Abstract
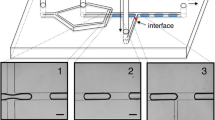
This work presents an alternative approach for visualizing and monitoring in real-time emulsification process using an optical microscope and a stroboscopic light controlled by a homemade circuit implemented in an Arduino microcontroller. The microfluidic device was built from glass tapered capillaries coaxially aligned in a co-flow configuration. The implemented optical visualization allows a clear in-situ identification of three oil-in-water emulsification regimes. The drop frequency production is determined in each case by matching the stroboscopic light frequency as integer submultiples of the frequency of the train of drops. The droplet production quality of each emulsification regime is associated with the polydispersity index of the collected emulsions. The frequency of each regime and their flow rates are related to a mathematical model that predicts the behavior of the frequencies upon the capillary number showing the applicability of the stroboscopic system to obtain qualitative information. The in situ monitoring of dripping regime varying the flow conditions enable the production and collection of emulsions with controlled drop size, which also are validated with a mathematical model.








Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Arriaga LR, Datta SS, Kim SH et al (2014) Ultrathin shell double emulsion templated giant unilamellar lipid vesicles with controlled microdomain formation. Small (Weinheim an der Bergstrasse, Germany) 10(5):950–6. https://doi.org/10.1002/smll.201301904
Arriaga LR, Huang Y, Kim SH et al (2019) Single-step assembly of asymmetric vesicles. Lab Chip 19(5):749–756. https://doi.org/10.1039/c8lc00882e
Bailey L, Vavolil PJ, Vishwapathi V et al (2021) Electroformation of particulate emulsions using lamellar and nonlamellar lipid self-assemblies. Langmuir 37(49):14,527-14,539. https://doi.org/10.1021/acs.langmuir.1c02721
Baroud CN (2010) Dynamics of microfluidic droplets. Lab Chip 10:2032–2045. https://doi.org/10.1039/c001191f
Chu LY, Utada AS, Shah RK et al (2007) Controllable monodisperse multiple emulsions. Angew Chem - Int Ed 46(47):8970–8974. https://doi.org/10.1002/anie.200701358
Clayton KN, Salameh JW, Wereley ST et al (2016) Physical characterization of nanoparticle size and surface modification using particle scattering diffusometry. Biomicrofluidics 10(5):1–14. https://doi.org/10.1063/1.4962992
Fan Y (2018) Low-cost microfluidics: materials and methods. Micro Nano Lett 13(10):1367–1372. https://doi.org/10.1049/mnl.2018.5169
Gao Z, Peng H, Zhu M et al (2019) A facile strategy for visualizing and modulating droplet-based microfluidics. Micromachines 10(5):1–11. https://doi.org/10.3390/mi10050291
Garstecki P, Fuerstman MJ, Stone HA et al (2006) Formation of droplets and bubbles in a microfluidic T-junction-scaling and mechanism of break-up. Lab Chip 6(3):437. https://doi.org/10.1039/b510841a
Ge X, Rubinstein BY, He Y et al (2021) Double emulsions with ultrathin shell by microfluidic step-emulsification. Lab Chip 21(8):1613–1622. https://doi.org/10.1039/d0lc01044h
Grundy MML, Wilde PJ (2021) Bioaccessibility and digestibility of lipids from food. Springer, Cham. https://doi.org/10.1007/978-3-030-56909-9
Guerrero J, Chang YW, Fragkopoulos AA et al (2020) Capillary-based microfluidics-coflow, flow-focusing, electro-coflow, drops, jets, and instabilities. Small 16(9):1–15. https://doi.org/10.1002/smll.201904344
Hussain M, Liu X, Tang S et al (2022) Rapid detection of Pseudomonas aeruginosa based on lab-on-a-chip platform using immunomagnetic separation, light scattering, and machine learning. Anal Chim Acta 1189(339):223. https://doi.org/10.1016/j.aca.2021.339223
Kim SH, Kim JW, Cho JC et al (2011) Double-emulsion drops with ultra-thin shells for capsule templates. Lab Chip 11(18):3162–6. https://doi.org/10.1039/c1lc20434c
Kovalchuk NM, Simmons MJ (2023) Review of the role of surfactant dynamics in drop microfluidics. Adv Colloid Interface Sci 312(January):102,844. https://doi.org/10.1016/j.cis.2023.102844
Lee H, Xu L, Koh D et al (2014) Various on-chip sensors with microfluidics for biological applications. Sensors (Switzerland) 14(9):17,008-17,036. https://doi.org/10.3390/s140917008
Martino C, Lee TY, Kim SH et al (2015) Microfluidic generation of PEG-b-PLA polymersomes containing alginate-based core hydrogel. Biomicrofluidics 10(1063/1):4914112
