Summary
Background
Diverticular disease appears to be one of the most common conditions in the Western world. The standard approach in treatment of diverticular disease is a laparoscopic resection, usually after an inflammation-free time of 4 to 6 weeks. The aim of this study was to evaluate the timing of operation.
Materials and methods
A total of 61 patients underwent left-sided colonic resection because of diverticular disease between January 2017 and February 2020. Because of complicated diverticulitis (CDD stage 2a or 2b) 37 patients were treated either early within 7 days after first symptoms (group A: n = 17) or delayed about 6 weeks after the first contact and conservative therapy (group B: n = 20).
Results
Overall mortality was 0%. The average operation time was shorter in the early elective group (group A: 140.4 min vs. group B: 151.2 min; p = 0.29). The hospital stay (group A: 9.9 days vs. group B: 16.9 days) and the postoperative stay (group A: 4.8 days vs. group B: 8.1 days) were significantly longer in group B (p = 0.01). We observed—although not reliable due to the low number of patients—more postoperative complications in the delayed group (group A: 5.9% vs. group B: 15.0%; p = 0.61).
Conclusion
The data in this study confirm the early operation as safe and efficient due to lower costs. We can recommend an early approach in selected cases with the first episode of a complicated diverticulitis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diverticular disease was first described in 1928 by Finny et al. [1]. Its complications continue to be a burden on health care systems. In the United States the most common gastrointestinal tract diagnoses are diverticulitis and diverticular hemorrhage among hospitalized patients [2, 3]. Diverticular disease appears to be one of the most common conditions in the Western world. Compared to the 1990s, the incidence of diverticulitis increased by 50% from 2000 to 2007 [3]. The prevalence of diverticulosis is about 28–45% and over 60% at the age of 70 years [4]. Age, BMI, and male sex are well-known risk factors for diverticulosis.
Diverticular disease was historically classified by Hinchey et al. [5]. This classification strongly depends on intraoperative surgical findings and does not implicate a recommandation which operative strategy should be chosen for different occurence of diverticular disease. The Hansen–Stock classification also included chronic diverticulitis and uncomplicated cases and was more commonly used in Europe in the past [6]. In 2014 a novel classification was introduced according to the Hansen–Stock classification (classification of diverticular disease, CDD) [4].
The treatment of diverticulitis is still under debate: in uncomplicated diverticulitis a change of paradigm has taken place in recent years and moved away from automatic treatment using antibiotics [7,8,9]. While in the past elective surgery was indicated very early after two attacks of uncomplicated diverticulitis, the indication switched to complicated diverticulitis cases regardless of the number of previous attacks. Studies showed that the number of attacks of diverticulitis is not necessarily a predictive factor in defining the suitability of surgery [10]. Only a fraction of 5–7% develop a complicated diverticulitis during recurrent attacks [11, 12]. One of the main questions in surgery is which patients would profit from diverticulitis surgery? It should be considered that surgery carries significant risks in terms of morbidity and mortality.
There are currently two options regarding the time of surgery: the delayed elective variant, in which the operation is done during an inflammation-free stage [13, 14], and the early elective option, in which the operation is performed in the acute inflammation stage [15, 16]. Delayed surgery can lead to more adhesions and fibrosis, which are possibly associated with higher morbidity. An early operation within the first week after diagnosis was adapted from practice in the therapy of acute cholecystitis [17], although acute cholecystitis and complicated diverticulitis are two completely different entities. Both options have been in concurrent use in the past [14, 18].
The aim of this study was to retrospectively evaluate the outcome of laparoscopic sigmoid resection in two groups (A and B) differing only in the timing of surgery for acute complicated diverticular disease with either abscessing or covered perforation according to CDD stage 2a and 2b [4].
Materials and methods
After an institutional review board, data were obtained from a retrospectively maintained database of our department. 61 patients underwent laparoscopic left-sided colonic resection because of diverticular disease between January 2017 and February 2020. 37 patients were treated because of complicated diverticulitis according to CDD stage 2a or 2b. Every patient received a CT scan before therapy to determinate the stage according CDD [19]. In the case of complicated diverticulitis, the patients were transferred to surgery either early (group A) within the first week after the CT scan (Fig. 1) or delayed (group B) about 4 to 6 weeks after the CT scan. A coloscopy was performed routinely 3 months after the operation in the early group or the day before operation in the delayed patients. The timing of operation (either early or delayed) was left to patients’ preferences, clinical manifestation, and operation theatre resources. All the patients initially received intravenous antibiotic therapy directly after diagnosis. None of the patients in either group had previous attacks.
