Abstract
Purpose
To clarify the factors predictive of progression from ocular myasthenia gravis (OMG) to generalized MG (GMG) among patients with positive antiacetylcholine receptor antibodies (AChRAb) and to evaluate the efficacy of our OMG treatment protocol including tacrolimus (TAC).
Study design
Retrospective study.
Methods
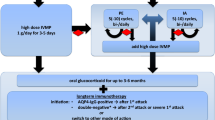
Fifty-two patients with AChRAb positivity and ptosis and/or eye movement disturbance showing diurnal fluctuation and fatigability were included. First, we compared the clinical characteristics of patients without OMG progression (p-OMG) with those of patients whose OMG transformed to GMG (TMG). Second, we subdivided the p-OMG patients according to the time of approval of TAC into the Before group and the After group. We compared the dosage and period of prednisolone administration (PSL) of the 2 groups. Third, we evaluated the responses to OMG treatment using the Myasthenia Gravis Foundation of America Postintervention Status (MGFA-PS) scale.
Results
Forty-one patients were classified into the p-OMG group, and 11, into the TMG group. The AChRAb level (P = 0.0006) and prevalence of thymoma (P = 0.001) were significantly higher in the TMG group than in the p-OMG group. In the p-OMG group, the MG composite score (P ≤ 0.0001) and AChRAb level (P = 0.005) improved after treatment. The periods of PSL administration at ≥ 20 mg/day (P = 0.009) and at 10 to 19 mg/day (P = 0.002) were significantly shorter in the After group. At the last follow-up, 78.0% of the p-OMG group patients had achieved MGFA-PS minimal manifestations or better status with PSL ≤ 5 mg/day.
Conclusions
The TMG cases were characterized by higher AChRAb levels and presence of thymoma. The P-OMG patients could be managed with our treatment protocol. Combined use of TAC was efficacious in patients with steroid-dependent p-OMG.
Similar content being viewed by others
References
Murai H, Yamashita N, Watanabe M, Nomura Y, Motomura M, Yoshikawa H, et al. Characteristics of myasthenia gravis according to onset-age: Japanese nationwide survey. J Neurol Sci. 2011;305:97–102.
Evoli A, Batocchi AP, Minisci C, Di Schino C, Tonali P. Therapeutic options in ocular myasthenia gravis. Neuromuscul Disord. 2001;11:208–16.
Li F, Hotter B, Swierzy M, Ismail M, Meisel A, Ruckert JC. Generalization after ocular onset in myasthenia gravis: a case series in Germany. J Neurol. 2018;265:2773–822.
Antonio-Santos AA, Eggenberger ER. Medical treatment options for ocular myasthenia gravis. Curr Opin Ophthalmol. 2008;19:468–78.
Mossman S, Vincent A, Newsom-Davis J. Myasthenia gravis without acetylcholine-receptor antibody: a distinct disease entity. Lancet. 1986;1:116–9.
Drachman DB. Myasthenia gravis. N Engl J Med. 1994;330:1797–810.
Palace J, Vincent A, Beeson D. Myasthenia gravis: diagnostic and management dilemmas. Curr Opin Neurol. 2001;14:583–9.
Hoch W, McConville J, Helms S, Newsom-Davis J, Melms A, Vincent A. Auto-antibodies to the receptor tyrosine kinase MuSK in patients with myasthenia gravis without acetylcholine receptor antibodies. Nat Med. 2001;7:365–8.
O'Hare M, Doughty C. Update on ocular myasthenia gravis. Semin Neurol. 2019;39:749–60.
Mittal MK, Barohn RJ, Pasnoor M, McVey A, Herbelin L, Whittaker T, et al. Ocular myasthenia gravis in an academic neuro-ophthalmology clinic: clinical features and therapeutic response. J Clin Neuromuscul Dis. 2011;13:46–52.
Apinyawasisuk S, Chongpison Y, Thitisaksakul C, Jariyakosol S. Factors affecting generalization of ocular myasthenia gravis in patients with positive acetylcholine receptor antibody. Am J Ophthalmol. 2020;209:10–7.
Jaretzki A 3rd, Barohn RJ, Ernstoff RM, Kaminski HJ, Keesey JC, Penn AS, et al. Myasthenia gravis: recommendations for clinical research standards. Task Force of the Medical Scientific Advisory Board of the Myasthenia Gravis Foundation of America. Neurology. 2000;55:16–23.
Burns TM, Conaway MR, Cutter GR, Sanders DB. Construction of an efficient evaluative instrument for myasthenia gravis: the MG composite. Muscle Nerve. 2008;38:1553–622.
Katzberg HD, Bril V. A comparison of electrodiagnostic tests in ocular myasthenia gravis. J Clin Neuromuscul Dis. 2005;6:109–13.
Hendricks TM, Bhatti MT, Hodge DO, Chen JJ. Incidence, epidemiology, and transformation of ocular myasthenia gravis: a population-based study. Am J Ophthalmol. 2019;205:99–105.
Ayzenberg I, Ellrichmann G, Schroeder C, Tonges L, Klasing A, Pappa V, et al. Brainstem encephalitis with low-titer acetylcholine receptor antibodies mimicking myasthenia gravis. Front Neurol. 2019;10:829.
