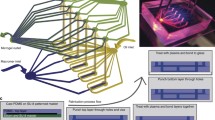
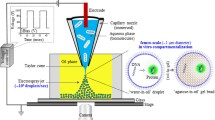
Abstract
Fabrication of multi-chamber microcapsules is of great interest in biotechnology, 3D cell culture, microfluidics, and other applications. The centrifuge-based droplet shooting device (CDSD) is a valuable method for multi-chamber microcapsule production, but is limited by loading difficulty and low volumes. We present a new CDSD system that is easy to load for multi-chamber high- or low-volume applications. We describe the structure, characteristics, and principle demonstration of the device. We experimentally determine trends of droplet shape differences and the resulting conditions of glob formation (due to collision with the centrifuge wall) with > 97% accuracy which compares well with published studies. The deformation parameter was shown for the first time relative to centrifugal force at the tip, as well as the angle at droplet splashdown, tip gap, and other features. We showed the formation of dimpled droplets, prolate spheroid, and other shaped microcapsules. A variety of contents were individually addressed into the multi-chamber droplets (cells, fluorescent, and magnetic nanoparticles). Encapsulated cancer (KLE) cells were cultured within alginate hydrogel droplets with > 60% viability for 4 days. Cell release post-encapsulation was demonstrated. Six-compartment droplets were produced from low (2 µL/reservoir) and high (~ 100 µL/reservoir) volumes. We report the highest droplet volume production of any multi-chamber CDSD for small, uniform droplets (7 × 105 droplets; 0.431 g; 106 μm diameter). This newly developed CDSD enables improved placement of nanoparticles or cells within multi-chamber microscale droplets for 3D cell culture and other applications.
Graphical abstract


















Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Alessandri K et al (2013) Cellular capsules as a tool for multicellular spheroid production and for investigating the mechanics of tumor progression in vitro. Proc Natl Acad Sci 110(37):14843–14848
Azimi-Boulali J et al (2020) Droplet and particle generation on centrifugal microfluidic platforms: a review. Micromachines (Basel) 11(6):603
Badalan M et al (2022) Physical analysis of the centrifugal microencapsulation process. Ind Eng Chem Res 61(30):10891–10914
Badalan M et al (2023) A soft landing approach for the centrifugal microgel synthesis process. Comptes Rendus Mécanique 351:83–102
Barnes JM et al (2012) Resistance to fluid shear stress is a conserved biophysical property of malignant cells. PLoS One 7(12):e50973
Bidarra SJ et al (2011) Injectable in situ crosslinkable RGD-modified alginate matrix for endothelial cells delivery. Biomaterials 32(31):7897–7904
Cao N et al (2012) Influence of calcium ions on cell survival and proliferation in the context of an alginate hydrogel. Int Schol Res Not 2012:1–9
Chen Z et al (2016) Spinning micropipette liquid emulsion generator for single cell whole genome amplification. Lab Chip 16(23):4512–4516
Chen Z et al (2017) Centrifugal micro-channel array droplet generation for highly parallel digital PCR. Lab Chip 17(2):235–240
Choi CH et al (2010) Surface-tension-induced synthesis of complex particles using confined polymeric fluids. Angew Chem 122(42):7914–7918
Daly AC et al (2020) Hydrogel microparticles for biomedical applications. Nat Rev Mater 5(1):20–43
Eral HB et al (2016) Governing principles of alginate microparticle synthesis with centrifugal forces. Langmuir 32(28):7198–7209
Goncu B, Yucesan E (2020) Microencapsulation for clinical applications and transplantation by using different alginates. In: Nano-and microencapsulation-techniques and applications. IntechOpen
Hayakawa M et al (2016) Complex-shaped three-dimensional multi-compartmental microparticles generated by diffusional and Marangoni microflows in centrifugally discharged droplets. Sci Rep 6(1):1–9
Kang S-M et al (2019) Centrifugal force-driven modular micronozzle system: generation of engineered alginate microspheres. Sci Rep 9(1):1–10
Kim JH et al (2014) Droplet microfluidics for producing functional microparticles. Langmuir 30(6):1473–1488