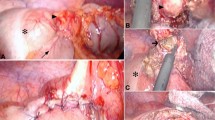
41-year-old male patient with covered perforation in acute diverticulitis according to CDD stage 2b [19]. Early approach: a CT scan before operation (red arrow covered perforation into sigmoid mesocolon), b intraoperative situs (asterisk abscess formation), c after incision of the peritoneum, exposure of the holy plane, and dissection of the sigmoid vessels (2 asterisks), d mobilization of sigmoid and descending colon
Perforated diverticulitis according to stage CDD 2c was excluded from our study, as were chronic diverticulitis and associated fistulas or stenosis (CDD stage 3).
The American Society of Anesthesiologists (ASA) score was used to compare the comorbidities of the two groups [20]. The ASA score was defined by our anesthesiologic team preoperatively in accordance with patients’ physical conditions.
The resections were performed by our surgical team at the Feldkirch academic teaching hospital. The majority of cases (94.6%) were performed in a reduced-port technique using an umbilical single-port system (OCTO™ Port, Dalim SurgNet, Seoul, South Korea) and an additional 5‑mm trocar suprasymphyseal, the minority of patients (5.4%) were treated with a conventional multiport technique with a Pfannenstiel incision for specimen retraction. Manual bowel preparation was done in all patients routinely. The severity of complications was graded according the Clavien–Dindo classification for surgical complications [21].
The duration of hospital stay was recorded from first presentation to the patient’s discharge from hospital. All hospital stays due to the same reason (ICD 10 code: K57.2-) were added up and resulted in length of hospital stay.
All data were evaluated using Microsoft® Office Excel (version 2019) and the statistical analyses were performed using Java-based tools. Quantitative datasets were analyzed using the two-tailed t‑test. We used the chi-square test or, if necessary, the Fisher’s exact test for category date. Quantitative data were represented by the average and its corresponding standard deviation. Differences were regarded as statistically significant at p < 0.05.
Results
Data from a cohort of 37 patients were retrospectively examined. All patients received surgery because of complicated diverticulitis, either CDD stage 2a or 2b. The patients were divided in two groups according to the timing of operation. Group A (early resection) consisted of 17 electively operated patients (45.9%), while group B (delayed resection) consisted of 20 cases (54.1%). Patients’ characteristics in group A and group B are shown in Table 1.
Group A consisted of 11 males and 6 females with an average age of 56.2 years (± 11). The average BMI was in this group 25.2 kg/m2 (± 2 kg/m2). Comorbidities were recorded in 23.5% (n = 4) of the patients in this group, the average ASA score was 2. In 70.6% (n = 12) of cases surgery was indicated because of CDD stage 2b, in 29.4% (n = 5) because of CDD stage 2a.
Group B consisted of 9 males and 11 females with a mean age of 59.9 years (± 12). The average BMI in this group was 25.4 kg/m2 (± 4.2 kg/m2). Comorbidities were recorded in 40.0% (n = 8) of the patients in this group, 50.0% (n = 10) were ASA score 2. In 85.0% (n = 17) of cases surgery was indicated because of CDD stage 2b, in 15.0% (n = 3) because of CDD stage 2a.
Data regarding the outcome of both groups are shown in Table 2.
The average operation time in group A was 140.7 min (± 24.6) and 151.2 min (± 38.9) in group B, which was not significant (p = 0.29). No ICU time was necessary in either group. The total hospital stay differed significantly (p = 0.01): In group A the total hospital stay was 9.9 days (± 2.4), in group B 16.9 days (± 9.6). Also, the postoperative stay was significantly longer in group B than in group A (p = 0.01). In group B we observed a median postoperative stay of 8.1 days (± 4.8), in group A a median postoperative stay of 4.8 days (± 1.2).