Rajput R, Sachdev A, Din N, Damato EM, Murray A. False positive acetylcholine receptor antibodies in a case of unilateral chronic progressive external ophthalmoplegia: case report and review of literature. Orbit. 2018;37:385–8.
Tanaka Y, Satomi K. Overlap of myasthenia gravis and Miller Fisher syndrome. Intern Med. 2016;55:1917–8.
Plested CP, Tang T, Spreadbury I, Littleton ET, Kishore U, Vincent A. AChR phosphorylation and indirect inhibition of AChR function in seronegative MG. Neurology. 2002;59:1682–8.
Peeler CE, De Lott LB, Nagia L, Lemos J, Eggenberger ER, Cornblath WT. Clinical utility of acetylcholine receptor antibody testing in ocular myasthenia gravis. JAMA Neurol. 2015;72:1170–4.
Teo KY, Tow SL, Haaland B, Gosavi TD, Jing-Liang L, Yew Long LO, et al. Low conversion rate of ocular to generalized myasthenia gravis in Singapore. Muscle Nerve. 2018;57:756–60.
Mazzoli M, Ariatti A, Valzania F, Kaleci S, Tondelli M, Nichelli PF, et al. Factors affecting outcome in ocular myasthenia gravis. Int J Neurosci. 2018;128:15–24.
Aguirre F, Villa AM. Prognosis of ocular myasthenia gravis in an Argentinian population. Eur Neurol. 2018;79:113–7.
Lindstrom JM, Seybold ME, Lennon VA, Whittingham S, Duane DD. Antibody to acetylcholine receptor in myasthenia gravis: prevalence, clinical correlates, and diagnostic value. Neurology. 1976;26:1054–9.
Kupersmith MJ, Latkany R, Homel P. Development of generalized disease at 2 years in patients with ocular myasthenia gravis. Arch Neurol. 2003;60:243–8.
Kupersmith MJ. Ocular myasthenia gravis: treatment successes and failures in patients with long-term follow-up. J Neurol. 2009;256:1314–20.
Mullaney P, Vajsar J, Smith R, Buncic JR. The natural history and ophthalmic involvement in childhood myasthenia gravis at the hospital for sick children. Ophthalmology. 2000;107:504–10.
Kupersmith MJ, Ying G. Ocular motor dysfunction and ptosis in ocular myasthenia gravis: effects of treatment. Br J Ophthalmol. 2005;89:1330–4.
Ozawa Y, Uzawa A, Kanai T, Oda F, Yasuda M, Kawaguchi N, et al. Efficacy of high-dose intravenous methylprednisolone therapy for ocular myasthenia gravis. J Neurol Sci. 2019;402:12–5.
Komiyama A, Arai H, Kijima M, Hirayama K. Extraocular muscle responses to high dose intravenous methylprednisolone in myasthenia gravis. J Neurol Neurosurg Psychiatry. 2000;68:214–7.
Jaretzki A 3rd, Barohn RJ, Ernstoff RM, Kaminski HJ, Keesey JC, Penn AS, et al. Myasthenia gravis: recommendations for clinical research standards. Task Force of the Medical Scientific Advisory Board of the Myasthenia Gravis Foundation of America. Ann Thorac Surg. 2000;70:327–34.
Gilhus NE. Autoimmune myasthenia gravis. Expert Rev Neurother. 2009;9:351–8.
Sanders DB, Evoli A. Immunosuppressive therapies in myasthenia gravis. Autoimmunity. 2010;43:428–35.
Grob D, Brunner N, Namba T, Pagala M. Lifetime course of myasthenia gravis. Muscle Nerve. 2008;37:141–9.
Masuda M, Utsugisawa K, Suzuki S, Nagane Y, Kabasawa C, Suzuki Y, et al. The MG-QOL15 Japanese version: validation and associations with clinical factors. Muscle Nerve. 2012;46:166–73.
Zhang Z, Yang C, Zhang L, Yi Q, Hao Z. Efficacy and safety of tacrolimus in myasthenia gravis: a systematic review and meta-analysis. Ann Indian Acad Neurol. 2017;20:341–7.
Nishida Y, Takahashi YK, Kanai T, Nose Y, Ishibashi S, Sanjo N, et al. Safety of tapering tacrolimus dose in patients with well-controlled anti-acetylcholine receptor antibody-positive myasthenia gravis. Euro J Neurol. 2020;27:100–4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Y. Isshiki, None; O. Mimura, Lecture fee (Santen, Senju, Otsuka, Pfizer, Alcon, GSK); F. Gomi, Grant, Lecture fee (Pfizer, Novartis, Santen, Senju), Grant (Alcon), Lecture fee (Bayer).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Corresponding Author: Yoshihiko Isshiki
About this article
Cite this article
Isshiki, Y., Mimura, O. & Gomi, F. Clinical features and treatment status of antiacetylcholine receptor antibody-positive ocular myasthenia gravis. Jpn J Ophthalmol 64, 628–634 (2020). https://doi.org/10.1007/s10384-020-00770-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-020-00770-z