Mehrnezhad A, Kwak TJ, Kim S et al (2019) Moving shot, an affordable and high-throughput setup for direct imaging of fast-moving microdroplets. Microsyst Technol 25(9):3417–3423. https://doi.org/10.1007/s00542-018-4272-9
Ravera F, Dziza K, Santini E et al (2021) Emulsification and emulsion stability: the role of the interfacial properties. Adv Colloid Interface Sci 288(102):344. https://doi.org/10.1016/j.cis.2020.102344
rgco (2019) Portable precision stroboscope. https://www.instructables.com/Portable-Precision-Stroboscope/
Schramm LL (2006) Emulsions, foams, and suspensions: fundamentals and applications. Wiley. https://doi.org/10.1002/3527606750
Schroën K, Wu L, Corstens M (2022) Food-grade microgel capsules tailored for anti-obesity strategies through microfluidic preparation. Curr Opin Food Sci 45:1–7. https://doi.org/10.1016/j.cofs.2022.100816
Seo H, Lee H (2021) Recent developments in microfluidic synthesis of artificial cell-like polymersomes and liposomes for functional bioreactors. Biomicrofluidics 15(2):1–13. https://doi.org/10.1063/5.0048441
Shepherd SJ, Issadore D, Mitchell MJ (2021) Microfluidic formulation of nanoparticles for biomedical applications. Biomaterials 274(March):120,826. https://doi.org/10.1016/j.biomaterials.2021.120826
Song H, Tice JD, Ismagilov RF (2003) Zuschriften. Angew Chem 115:791–796
Sun Z, Yan X, Xiao Y et al (2022) Pickering emulsions stabilized by colloidal surfactants: role of solid particles. Particuology 64:153–163. https://doi.org/10.1016/j.partic.2021.06.004
Suzuki H, Lee IYS, Okuno Y (2010) Stability and dancing dynamics of acoustic single bubbles in aqueous surfactant solution. Int J Phys Sci 5(3):176–181
Tuljak M, Lajevec D, Štanc R et al (2022) Low-cost programable stroboscopic illumination with sub-microsecond pulses for high-throughput microfluidic applications. HardwareX 12:1–11. https://doi.org/10.1016/j.ohx.2022.e00367
Umbanhowar PB, Prasad V, Weitz Da (2000) Monodisperse emulsion generation via drop break off in a coflowing stream. Langmuir 16(2):347–351. https://doi.org/10.1021/la990101e
Utada AS, Lorenceau E, Link DR, Kaplan PD, Stone HA, Weitz DA (2005) Monodisperse double emulsions generated from a microcapillary device. Science (New York, NY) 308:537–542
Utada A, Fernandez-Nieves A, Stone H et al (2007) Dripping to jetting transitions in coflowing liquid streams. Phys Rev Lett 99(9):094,502. https://doi.org/10.1103/PhysRevLett.99.094502
Van Ven F (1977) Handbook of stroboscopy. GenRad, Concord, Massachusetts, USA
Vladisavljević GT, Al Nuumani R, Nabavi SA (2017) Microfluidic production of multiple emulsions. Micromachines. https://doi.org/10.3390/mi8030075
Wang ZL (2022) Universal self-scalings in a micro-co-flowing. Chem Eng Sci 262(117):956. https://doi.org/10.1016/j.ces.2022.117956
Wang T, Andersen SI, Shapiro A (2020) Coalescence of oil droplets in microchannels under brine flow. Colloids Surf A Physicochem Eng Asp 598(April):124,864. https://doi.org/10.1016/j.colsurfa.2020.124864
Wang Y, Shang L, Zhao Y et al (2022) Microfluidic generation of multicomponent soft biomaterials. Engineering 13:128–143. https://doi.org/10.1016/j.eng.2021.02.026
Winkler S, Grünberger A, Bahnemann J (2021) Microfluidics in biotechnology: Quo Vadis. Adv Biochem Eng/Biotechnol 179(Book series):355–380. https://doi.org/10.1007/10_2020_162
Wu L, Liu X, Zhao Y et al (2017) Role of local geometry on droplet formation in axisymmetric microfluidics. Chem Eng Sci 163:56–67. https://doi.org/10.1016/j.ces.2017.01.022
Zhang W, Qu L, Pei H et al (2019) Controllable fabrication of inhomogeneous microcapsules for triggered release by osmotic pressure. Small. https://doi.org/10.1002/smll.201903087
Zhu P, Wang L (2017) Passive and active droplet generation with microfluidics: a review. Lab Chip 17(1):34–75. https://doi.org/10.1039/C6LC01018K
Acknowledgements
This work was partially funded by Consejo de la Investigación Científica, Universidad Michoacana de San Nicolás de Hidalgo (UMSNH), Research Project 2023. Luis M. Montes-de-Oca acknowledges support from the Mexican Council on Humanities, Science and Technology (CONAHCYT) for a postdoctoral fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Montes-de-Oca, L.M., Espinosa, G. & Martínez-Torres, P. Identification of emulsification regimes in co-flow microfluidics using stroboscopic LED illumination. Microfluid Nanofluid 27, 61 (2023). https://doi.org/10.1007/s10404-023-02671-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-023-02671-2