Abstract
Purpose
To report the clinical findings in 6 patients who developed night blindness after long-term hemodialysis.
Study design
Retrospective case series.
Patients and methods
The medical charts of the 6 patients were examined. The fundus photographs, spectral-domain optical coherence tomographic (SD-OCT) images, full-field ERGs, and blood chemistry panels were analyzed.
Results
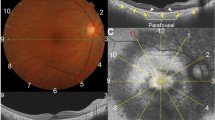
The mean age of the 6 patients (4 men) at the time of diagnosis was 69.1 ± 5.9 years. The mean duration of the hemodialysis was 21.8 ± 13.4 years (7–41 years). The visual acuity of the patients was preserved at 20/30 or better except in 1 eye. Ophthalmoscopy showed white flecks that were scattered over the midperipheral retina in all the eyes. SD-OCT showed mild macular degeneration in 5 eyes. The scotopic ERGs elicited by dim flashes were absent, and those elicited by bright flashes had negative waveforms. The photopic ERGs were relatively well preserved. These data indicated a rod-specific dysfunction that may account for the night blindness. The plasma concentration of vitamin A was within the normal range in 4 of the patients and slightly lower than the normal limit in 1 of the patients. Administration of vitamin A was performed for 1 patient, and the symptom of night blindness and scotopic ERGs were improved 3 months later.
Discussion
Long-term hemodialysis can be associated with the night blindness that may be caused by vitamin A deficiency, even though the plasma concentration of vitamin A in these patients was within the normal range.




Similar content being viewed by others
References
Thirkill CE, FitzGerald P, Sergott RC, Roth AM, Tyler NK, Kaltner JL. Cancer-associated retinophaty (CAR syndrome) with antibodies reacting with retinal, optic-nerve, and cancer cells. N Engl J Med. 1989;321:1589–94.
Milam AH, Saari JC, Jacobson SG, Lubinski WP, Feun LG, Alexander KR. Autoantibodies against retinal bipolar cells in cutaneous melanoma-associated retinopathy. Invest Ophthalmol Vis Sci. 1993;34:91–100.
Ueno S, Inooka D, Nakanishi A, Okado S, Yasuda S, Kominami T, et al. Clinical course of paraneoplastic retinopathy with anti-TRPM1 autoantibody in Japanese cohort. Retina. 2019;39:2410–8.
Tsang SH, Sharma T. Autoimmune retinopathy. Adv Exp Med Biol. 2018;1085:223–6.
Harris EW, Loewenstein JI, Azar D. Vitamin A deficiency and its effects on the eye. Int Ophthalmol Clin. 1998;38:155–61.
Kelsey JH, Arden GB. Acquired unilateral loss of dark adaptation. Br J Ophthalmol. 1971;55:38–43.
Kondo M, Nakamura M, Tanikawa A, Kachi S, Hirai T, Miyake Y. Acquired unilateral night blindness with negative ERG: nine-year follow-up. Retina. 2005;25:519–21.
Tsunoda K, Fujinami K, Yoshitake K, Iwata T. Late-onset night blindness with peripheral flecks accompanied by progressive trickle-like macular degeneration. Doc Ophthalmol. 2019;139:171–84.
Ueno S, Inooka D, Meinert M, Ito Y, Tsunoda K, Fujinami K, et al. Three cases of acute-onset bilateral photophobia. Jpn J Ophthalmol. 2019;63:172–80.
Nishida T, Sawada A, Mochizuki K, Niwa Y, Hayakawa K. Case of acquired night blindness in a hemodialysis patient. Can J Ophthalmol. 2013;48:e148–51.
McCulloch DL, Marmor MF, Brigell MG, Hamilton R, Holder GE, Tzekov R, et al. ISCEV standard for full-field clinical electroretinography (2015 update). Doc Ophthalmol. 2015;130:1–12.
Hayashi T, Goto-Omoto S, Takeuchi T, Gekka T, Ueoka Y, Kitahara K. Compound heterozygous RDH5 mutations in familial fleck retina with night blindness. Acta Ophthalmol. 2006;84:254–8.
Spaide RF, Curcio CA. Drusen characterization with multimodal imaging. Retina. 2010;30:1441–54.
Zaharova E, Sherman J. The use of SD-OCT in the differential diagnosis of dots, spots and other white retinal lesions. Eye Brain. 2011;3:69–80.
Fukukita H, Ito Y, Iwase T, Kaneko H, Yasuda S, Kataoka K, et al. Inner macular changes after vitrectomy with internal limiting membrane peeling for rhegmatogenous retinal detachment: similarity with Alport syndrome. Retina. 2019;39:2332–40.
Hayashi T, Hosono K, Kurata K, Katagiri S, Mizobuchi K, Ueno S, et al. Coexistence of GNAT1 and ABCA4 variants associated with Nougaret-type congenital stationary night blindness and childhood-onset cone-rod dystrophy. Doc Ophthalmol. 2020;140:147–57.