88.2% (n = 15) of the cases in group A were operated with a reduced-port technique, 11.8% (n = 2) in a conventional multiport laparoscopic technique. In group B 100% (n = 20) were treated with the reduced-port technique. We observed a conversion rate of 15.0% (n = 3) in group B, no conversions were necessary in group A (p = 0.23). We found no intraoperative complications in group A and one patient with a ureteral injury (5.0%) in group B (p = 1.00). The postoperative complication rate did not differ significantly between the groups (p = 0.61), but we observed more complications in the delayed group. In group A 5.9% (n = 1) suffered a postoperative complication, in group B 15.0% (n = 3). The anastomotic leakage rate was 5.9% (n = 1) in group A and 10% (n = 2) in group B, which was not significant (p = 1.00). Anastomotic bleeding was not observed in either group, whereas an infected hematoma occurred in one case of group B. Re-laparoscopy plus a diverting ileostomy and Endo VAC was necessary in the case of group A. A Hartmann procedure was performed in one case of anastomotic leakage of group B, the other leakage could be treated laparoscopically with a diverting ileostomy. A CT intervention was indicated in the case of the infected hematoma. No patient died in the course of treatment.
Because more complicated diverticulitis cases according to CDD stage 2b occurred in group B, we also analyzed both approaches according to their stage of diverticulitis. Subgroup analysis of CDD stage 2a is shown in Table 3, CDD stage 2b is shown in Table 4.
The average operation time according the subgroup of CDD stage 2a was 144.4 min (± 15.78 min) in group A and 158 min (± 23.06 min) in group B (p = 0.35). The total hospital stay was 9.8 days (± 1.30) in group A and 20.66 days (± 11.01) in group B, which was significant (p = 0.05). The postoperative stay was 15 days (± 7.93) in group B, whereas in group A the average postoperative stay was 4.4 days (± 0.89) (p = 0.02). This was mainly caused by two complications which we observed in the delayed group of CDD stage 2a, and consequently not significant (p = 0.11).
The average operation time according the subgroup of CDD stage 2b was 138.75 min (± 27.97) in group A and 150 min (± 36.99) in group B (p = 0.38). The total hospital stay was 10 days (± 2.76) in group A and 16.17 days (± 9.53) in group B, which was significant (p = 0.04). The postoperative stay was 6.82 days (± 3.00) in group B, whereas in group A the average postoperative stay was 5 days (± 1.34) (p = 0.06). In both groups one complication occurred (p = 1.00).
Discussion
Diverticulitis surgery can result in a high morbidity and mortality rate in case of any postoperative complication. In light of this, patient selection and indication becomes even more important. 2.7 million outpatient visits and 200,000 inpatient admissions at a cost of more than $ 2 billion were seen in the United States annually [22,23,24]; therefore, diverticulitis is a burden for the health system. The prevalence of diverticulosis between 50 and 70 years is more than 30% [4, 25, 26] and the incidence among individuals between 40 and 49 years rose by 132% between 1980 and 2007 [24, 27].
The surgical standard technique should be a laparoscopic sigmoid resection regardless of the stage of diverticulitis [28, 29]. Whereas two episodes of mild diverticulitis were enough for the indication for surgery in the past, the course of treatment has shifted toward more conservative therapy regardless of the number of attacks [10, 30,31,32,33]. However, in 2015, Schildberg et al. described that 40% of all diverticulitis cases in Germany received an operation, most of these cases were staged as an uncomplicated diverticulitis [34]. Other studies showed that 38% of patients with recurrent diverticulitis would have preferred an earlier operation [35]. We know that the complication rate varies depending on diverticulitis stage. The risk profile is higher in patients with complicated diverticulitis [15, 36].
The patients’ characteristics in our study are comparable to the literature in terms of proportion of females and age. The operative time, the total hospital stay, and the postoperative stay differ to the literature. We observed better results in the early operated group. An overall anastomotic leakage rate of 8.1% is comparable to the literature; 5.4% occurred in the delayed group. This may be caused by different study groups, although not statistically significant at this sample size: the delayed group had 85% cases of complicated diverticulitis with a macro-abscess (compared to 70% in the early group) and comorbidities were more frequent (40%) compared to the early group (23.5%). In the subgroup analysis the total hospital stay was significantly longer in group B. Also, postoperative stay and the operative time were expanded, but not significantly.
The most efficient approach to surgical treatment regarding the timing of operation in complicated diverticulitis is still under debate. The most recent studies prefer the operation during the inflammation-free interval [13, 14]. It was shown that the minimum safe period between acute inflammation and surgery should be at least 4–6 weeks and this was the time interval we used for group B [37].
During the study period, the decision for surgery was indicated according to the recommendation of the guidelines [4]. A CT scan to classify the stage of diverticulitis is necessary. A coloscopy should be performed in all cases if diverticular surgery is indicated. A recently published systematic review of Koo et al. showed that in patients treated for CT-confirmed complicated diverticulitis, malignancies occurred in 6.14% [38]. In case of an early approach, preoperative endoscopy is recommended in cancer-suspicious cases. In all other (“clear”) cases, especially in case of covered perforation, we would suggest the coloscopy 3 months after the operation. In delayed cases we performed the endoscopy preoperatively 1 day before surgery.
