Abstract
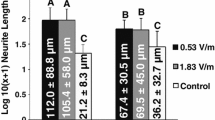
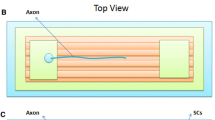
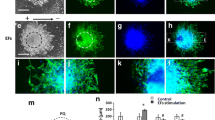
Schwann cells (SCs) are PNS glia that play numerous support functions including myelination of axons. After PNS injury, SCs facilitate regeneration by phagocytosing cellular debris and providing physical and biochemical cues to guide axon growth. This reparative phenotype suggests SCs could be critical cellular targets for enhancing nerve regeneration. One method for altering cell morphology and motility is the application of direct current (DC) electric fields (EFs). Endogenous EFs have physiologic relevance during embryogenesis and serve as guidance and polarization cues. While much literature exists on EFs and CNS and PNS neurons, the effects of EFs on SCs have not been extensively studied. In this work, cell alignment, migration, and morphology of rat SCs were measured in response to several EF stimulation regimes including constant DC, 50% duty cycle DC and oscillating DC. SCs were found to re-orient perpendicular to field lines and respond to DC EFs as low as 75 mV/mm. EF exposure promoted directed migration, with travel towards the cathode at a mean rate of 7.5 µm/h. The data highlight the utility of EFs in modulating SC morphology, alignment and migration. Results may have implications for using EFs to attract and realign SCs at the site of PNS trauma.








Similar content being viewed by others
References
Alexander, J. K., B. Fuss, and R. J. Colello. Electric field-induced astrocyte alignment directs neurite outgrowth. Neuron Glia Biol. 2:93–103, 2006.
Arthur-Farraj, P. J., M. Latouche, D. K. Wilton, S. Quintes, E. Chabrol, A. Banerjee, A. Woodhoo, B. Jenkins, M. Rahman, M. Turmaine, G. K. Wicher, R. Mitter, L. Greensmith, A. Behrens, G. Raivich, R. Mirsky, and K. R. Jessen. c-Jun reprograms Schwann cells of injured nerves to generate a repair cell essential for regeneration. Neuron 75:633–647, 2012.
Bonazzi, D., and N. Minc. Dissecting the molecular mechanisms of electrotactic effects. Adv. Wound Care 3:139–148, 2014.
Borgens, R. B. Electrically mediated regeneration and guidance of adult mammalian spinal axons into polymeric channels. Neuroscience 91:251–264, 1999.
Borgens, R. B., K. R. Robinson, J. W. Vanable, and M. E. McGinnis. Artificially controlling axonal regeneration and development by applied electric fields. In: Electric Fields in Vertebrate Repair. New York: Alan R. Liss, 1989, pp. 164–165.
Borgens, R. B., R. Shi, T. J. Mohr, and C. B. Jaeger. Mammalian cortical astrocytes align themselves in a physiological voltage gradient. Exp. Neurol. 128:41–49, 1994.
Borgens, R., J. Toombs, A. Blight, M. Mcginnis, M. Bauer, W. Widmer, and J. Cook. Effects of applied electric-fields on clinical cases of complete paraplegia in dogs. Restor. Neurol. Neurosci. 5:305–322, 1993.
Borgens, R. B., J. P. Toombs, G. Breur, W. R. Widmer, D. Waters, A. M. Harbath, P. March, and L. G. Adams. An imposed oscillating electrical field improves the recovery of function in neurologically complete paraplegic dogs. J. Neurotrauma 16:639–657, 1999.
Brown, M. J., and L. M. Loew. Electric field directed fibroblast locomotion involves cell surface molecular reorganization and is calcium independent. J. Cell Biol. 127:117–128, 1994.
Bunge, R. P. The role of the Schwann cell in trophic support and regeneration. J. Neurol. 242:S19–S21, 1994.
Bunge, R. P., and M. B. Bunge. Interrelationship between Schwann cell function and extracellular matrix production. Trends Neurosci. 6:499–505, 1983.
Chang, H. F., Y. S. Lee, T. K. Tan, and J. Y. Cheng. Pulsed DC electric field-induced differentiation of cortical neural precursor cells. PLoS ONE 11:e0158133, 2016.
Cooper, M. S., and R. E. Keller. Perpendicular orientation and directional migration of amphibian neural crest cells in dc electrical fields. Proc. Natl Acad. Sci. U.S.A. 81:160–164, 1984.
Hall, S. M. Regeneration in cellular and acellular autografts in the peripheral nervous system. Neuropathol. Appl. Neurobiol. 12:27–46, 1986.
Hall, S. M. The effect of inhibiting Schwann cell mitosis on the re-innervation of acellular autografts in the peripheral nervous system of the mouse. Neuropathol. Appl. Neurobiol. 12:401–414, 1986.
Huang, Y. J., J. Samorajski, R. Kreimer, and P. C. Searson. The influence of electric field and confinement on cell motility. PLoS ONE 8:e59447, 2013.
Jaffe, L. F. The role of ionic currents in establishing developmental pattern. Philos. Trans. R. Soc. Lond. B Biol. Sci. 295:553–566, 1981.
Jessen, K. R., and R. Mirsky. The repair Schwann cell and its function in regenerating nerves. J. Physiol. 594:3521–3531, 2016.
Koppes, A. N., A. L. Nordberg, G. Paolillo, N. Goodsell, H. Darwish, L. Zhang, and D. M. Thompson. Electrical stimulation of Schwann cells promotes sustained increases in neurite outgrowth. Tissue Eng. Part A 20:494–506, 2013.
