett, K. W. Johnston, W. C. Krupski, and J. S. Matsumura. Guidelines for the treatment of abdominal aortic aneurysms: report of a subcommittee of the Joint Council of the American Association for Vascular Surgery and Society for Vascular Surgery. J. Vasc. Surg. 37:1106–1117, 2003.
Buchanan, J. R., C. Kleinstreuer, and J. K. Comer. Rheological effects on pulsatile hemodynamics in a stenosed tube. Comput. Fluids 29:695–724, 2000.
Busa-Fekete, R., B. Kégl, G. Szarvas, O. Chapelle, Y. Chang, and T.-Y. Liu. Ranking by calibrated AdaBoost. In: Learning to Rank Challenge, volume 14, pp. 37–48. 2011.
Chaikof, E. L., J. D. Blankensteijn, P. L. Harris, G. H. White, C. K. Zarins, V. M. Bernhard, J. S. Matsumura, J. May, F. J. Veith, M. F. Fillinger, R. B. Rutherford, and K. Craig Kent. Reporting standards for endovascular aortic aneurysm repair. J. Vasc. Surg. 35(5):1048–1060, 2002.
Chaikof, E. L., R. L. Dalman, M. K. Eskandari, B. M. Jackson, W. A. Lee, M. A. Mansour, T. M. Mastracci, M. Mell, M. H. Murad, L. L. Nguyen, G. S. Oderich, M. S. Patel, M. L. Schermerhorn, and B. W. Starnes. The Society for Vascular Surgery practice guidelines on the care of patients with an abdominal aortic aneurysm. J. Vasc. Surg. 67(1):2–77, 2018.
Chaikof, E. L., D. C. Brewster, R. L. Dalman, M. S. Makaroun, K. A. Illig, G. A. Sicard, C. H. Timaran, G. R. Upchurch, and F. J. Veith. The care of patients with an abdominal aortic aneurysm: the Society for Vascular Surgery practice guidelines. J. Vasc. Surg. 50:49, 2009.
Darling, R. C., C. R. Messina, D. C. Brewster, and L. W. Ottinger. Autopsy study of unoperated abdominal aortic aneurysms. The case for early resection. Circulation 56:161–4, 1977.
DeLong, E. R., D. M. DeLong, and D. L. Clarke-Pearson. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837–45, 1988.
Detmer, F. J., Chung, B. J., Jimenez, C., Hamzei-Sichani, F., Kallmes, D., Putman, C., and Cebral, J. R.. Associations of hemodynamics, morphology, and patient characteristics with aneurysm rupture stratified by aneurysm location. Neuroradiology 61(3), 275-284, 2019.
Detmer, F. J., Fajardo-Jimnez, D., Mut, F., Juchler, N., Hirsch, S., Pereira, V. M., and Cebral, J. R.. External validation of cerebral aneurysm rupture probability model with data from two patient cohorts. Acta Neurochir. 160(12), 2425–2434., 2018.
Di Achille, P., G. Tellides, C. A. Figueroa, and J. D. Humphrey. A haemodynamic predictor of intraluminal thrombus formation in abdominal aortic aneurysms. Proc. R. Soc. A 470:20140163–20140163, 2014.
Duellman, T., C. L. Warren, J. Matsumura, and J. Yang. Analysis of multiple genetic polymorphisms in aggressive-growing and slow-growing abdominal aortic aneurysms. J. Vasc. Surg. 60:613–621, 2014.
Efron, B. and R. Tibshirani. Bootstrap methods for standard errors, confidence intervals, and other measures of statistical accuracy. Stat. Sci. 1:54–75, 1986.
Esmaily Moghadam, M., Y. Bazilevs, T. Y. Hsia, I. E. Vignon-Clementel, and A. L. Marsden. A comparison of outlet boundary treatments for prevention of backflow divergence with relevance to blood flow simulations. Comput. Mech. 48:277–291, 2011.
Finol, E. A. and C. H. Amon. Blood flow in abdominal aortic aneurysms: pulsatile flow hemodynamics. J. Biomech. Eng. 123:474, 2001.