Dryja TP, Hahn LB, Reboul T, Arnaud B. Missense mutation in the gene encoding the alpha subunit of rod transducin in the Nougaret form of congenital stationary night blindness. Nat Genet. 1996;13:358–60.
Dryja TP, Berson EL, Rao VR, Oprian DD. Heterozygous missense mutation in the rhodopsin gene as a cause of congenital stationary night blindness. Nat Genet. 1993;4:280–3.
Gal A, Orth U, Baehr W, Schwinger E, Rosenberg T. Heterozygous missense mutation in the rod cGMP phosphodiesterase beta-subunit gene in autosomal dominant stationary night blindness. Nat Genet. 1994;7:64–8.
Szabo V, Kreienkamp HJ, Rosenberg T, Gal A. pGln200Glu, a putative constitutively active mutant of rod alpha-transducin (GNAT1) in autosomal dominant congenital stationary night blindness. Hum Mutat. 2007;28:741–2.
Sandberg MA, Pawlyk BS, Dan J, Arnaud B, Dryja TP, Berson EL. Rod and cone function in the Nougaret form of stationary night blindness. Arch Ophthalmol. 1998;116:867–72.
Apushkin MA, Fishman GA. Improvement in visual function and fundus findings for a patient with vitamin A-deficient retinopathy. Retina. 2005;25:650–2.
Genead MA, Fishman GA, Lindeman M. Fundus white spots and acquired night blindness due to vitamin A deficiency. Doc Ophthalmol. 2009;119:229–33.
Aleman TS, Garrity ST, Brucker AJ. Retinal structure in vitamin A deficiency as explored with multimodal imaging. Doc Ophthalmol. 2013;127:239–43.
Smith FR, Goodman DS. The effects of diseases of the liver, thyroid, and kidneys on the transport of vitamin A in human plasma. J Clinl Invest. 1971;50:2426–36.
Yeung SC, You Y, Howe KL, Yan P. Choroidal thickness in patients with cardiovascular disease: a review. Surv Ophthalmol. 2020;65:473–86.
Read SA, Fuss JA, Vincent SJ, Collins MJ, Alonso-Caneiro D. Choroidal changes in human myopia: insights from optical coherence tomography imaging. Clin Exp Optom. 2019;102:270–85.
Yamamoto H, Simon A, Eriksson U, Harris E, Berson EL, Dryja TP. Mutations in the gene encoding 11-cis retinol dehydrogenase cause delayed dark adaptation and fundus albipunctatus. Nat Genet. 1999;22:188–91.
Maw MA, Kennedy B, Knight A, Bridges R, Roth KE, Mani EJ, et al. Mutation of the gene encoding cellular retinaldehyde-binding protein in autosomal recessive retinitis pigmentosa. Nat Genet. 1997;17:198–200.
Naz S, Ali S, Riazuddin SA, Farooq T, Butt NH, Zafar AU, et al. Mutations in RLBP1 associated with fundus albipunctatus in consanguineous Pakistani families. Br J Ophthalmol. 2011;95:1019–24.
Hipp S, Zobor G, Glockle N, Mohr J, Kohl S, Zrenner E, et al. Phenotype variations of retinal dystrophies caused by mutations in the RLBP1 gene. Acta Ophthalmol. 2015;93:e281–6.
Schatz P, Preising M, Lorenz B, Sander B, Larsen M, Rosenberg T. Fundus albipunctatus associated with compound heterozygous mutations in RPE65. Ophthalmology. 2011;118:888–94.
Li SQ, Xiao XS, Yi Z, Sun WM, Wang PF, Zhang QJ. RPE65 mutation frequency and phenotypic variation according to exome sequencing in a tertiary centre for genetic eye diseases in China. Acta Ophthalmol. 2020;98:e181–90.
Littink KW, van Genderen MM, van Schooneveld MJ, Visser L, Riemslag FC, Keunen JE, et al. A homozygous frameshift mutation in LRAT causes retinitis punctata albescens. Ophthalmology. 2012;119:1899–906.
Katagiri S, Hayashi T, Nakamura M, Mizobuchi K, Gekka T, Komori S, et al. RDH5-related fundus albipunctatus in a large Japanese cohort. Invest Ophthalmol Vis Sci. 2020;61:53.
Mochizuki K, Murase H, Imose M, Kawakami H, Sawada A. Improvement of scotopic electroretinograms and night blindness with recovery of serum zinc levels. Jpn J Ophthalmol. 2006;50:532–6.
Acknowledgements
We thank Professor Emeritus Duco Hamasaki (Bascom Palmer Eye Institute) for discussions and for editing the final version of the manuscript. This work was supported by the Japan Society for the Promotion of Science (JSPS) Kakenhi grant numbers 19K09928 (to S.U.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
S. Ueno, None; S. Okado, None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Corresponding Author: Shinji Ueno
About this article
Cite this article
Ueno, S., Okado, S. Acquired night blindness due to rod dysfunction after long-term hemodialysis. Jpn J Ophthalmol 66, 1–7 (2022). https://doi.org/10.1007/s10384-021-00883-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-021-00883-z