Koppes, A. N., A. M. Seggio, and D. M. Thompson. Neurite outgrowth is significantly increased by the simultaneous presentation of Schwann cells and moderate exogenous electric fields. J. Neural Eng. 8:1–13, 2011.
Li, R., Z. Liu, Y. Pan, L. Chen, Z. Zhang, and L. Lu. Peripheral nerve injuries treatment: a systematic review. Cell Biochem. Biophys. 68:449–454, 2014.
Mackinnon, S. E., and A. L. Dellon. Nerve repair and nerve grafts. In: Surgery of the Peripheral Nerve, edited by S. E. Mackinnon. New York: Thieme, 1988.
Martin, J. R., and H. D. Webster. Mitotic Schwann cells in developing nerve: their changes in shape, fine structure, and axon relationships. Dev. Biol. 32:417–431, 1973.
McCaig, C. D. Spinal neurite reabsorption and regrowth in vitro depend on the polarity of an applied electric field. Development 100:31–41, 1987.
McLaughlin, S., and M. M. Poo. The role of electro-osmosis in the electric-field-induced movement of charged macromolecules on the surfaces of cells. Biophys. J. 34:85–93, 1981.
Nuccitelli, R., and C. A. Erickson. Embryonic cell motility can be guided by physiological electric fields. Exp. Cell Res. 147:195–201, 1983.
Orida, N., and M. Poo. Electrophorectic movement and localisation of acetylcholine receptors in the embryonic muscle cell membrane. Nature 275:31–35, 1978.
Ozkucur, N., S. Perike, P. Sharma, and R. Funk. Persistent directional cell migration requires ion transport proteins as direction sensors and membrane potential differences in order to maintain directedness. BMC Cell Biol. 12:1–13, 2011.
Pan, L., and R. B. Borgens. Perpendicular organization of sympathetic neurons within a required physiological voltage. Exp. Neurol. 222:161–164, 2010.
Pan, L., and R. B. Borgens. Strict perpendicular orientation of neural crest-derived neurons in vitro is dependent on an extracellular gradient of voltage. J. Neurosci. Res. 90:1335–1346, 2012.
Poo, M., J. W. Lam, N. Orida, and A. W. Chao. Electrophoresis and diffusion in the plane of the cell membrane. Biophys. J. 26:1–22, 1979.
Rajnicek, A. M., N. A. R. Gow, and C. D. McCaig. Electric field-induced orientation of rat hippocampal neurons in vitro. Exp. Physiol. 77:229–232, 1992.
Rajnicek, A. M., K. R. Robinson, and C. D. McCaig. The direction of neurite growth in a weak DC electric field depends on the substratum: contributions of adhesivity and net surface charge. Dev. Biol. 203:412–423, 1998.
Seggio, A. M., A. Narayanaswamy, B. Roysam, and D. M. Thompson. Self-aligned Schwann cell monolayers demonstrate an inherent ability to direct neurite outgrowth. J. Neural Eng. 7(4):046001, 2010.
Shapiro, S., R. Borgens, R. Pascuzzi, K. Roos, M. Groff, S. Purvines, R. B. Rodgers, S. Hagy, and P. Nelson. Oscillating field stimulation for complete spinal cord injury in humans: a phase 1 trial. J. Neurosurg. Spine 2:3–10, 2005.
Shi, R., and R. B. Borgens. Three-dimensional gradients of voltage during development of the nervous system as invisible coordinates for the establishment of embryonic pattern. Dev. Dyn. 202:101–114, 1995.
Siemionow, M., and G. Brzezicki. Current techniques and concepts in peripheral nerve repair. Int. Rev. Neurobiol. 87:141–172, 2009.
Son, Y. J., and W. J. Thompson. Schwann cell processes guide regeneration of peripheral axons. Neuron 14:125–132, 1995.
Stump, R. F., and K. R. Robinson. Xenopus neural crest cell migration in an applied electrical field. J. Cell Biol. 97:1226–1233, 1983.
Talat, K., S. Sayers, and N. Chauhan. Effect of applied electric field on astrocytic scar formation after spinal cord injury. In: Electricity and Magnetism in Biology and Medicine, edited by F. Bersani. Boston: Springer, 1999, pp. 887–890.
Yao, L., Y. Li, J. Knapp, and P. Smith. Exploration of molecular pathways mediating electric field-directed Schwann cell migration by RNA-seq. J. Cell. Physiol. 230:1515–1524, 2015.
Yao, L., and L. Yongchao. The role of direct current electric field-guided stem cell migration in neural regeneration. Stem Cell Rev. 12:365–375, 2016.
Zhao, M., J. V. Forrester, and C. D. McCaig. A small physiological electric field orients cell division. Proc. Natl. Acad. Sci. U.S.A. 96:4942–4946, 1999.
Acknowledgments
This study was completed at the Center for Paralysis Research with funding provided by the State of Indiana. The authors thank Megan Saenger and Bhavani Gopalakrishnan for their assistance with cell culture.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Joel Stitzel oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bunn, S.J., Lai, A. & Li, J. DC Electric Fields Induce Perpendicular Alignment and Enhanced Migration in Schwann Cell Cultures. Ann Biomed Eng 47, 1584–1595 (2019). https://doi.org/10.1007/s10439-019-02259-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-019-02259-4