Grytsan, A., P. N. Watton, and G. A. Holzapfel. A thick-walled fluid solid-growth model of abdominal aortic aneurysm evolution: application to a patient-specific geometry. J. Biomech. Eng. 137:031008, 2015.
Haller, S. J., J. D. Crawford, K. M. Courchaine, C. J. Bohannan, G. J. Landry, G. L. Moneta, A. F. Azarbal, and S. Rugonyi. Intraluminal thrombus is associated with early rupture of abdominal aortic aneurysm. J. Vasc. Surg. 67:1051–1058, 2018.
Hansen, K. B., A. Arzani, and S. C. Shadden. Mechanical platelet activation potential in abdominal aortic aneurysms. J. Biomech. Eng., 137(4), 041005, 2015.
Jasak, H. Error Analysis and Estimation for the Finite Volume Method with Applications to Fluid Flows. Ph.D. thesis, Imperial College, University of London, 1996.
Jasak, H. and H. G. Weller. Application of the finite volume method and unstructured meshes to linear elasticity. J. Numer. Methods Eng. 48:267–287, 2000.
Joly, F., G. Soulez, D. Garcia, S. Lessard, and C. Kauffmann. Flow stagnation volume and abdominal aortic aneurysm growth: Insights from patient-specific computational flow dynamics of Lagrangian-coherent structures. Comput. Biol. Med. 92:98–109, 2018.
Kaibara, M. Rheology of blood coagulation. Biorheology 33:101–117, 1996.
Kauffmann, C., A. Tang, A. Dugas, E. Therasse, V. Oliva, and G. Soulez. Clinical validation of a software for quantitative follow-up of abdominal aortic aneurysm maximal diameter and growth by CT angiography. Eur. J. Radiol. 77:502–508, 2011.
Kazi, M., Thyberg, J., Religa, P., Roy, J., Eriksson, P., Hedin, U., and Swedenborg, J. Influence of intraluminal thrombus on structural and cellular composition of abdominal aortic aneurysm wall . J. Vasc. Surg., 38(6), 1283-1292, 2003.
Les, A. S., S. C. Shadden, C. A. Figueroa, J. M. Park, M. M. Tedesco, R. J. Herfkens and C. A. Taylor. Quantification of hemodynamics in abdominal aortic aneurysms during rest and exercise using magnetic resonance imaging and computational fluid dynamics. Ann. Biomed. Eng., 38(4), 1288-1313, 2010
Lipton, Z. C., D. C. Kale, C. Elkan, and R. Wetzel. Learning to Diagnose with LSTM Recurrent Neural Networks. arXiv preprint arXiv:1511.03677, 2015.
Marcinkowska-Gapińska, A., J. Gapinski, W. Elikowski, F. Jaroszyk, and L. Kubisz. Comparison of three rheological models of shear flow behavior studied on blood samples from post-infarction patients. Med. Biol. Eng. Comput. 45:837–844, 2007.
Martufi, G., M. Lindquist Liljeqvist, N. Sakalihasan, G. Panuccio, R. Hultgren, J. Roy, and T. C. Gasser. Local Diameter, Wall Stress, and Thrombus Thickness Influence the Local Growth of Abdominal Aortic Aneurysms. J. Endovasc. Therapy 23, 2016.
Mills, C. J., I. T. Gabe, J. H. Gault, D. T. Mason, J. Ross, E. Braunwald, and J. P. Shillingford. Pressure-flow relationships and vascular impedance in man. Cardiovasc. Res. 4:405–417, 1970.
Moore, J., S. Maier, D. Ku, and P. Boesiger. Hemodynamics in the abdominal aorta: a comparison of in vitro and in vivo measurements. J. Appl. Physiol. 76:1520–1527, 1994.
ORourke, M. J., J. P., McCullough, and S. Kelly. An investigation of the relationship between hemodynamics and thrombus deposition within patient-specific models of abdominal aortic aneurysm. Proc. Inst. Mech. Eng. H 226(7), 548-564, 2012.
Ouriel, K., R. M. Green, C. Donayre, C. K. Shortell, J. Elliott, and J. A. DeWeese. An evaluation of new methods of expressing aortic aneurysm size: relationship to rupture. J. Vasc. Surg. 15:12–20, 1992.