With the first “early” cases we operated, we started to recognize that the hospital stays and the postoperative stays were shorter. Furthermore, chronic fibrotic adhesions were not that distinctive than in the delayed group. The inflammation was in an acute stage, the subjective surgical confidence was higher intraoperatively, separation of the layers appeared easier, and this resulted in a trend toward shorter operative time in the early resection group. The stage of inflammation in diverticulitis surgery is the key to safe and successful surgery. Zdichavsky et al. showed in 2013 that in histopathological examination of surgical specimens in early resected patients, the stage of inflammation was more acute than in delayed cases [16]. Our intraoperative findings confirmed this study in complicated diverticulitis disease (Fig. 2).
Histopathological examination of a covered perforation in acute diverticulitis according CDD stage 2b [19]. a Hematoxylin and eosin (HE) stain shows mucosa (asterisk), the area of perforation (arrow), and abscess formation (2 asterisks). b HE stain in higher resolution shows mucosa with inflammation (asterisk), food residue (2 asterisks), and abscess formation (circle)
We observed—although not significant—more postoperative complications in the delayed group. As mentioned before, this is not significant, but may be a trend toward a better outcome in early operated patients. Regardless of the timing of surgery in diverticular disease, the importance of early diagnosis in case of complications and rapid complication management by experienced colorectal surgeons is generally necessary in colorectal surgery.
A recommendation according to the timing of surgery for complicated diverticulitis cannot be given on the basis of our data. A trend toward shorter operative time and reduced hospital stays is shown in this study. The low number of patients and retrospective analysis are the main limitations of this study. An early operation approach is safe and showed comparable morbidity in the literature as well as in our study [16]. Detailed patient history is required not to miss past episodes of diverticulitis. In these cases, a delayed operation might be the better approach.
Conclusion
The indication for surgery in diverticulitis should be considered very carefully. The timing of the operation is still under debate. In our study we could show no difference between an early approach and a delayed operation in terms of complications, as shown in the literature in the past. But there was a significant difference in hospital stay, which results in more costs in the delayed group. This might be a reason for the consideration for an early approach. A further randomized trial is needed.
Abbreviations
- ASA:
-
American Society of Anesthesiologists
- BMI:
-
Body mass index
- CDD:
-
Classification of Diverticular Disease
- CT:
-
Computer tomography
- ICD:
-
International Classification of Disease
- ICU:
-
Intensive care unit
- HE:
-
Hematoxylin and eosin
References
Finney JMT. Diverticulitis and its surgical treatment. Proc Interstate Post-grad Med Assembly North Am. 1928;55:57–65.
Talabani JA, Lydersen S, Ness-Jensen E, et al. Risk factors of admission for acute colonic diverticulitis in a population-based cohort study: the North Trondelag Health Study, Norway. World J Gastroenterol. 2016;22(48):10663–72. https://doi.org/10.3748/wjg.v22.i48.10663.
Lee TH, Setty PT, Parthasarathy G, et al. Aging, obesity, and the incidence of diverticulitis: a population-based Study. Mayo Clin Proc. 2018;93(9):1256–65. https://doi.org/10.1016/j.mayocp.2018.03.005.
Leifeld L, Germer CT, Böhm S, Dumoulin FL, Häuser W, Kreis M, Labenz J, Lembcke B, Post S, Reinshagen M, Ritz JP, Sauerbruch T, Wedel T, von Rahden B, Kruis W. S2k-Leitlinie Divertikelkrankheit/Divertikulitis [S2k guidelines diverticular disease/diverticulitis]. Z Gastroenterol. 2014;52(7):663–710. https://doi.org/10.1055/s-0034-1366692. PMID: 25026009
Hinchey EJ, Schaal PG, Richards GK. Treatment of perforated diverticular disease of the colon. Adv Surg. 1978;12:85–109.
Hansen O, Stock W. Prophylaktische Operation bei der Divertikelkrankheit des Kolons – Stufenkonzept durch exakte Stadieneinteilung (article in German). Langenbecks Arch Chir. 1999;384(suppl 2):1257–60.
Chabok A, Påhlman L, Hjern F, et al. Randomized clinical trial of antibiotics in acute uncomplicated diverticulitis. Br J Surg. 2012;99(4):532–9. https://doi.org/10.1002/bjs.8688.
