Summary
Background
Wound healing deficits and subsequent surgical site infections are potential complications after surgical procedures, resulting in increased morbidity and treatment costs. Closed-incision negative-pressure wound therapy (ciNPWT) systems seem to reduce postoperative wound complications by sealing the wound and reducing tensile forces.
Materials and methods
We conducted a collaborative English literature review in the PubMed database including publications from 2009 to 2020 on ciNPWT use in five surgical subspecialities (orthopaedics and trauma, general surgery, plastic surgery, cardiac surgery and vascular surgery). With literature reviews, case reports and expert opinions excluded, the remaining 59 studies were critically summarized and evaluated with regard to their level of evidence.
Results
Of nine studies analysed in orthopaedics and trauma, positive results of ciNPWT were reported in 55.6%. In 11 of 13 (84.6%), 13 of 15 (86.7%) and 10 of 10 (100%) of studies analysed in plastic, vascular and general surgery, respectively, a positive effect of ciNPWT was observed. On the contrary, only 4 of 12 studies from cardiac surgery discovered positive effects of ciNPWT (33.3%).
Conclusion
ciNPWT is a promising treatment modality to improve postoperative wound healing, notably when facing increased tensile forces. To optimise ciNPWT benefits, indications for its use should be based on patient- and procedure-related risk factors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Wound complications following surgery may include dehiscence, prolonged secretion and infection. Surgical site infections (SSI) account for approximately 20% of all hospital-acquired infections and result in a prolonged length of stay, increased readmission rate and higher treatment costs [1]. In 2 to 5% of patients undergoing inpatient elective surgery, SSI develop after an average length of stay of 9.7 days [1, 2]. Overall, costs of SSI treatment are estimated to amount to 3.5 to 10 billion dollars in the United States per year [2]. About 60% of SSI are said to be preventable [2]. Several risk factors associated with SSI have been identified and can be divided into intrinsic (i.e. patient-related) and extrinsic (i.e. procedure-related) factors. Intrinsic risk factors can be subdivided into modifiable (e.g. obesity, alcoholism and smoking, but also diabetes, immunosuppression) and non-modifiable (patient age, skin quality and/or microbial environment) [1]. Likewise, extrinsic factors can be based on the type of surgical procedure, surgical protocols used for perioperative management and other factors related to the healthcare facility (e.g. surgical caseload per operating theatre, condition of sterile equipment and anaesthetic/surgical infrastructure) [2].
Postoperative wound management can be optimized by surgical suture material and wound dressings, as well as by timing and frequency of dressing changes [1]. Closed-incision negative-pressure wound therapy (ciNPWT) systems represent a relatively new modulator in this sector.
Closed-incision negative-pressure wound therapy
Postoperative wound management consists of dressing changes based on the postoperative protocol as outlined by the surgeon or facility, and usually involves repetitive intervals for removal of dressings, disinfection and the re-application of a wound dressing [3]. However, every dressing change disrupts the wound’s microenvironment and potentially increases the risk of SSI. Thus, dressing materials have been developed to remain in place for several days to improve barrier function and support the wound healing process [3, 4].
Closed-incision negative-pressure wound therapy (ciNPWT) has gained importance during the past 20 years [3, 5]. In practice, ciNPWT is applied to a closed/sutured, previously cleaned incision under sterile conditions. A foam is placed on the wound, with or without interposed non-adherent dressings, followed by an occlusive drape [3]. Subsequently, negative pressure is applied to the closed system via a suction mechanism (Fig. 1). Immediate effects of ciNPWT include maintenance of the wound’s microenvironment, protection from external contamination, reduction of tensile forces at the wound edges, enhancement of appositional strength, normalization of stress distribution and improvement of skin perfusion [3]. Decreased formation of haematoma and seroma were linked to improved vascular and lymphatic flow, and may be an additional mid-term benefit of ciNPWT [3]. In the long term, application of ciNPWT was associated with improved mechanical strength of the scar due to a downregulation of genes associated with hypoxia, scarring, impaired wound healing, inflammation and delayed re-epithelialization in early wound healing stages [3, 6]. The increased use of ciNPWT after surgery is reflected by the rising number of publications and manufacturers selling ciNPWT devices.
The aim of this review was to provide an overview of the medical literature on ciNPWT in different surgical subspecialties and derive best-practice recommendations highlighting their level of evidence.
Methods
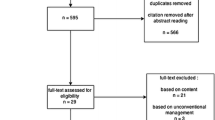
MEDLINE and PubMed were searched for studies analysing ciNPWT use in predefined surgical fields, i.e. trauma and orthopaedics, general surgery, plastic surgery, vascular surgery and cardiac surgery, published from 2009 to 2020. Retrospective case series, case–control studies, prospective non-randomised comparative studies, prospective randomised controlled trials (RCTs), systematic reviews and meta-analyses published in English language were eligible. Case reports, expert opinions, articles in languages other than English, non-systematic reviews, animal studies and study protocols were excluded. All studies included were subsequently classified into levels of evidence (ranging from I to V) according to the Oxford Centre for Evidence-Based Medicine (OCEBM) [7]. Studies were summarised according to the main surgical field where ciNPWT usage had been analysed into the subsections “trauma and orthopaedics”, “plastic surgery”, “cardiac surgery”, “vascular surgery” and “general surgery”. The latter subsection also contained meta-analyses grouping together studies from different surgical specialities.
Results
Trauma and orthopaedics
Total hip (THA) and knee arthroplasties (TKA) are increasingly performed for osteoarthritis of hip and knee [8]. Although wound complications are considered a rare event, they may have dramatic consequences, potentially leading to development of early periprosthetic joint infections [9]. Operative interventions for hand trauma were frequently associated with postoperative wound healing deficits. Proximal tibial and distal tibial (pilon) fractures can be accompanied by SSI in up to 27.0 and 40% of cases, respectively [10,11,12]. Consequently, prevention of wound healing problems in both elective and emergency settings is of increasing importance. Overall, we identified three retrospective studies, five RCTs and one systematic review published on the use of ciNPWT in orthopaedic or trauma cases (Table 1).