Parkinson, F., S. Ferguson, P. Lewis, I. M. Williams, C. P. Twine, and South East Wales Vascular Network. Rupture rates of untreated large abdominal aortic aneurysms in patients unfit for elective repair. J. Vasc. Surg. 61:1606–1612, 2015.
Raghavan, M. L., J. Kratzberg, E. M. Castro de Tolosa, M. M. Hanaoka, P. Walker, and E. S. da Silva. Regional distribution of wall thickness and failure properties of human abdominal aortic aneurysm. J. Biomech. 39:3010–3016, 2006.
Reeps, C., A. Maier, J. Pelisek, F. Härtl, V. Grabher-Meier, W. A. Wall, M. Essler, H. H. Eckstein, and M. W. Gee. Measuring and modeling patient-specific distributions of material properties in abdominal aortic aneurysm wall. Biomech. Model. Mechanobiol. 12:717–733, 2013.
Roache, P. J. Roache perspective: a method for uniform reporting of grid refinement studies. J. Fluids Eng. 116(3):405–413, 1994
Rowland, E. M., Y. Mohamied, K. Yean Chooi, E. L. Bailey, and P. D. Weinberg. Comparison of statistical methods for assessing spatial correlations between maps of different arterial properties. J. Biomech. Eng. 137:101003, 2015.
Shum, J., G. Martufi, E. Di Martino, C. B. Washington, J. Grisafi, S. C. Muluk, and E. A. Finol. Quantitative assessment of abdominal aortic aneurysm geometry. Ann. Biomed. Eng. 39:277–286, 2011.
Shum, J., A. Xu, I. Chatnuntawech, and E. A. Finol. A framework for the automatic generation of surface topologies for abdominal aortic aneurysm models. Ann. Biomed. Eng. 39:249–259, 2011.
Silverberg, E., C. C. Boring, and T. S. Squires. Cancer statistics, 1990. Cancer J. Clin. 40:9–26, 1990.
Solberg, S., Singh, K., Wilsgaard, T., and Jacobsen, B. K. Increased growth rate of abdominal aortic aneurysms in women. The Troms study. Eur. J. Vasc. Endovasc. Surg. 29(2):145–149, 2005.
Stenbaek, J., B. Kalin, and J. Swedenborg. Growth of thrombus may be a better predictor of rupture than diameter in patients with abdominal aortic aneurysms. Eur. J. Vasc. Endovasc. Surg. 20:466–469, 2000.
Stevens, R. R., A. Grytsan, J. Biasetti, J. Roy, M. L. Liljeqvist, and T. Christian Gasser. Biomechanical changes during abdominal aortic aneurysm growth. PLoS ONE 12:e0187421, 2017.
Strub, F., H. De Vries, J. Mary, B. Piot, A. Courvile, and O. Pietquin. End-to-end optimization of goal-driven and visually grounded dialogue systems. IJCAI Int. Joint Conf. Artif. Intell. 13:2765–2771, 2017.
Tanios, F., M. W. Gee, J. Pelisek, S. Kehl, J. Biehler, V. Grabher-Meier, W. A. Wall, H. H. Eckstein, and C. Reeps. Interaction of biomechanics with extracellular matrix components in abdominal aortic aneurysm wall. Eur. J. Vasc. Endovasc. Surg. 50:167–174, 2015.