Schug-Pass C, Geers P, Hügel O, et al. Prospective randomized trial comparing short-term antibiotic therapy versus standard therapy for acute uncomplicated sigmoid diverticulitis. Int J Colorectal Dis. 2010;25(6):751–9. https://doi.org/10.1007/s00384-010-0899-4.
Hjern F, Josephson T, Altman D, et al. Conservative treatment of acute colonic diverticulitis: Are antibiotics always mandatory? Scand J Gastroenterol. 2007;42(1):41–7. https://doi.org/10.1080/00365520600780650.
van de Wall BJM, Draaisma WA, Consten ECJ, et al. DIRECT trial. Diverticulitis recurrences or continuing symptoms: operative versus conservative treatment. A multicenter randomised clinical trial. BMC Surg. 2010;10:25. https://doi.org/10.1186/1471-2482-10-25.
Moreno AM, Wille-Jørgensen P. Long-term outcome in 445 patients after diagnosis of diverticular disease. Colorectal Dis. 2007;9(5):464–8. https://doi.org/10.1111/j.1463-1318.2006.01173.x.
Anaya DA, Flum DR. Risk of emergency colectomy and colostomy in patients with diverticular disease. Arch Surg. 2005;140(7):681–5. https://doi.org/10.1001/archsurg.140.7.681.
Siewert J. Frühelektive Chirurgie der akuten Divertikulitis des Colons. Chirurg. 1995;66(12):1182–9.
Reissfelder C, Buhr HJ, Ritz J‑P. What is the optimal time of surgical intervention after an acute attack of sigmoid diverticulitis: early or late elective laparoscopic resection? Dis Colon Rectum. 2006;49(12):1842–8. https://doi.org/10.1007/s10350-006-0730-z.
Warwas FB, Schneider B. Elective vs. early elective surgery in diverticular disease: a retrospective study on the optimal timing of non-emergency treatment. Int J Colorectal Dis. 2018;33(5):531–9. https://doi.org/10.1007/s00384-018-3022-x.
Zdichavsky M, Kratt T, Stüker D, et al. Acute and elective laparoscopic resection for complicated sigmoid diverticulitis: clinical and histological outcome. J Gastrointest Surg. 2013;17(11):1966–71. https://doi.org/10.1007/s11605-013-2296-0.
Gutt CN, Encke J, Köninger J, et al. Acute cholecystitis: early versus delayed cholecystectomy, a multicenter randomized trial (ACDC study, NCT00447304). Ann Surg. 2013;258(3):385–93. https://doi.org/10.1097/SLA.0b013e3182a1599b.
Reissfelder C, Buhr HJ, Ritz J‑P. Can laparoscopically assisted sigmoid resection provide uncomplicated management even in cases of complicated diverticulitis? Surg Endosc. 2006;20(7):1055–9. https://doi.org/10.1007/s00464-005-0529-3.
Pustelnik D, Elsholtz FHJ, Bojarski C, et al. Das CDD-System in der computertomografischen Diagnostik der Divertikelkrankheit (The CDD System in Computed Tomographic Diagnosis of Diverticular Disease). Rofo. 2017;189(8):740–7. https://doi.org/10.1055/s-0043-110770.
Little JP. Consistency of ASA grading. Anaesthesia. 1995;50(7):658–9.
Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250(2):187–96. https://doi.org/10.1097/SLA.0b013e3181b13ca2.
Peery AF, Crockett SD, Barritt AS, et al. Burden of gastrointestinal, liver, and pancreatic diseases in the United States. Gastroenterology. 2015;149(7):1731–1741.e3. https://doi.org/10.1053/j.gastro.2015.08.045.
Peery AF, Dellon ES, Lund J, et al. Burden of gastrointestinal disease in the United States: 2012 update. Gastroenterology. 2012;143(5):1179–1187.e3. https://doi.org/10.1053/j.gastro.2012.08.002.
Strate LL, Morris AM. Epidemiology, pathophysiology, and treatment of diverticulitis. Gastroenterology. 2019;156(5):1282–1298.e1. https://doi.org/10.1053/j.gastro.2018.12.033.
Jun S, Stollman N. Epidemiology of diverticular disease. Best Pract Res Clin Gastroenterol. 2002;16(4):529–42. https://doi.org/10.1053/bega.2002.0328.