Lee J, Kim J (2012) Multiphasic sensory alginate particle having polydiacetylene liposome for selective and more sensitive multitargeting detection. Chem Mater 24(14):2817–2822
Lee P, Rogers MA (2012) Effect of calcium source and exposure-time on basic caviar spherification using sodium alginate. Int J Gastron Food Sci 1(2):96–100
Li G et al (2017) Mechanical strengthened alginate/polyacrylamide hydrogel crosslinked by barium and ferric dual ions. J Mater Scie 52:8538–8545
Liu K et al (2012) Generation of disk-like hydrogel beads for cell encapsulation and manipulation using a droplet-based microfluidic device. Microfluid Nanofluid 13:761–767
Liu M et al (2016) On-chip preparation of calcium alginate particles based on droplet templates formed by using a centrifugal microfluidic technique. J Colloid Interface Sci 466:20–27
Madadelahi M et al (2020) Fluidic barriers in droplet-based centrifugal microfluidics: Generation of multiple emulsions and microspheres. Sens Actuators, B Chem 311:127833
Maeda K et al (2012) Controlled synthesis of 3D multi-compartmental particles with centrifuge-based microdroplet formation from a multi-barrelled capillary. Adv Mater 24(10):1340–1346
Miraftab M et al (2010) Inherently antimicrobial alchite fibres developed for wound care applications. In: Anand SC, Kennedy JF, Miraftab M, Rajendran S (eds) Medical and healthcare textiles. Woodhead Publishing, Sawston, pp 76–83
Nair K et al (2009) Characterization of cell viability during bioprinting processes. Biotechnol J 4(8):1168–1177
Nisisako T et al (2006) Synthesis of monodisperse bicolored Janus particles with electrical anisotropy using a microfluidic co-flow system. Adv Mater 18(9):1152–1156
Park YG et al (1998) Microencapsulation of islets and model beads with a thin alginate–ba2+ gel layer using centrifugation. Polym Adv Technol 9(10–11):734–739
Peng H-Y et al (2020) Mesoscale regulation of droplet templates to tailor microparticle structures and functions. Particuology 48:74–87
Qu Q et al (2022) Spatial confinement of multi-enzyme for cascade catalysis in cell-inspired all-aqueous multicompartmental microcapsules. J Colloid Interface Sci 626:768–774
Rahmani S et al (2016) Dual release carriers for cochlear delivery. Adv Healthc Mater 5(1):94–100
Sharpe S et al (2003) Mucosal immunization with PLGA-microencapsulated DNA primes a SIV-specific CTL response revealed by boosting with cognate recombinant modified vaccinia virus Ankara. Virology 313(1):13–21
Sugiura S et al (2005) Size control of calcium alginate beads containing living cells using micro-nozzle array. Biomaterials 26(16):3327–3331
Sun XT et al (2017) Multitarget sensing of glucose and cholesterol based on Janus hydrogel microparticles. Biosens Bioelectron 92:81–86
Tang G et al (2019) Gas-shearing fabrication of multicompartmental microspheres: a one-step and oil-free approach. Adv Sci 6(9):1802342
Tate T (1864) XXX. On the magnitude of a drop of liquid formed under different circumstances. Lond Edinb Dublin Philos Mag J Sci 27(181):176–180
Wu Y et al (2014) Self-propelled polymer multilayer Janus capsules for effective drug delivery and light-triggered release. ACS Appl Mater Interfaces 6(13):10476–10481
Yin S-N et al (2011) Versatile bifunctional magnetic-fluorescent responsive Janus supraballs towards the flexible bead display. Adv Mater 23(26):2915–2919
Yoshida S et al (2017) Compartmentalized spherical collagen microparticles for anisotropic cell culture microenvironments. Adv Healthc Mater 6(8):1601463
Funding
We gratefully acknowledge the funding for this research which was provided by The Ivan Bowen Family Foundation, and the Liautaud Family Foundation.
Author information
Authors and Affiliations
Contributions
KEP performed experiments, interpreted results, wrote, and edited the manuscript. AEA cultured and provided KLE cells, and edited the manuscript. MW-A secured funding, and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare having no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Petersen, K.E., Asangba, A.E. & Walther-Antonio, M. Multi-chamber microcapsule generation for cell culture using a high- and low-volume centrifuge-based droplet shooting device. Microfluid Nanofluid 27, 40 (2023). https://doi.org/10.1007/s10404-023-02651-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-023-02651-6