The Theano Development Team, R. Al-Rfou, G. Alain, A. Almahairi, C. Angermueller, D. Bahdanau, N. Ballas, F. Bastien, J. Bayer, A. Belikov, A. Belopolsky, Y. Bengio, A. Bergeron, J. Bergstra, V. Bisson, J. B. Snyder, N. Bouchard, N. Boulanger-Lewandowski, X. Bouthillier, A. de Brébisson, O. Breuleux, P.-L. Carrier, K. Cho, J. Chorowski, P. Christiano, T. Cooijmans, M.-A. Côté, M. Côté, A. Courville, Y. N. Dauphin, O. Delalleau, J. Demouth, G. Desjardins, S. Dieleman, L. Dinh, M. Ducoffe, V. Dumoulin, S. E. Kahou, D. Erhan, Z. Fan, O. Firat, M. Germain, X. Glorot, I. Goodfellow, M. Graham, C. Gulcehre, P. Hamel, I. Harlouchet, J.-P. Heng, B. Hidasi, S. Honari, A. Jain, S. Jean, K. Jia, M. Korobov, V. Kulkarni, A. Lamb, P. Lamblin, E. Larsen, C. Laurent, S. Lee, S. Lefrancois, S. Lemieux, N. Léonard, Z. Lin, J. A. Livezey, C. Lorenz, J. Lowin, Q. Ma, P.-A. Manzagol, O. Mastropietro, R. T. McGibbon, R. Memisevic, B. van Merriënboer, V. Michalski, M. Mirza, A. Orlandi, C. Pal, R. Pascanu, M. Pezeshki, C. Raffel, D. Renshaw, M. Rocklin, A. Romero, M. Roth, P. Sadowski, J. Salvatier, F. Savard, J. Schlüter, J. Schulman, G. Schwartz, I. V. Serban, D. Serdyuk, S. Shabanian, C. Simon, S. Spieckermann, S. R. Subramanyam, J. Sygnowski, J. Tanguay, G. van Tulder, J. Turian, S. Urban, P. Vincent, F. Visin, H. de Vries, D. Warde-Farley, D. J. Webb, M. Willson, K. Xu, L. Xue, L. Yao, S. Zhang, and Y. Zhang. Theano: a Python framework for fast computation of mathematical expressions. arXiv e-prints, p. 19, 2016.
Tzirakis, K., Y. Kamarianakis, E. Metaxa, N. Kontopodis, C. V. Ioannou, and Y. Papaharilaou. A robust approach for exploring hemodynamics and thrombus growth associations in abdominal aortic aneurysms. Biol. Eng. Comput. 55:1493–1506, 2017.
Vergara, C., D. Le Van, M. Quadrio, L. Formaggia, and M. Domanin. Large eddy simulations of blood dynamics in abdominal aortic aneurysms. Med. Eng. Phys. 47:38-46, 2017.
Womersley, J. R. Method for the calculation of velocity, rate of flow and viscous drag in arteries when the pressure gradient is known. J. Physiol. 127:553–563, 1955.
Wu, J. and S. C. Shadden. Coupled simulation of hemodynamics and vascular growth and remodeling in a subject-specific geometry. Ann. Biomed. Eng. 43(7), 1543-1554.
Xu, X., B. Wang, C. Ren, J. Hu, D. A. Greenberg, T. Chen, L. Xie, and K. Jin. Age-related impairment of vascular structure and functions. Aging Dis. 8:590–610, 2017.
Yushkevich, P. A., J. Piven, H. C. Hazlett, R. G. Smith, S. Ho, J. C. Gee, and G. Gerig. User-guided 3D active contour segmentation of anatomical structures: significantly improved efficiency and reliability. NeuroImage 31:1116–1128, 2006.
Zambrano, B. A., H. Gharahi, C. Y. Lim, F. A. Jaberi, J. Choi, W. Lee, and S. Baek. Association of intraluminal thrombus, hemodynamic forces, and abdominal aortic aneurysm expansion using longitudinal CT images. Ann. Biomed. Eng. 44:1502–1514, 2016.
Acknowledgements
The authors thank Daniel Stubbs and Compute Canada for the HPC support. The presented work relies on open-source projects and their community, especially OpenFOAM and ITKSnap (http://www.itksnap.org). This work has been supported by the Collaborative Research and Development Grants No. 460903-13 provided by the Natural Sciences and Engineering Research Council of Canada (NSERC) and the Industry-partnered Collaborative Research Grant No. 124294 from the Canadian Institutes of Health Research (CIHR).
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Umberto Morbiducci oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Joly, F., Soulez, G., Lessard, S. et al. A Cohort Longitudinal Study Identifies Morphology and Hemodynamics Predictors of Abdominal Aortic Aneurysm Growth. Ann Biomed Eng 48, 606–623 (2020). https://doi.org/10.1007/s10439-019-02375-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-019-02375-1