Delvaux M. Diverticular disease of the colon in Europe: epidemiology, impact on citizen health and prevention. Aliment Pharmacol Ther. 2003;18(Suppl 3):71–4. https://doi.org/10.1046/j.0953-0673.2003.01720.x.
Bharucha AE, Parthasarathy G, Ditah I, et al. Temporal trends in the incidence and natural history of diverticulitis: a population-based study. Am J Gastroenterol. 2015;110(11):1589–96. https://doi.org/10.1038/ajg.2015.302.
Pendlimari R, Touzios JG, Azodo IA, et al. Short-term outcomes after elective minimally invasive colectomy for diverticulitis. Br J Surg. 2011;98(3):431–5. https://doi.org/10.1002/bjs.7345.
Gaertner WB, Kwaan MR, Madoff RD, et al. The evolving role of laparoscopy in colonic diverticular disease: a systematic review. World J Surg. 2013;37(3):629–38. https://doi.org/10.1007/s00268-012-1872-x.
Shaikh S, Krukowski ZH. Outcome of a conservative policy for managing acute sigmoid diverticulitis. Br J Surg. 2007;94(7):876–9. https://doi.org/10.1002/bjs.5703.
Mueller MH, Glatzle J, Kasparek MS, et al. Long-term outcome of conservative treatment in patients with diverticulitis of the sigmoid colon. Eur J Gastroenterol Hepatol. 2005;17(6):649–54. https://doi.org/10.1097/00042737-200506000-00009.
Chautems RC, Ambrosetti P, Ludwig A, et al. Long-term follow-up after first acute episode of sigmoid diverticulitis: Is surgery mandatory? A prospective study of 118 patients. Dis Colon Rectum. 2002;45(7):962–6. https://doi.org/10.1007/s10350-004-6336-4.
Regenbogen SE, Hardiman KM, Hendren S, et al. Surgery for diverticulitis in the 21st century: a systematic review. JAMA Surg. 2014;149(3):292–303. https://doi.org/10.1001/jamasurg.2013.5477.
Schildberg CW, Schellerer V, Croner R, et al. Operationsindikation der Sigmadivertikulitis in Deutschland: Sind alle Operationen gerechtfertigt? (Surgical Indications for Diverticulitis in Germany: Are All Operations Justified?). Zentralbl Chir. 2015;140(6):585–90. https://doi.org/10.1055/s-0032-1328593.
Käser SA, Glauser PM, Basilicata G, et al. Timing of rectosigmoid resection for diverticular disease: the patient’s view. Colorectal Dis. 2012;14(3):e111–e6. https://doi.org/10.1111/j.1463-1318.2011.02801.x.
Ambrosetti P, Grossholz M, Becker C, et al. Computed tomography in acute left colonic diverticulitis. Br J Surg. 1997;84(4):532–4. https://doi.org/10.1046/j.1365-2168.1997.02576.x.
Rafferty J, Shellito P, Hyman NH, et al. Practice parameters for sigmoid diverticulitis. Dis Colon Rectum. 2006;49(7):939–44. https://doi.org/10.1007/s10350-006-0578-2.
Koo CH, Chang JHE, Syn NL, et al. Systematic Review and Meta-analysis on Colorectal Cancer Findings on Colonic Evaluation After CT-Confirmed Acute Diverticulitis. Dis Colon Rectum. 2020;63(5):701–9. https://doi.org/10.1097/DCR.0000000000001664.
Acknowledgements
The authors would like to thank Simone Minikus for English and spelling corrections and Dr. Susanne Dertinger (Institute for Pathology, LKH Feldkirch) for the histopathological pictures.
Author information
Authors and Affiliations
Contributions
P. Tschann, D. Lechner, B. Feurstein, P. Girotti, S. Adler, M. Hufschmidt, and I. Königsrainer contributed to the study design. P. Tschann, D. Lechner, and B. Feurstein carried out data acquisition. P. Tschann and D. Lechner contributed the data analysis. P. Tschann drafted the manuscript. P. Tschann, D. Lechner, B. Feurstein, P. Girotti, S. Adler, M. Hufschmidt, and I. Königsrainer reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
P. Tschann, D. Lechner, B. Feurstein, S. Adler, P. Girotti, M. Hufschmidt, and I. Königsrainer declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tschann, P., Lechner, D., Feurstein, B. et al. Early or delayed sigmoid resection in complicated diverticular disease? A single-center experience. Eur Surg 53, 16–22 (2021). https://doi.org/10.1007/s10353-020-00681-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10353-020-00681-w