Cooper et al. [13] performed a retrospective case–control study including 59 patients with periprosthetic fractures of the hip (n = 44) and knee (n = 15), as well as 8 patients with interprosthetic fractures. Of these, 27 were treated with an ciNPWT system following surgery. The remaining 40 patients underwent conventional wound care. During the postoperative course, the rate of deep SSI was significantly lower in patients treated with ciNPWT in comparison to controls (0.0% vs. 25.0%; p = 0.004), as were the overall wound complication rate (4.0% vs. 35.0%; p = 0.002) and the revision rate (4.0% vs. 25.0%; p = 0.021). The authors concluded that ciNPWT may be used to reduce wound healing deficits in patients following periprosthetic lower limb fractures (level IV) [13]. In another retrospective case–control study, 309 patients undergoing 403 major lower limb amputations for non-traumatic indications were analysed with a specific focus on the potential effect of ciNPWT on postoperative survival rates [14]. Most common indications for lower limb amputation were diabetes and chronic wounds. Altogether, 139 patients were treated with ciNPWT post amputation, and 170 received a conventional stump bandage. There was no significant difference in patient survival depending on the type of postoperative wound management used (ciNPWT vs. conventional stump bandage: 52.0 and 36.0% vs. 49.0 and 39.0% survival at 2 and 3 years, respectively; p = 0.970). Furthermore, there was no significant difference between the two groups with regards to rate of revision surgery (12.2% vs. 15.9%; ciNPWT vs. control; p = 0.740). The authors concluded that ciNPWT does not improve survival probability in patients undergoing major lower limb amputation for non-traumatic indications (level IV) [14]. Zayan et al. [15] published results of a retrospective case series including 4 patients with upper limb amputation and 21 patients with lower limb amputation. Seventeen operations were primary amputations, whilst eight interventions constituted revisions for failed amputation. All patients had been treated with ciNPWT following amputation. The SSI rate was 4.0%, whilst no seroma, haematoma or wound dehiscence was observed (level IV) [15].
In the RCT by Newman et al., 160 patients at a high risk of postoperative wound complications undergoing revision TKA or THA were randomly assigned to the intervention group (ciNPWT, n = 80) or control group (standard dressing; n = 80). Specifically, at least one of the following risk factors was present in any participant: human immunodeficiency virus infection (2.5%), history of organ transplant (1.3%), immunosuppression (16.3%), history of periprosthetic joint infection (35.6%), current smoker (12.5%), depression (41.9%), peripheral vascular disease (10.0%), use of anticoagulants other than aspirin (39.4%), diabetes (23.1%), body mass index >35 kg/m2 (33.1%), liver disease (5.0%), malnutrition (0.6%), inflammatory arthritis (18.1%), renal failure (7.5%) or presence of haematological/oncological disease (2.5%). In this high-risk cohort, the use of ciNPWT was associated with a significant reduction of postoperative wound complication rate in comparison to controls (10.1% vs. 23.8%; p = 0.022), as well as with regards to revision rate (2.5% vs. 12.5%; p = 0.017). However, readmission rate was comparable between patients treated with ciNPWT and controls (20.3% vs. 23.8%; p = 0.595). Based on their observations, the authors concluded that ciNPWT treatment may reduce the risk of wound healing deficits in high-risk patients undergoing revision arthroplasty (level II) [16]. Kim et al. [9] performed a systematic review and meta-analysis of patients receiving ciNPWT (n = 790) or conventional wound care (n = 1274) following primary TKA or THA. Again, the use of ciNPWT significantly reduced overall wound complication rate (9.3% vs. 12.8% for controls) as well as SSI rate (2.3% vs. 6.9% for controls). The overall wound complication rate for patients undergoing primary TKA or THA was lower than for patients undergoing revision arthroplasty [16]. Nevertheless, an equally significant protective effect of ciNPWT in patients undergoing primary arthroplasty was found (level I) [9]. Use of ciNPWT following primary THA or TKA was investigated in a single-centre RCT by Redfern et al. [17]. One hundred sixty-nine patients were treated with a ciNPWT system for 6–8 days after surgery, whilst 192 patients underwent conventional wound care. There was no significant difference in deep SSI rate between ciNPWT and control groups (1.0% vs. 1.25%; p = 0.810). However, the overall wound complication rate was significantly lower in cases where ciNPWT had been used (1.5% vs. 5.5%; p = 0.020). The authors concluded that ciNPWT may be applied following primary TKA or THA to improve wound healing (level II) [17]. A similar prospective RCT was performed by Keeney et al. in 2019 [18], analysing the effect of a ciNPWT device following primary or revision THA or TKA (n = 185) in comparison to standard wound dressings (n = 213). There was no significant difference in the rate of late deep or superficial SSI between ciNPWT-treated patients and controls (4.0% vs. 3.4%). However, obese patients (BMI >35 kg/m2) undergoing TKA were significantly less likely to develop any wound-related complications if ciNPWT had been administered (1.3% vs. 21.6%; p < 0.01). The authors concluded that ciNPWT may be effectively used following TKA, especially in those with high BMI (level II) [18]. In the RCT by Alga et al. [19], patients with conflict-related extremity wounds (n = 174) were randomly assigned to receive ciNPWT (n = 88) or conventional wound care (n = 86). At the fifth postoperative day, 49.0% (ciNPWT) and 60.0% (control) of wounds had healed (p = 0.212). There was no significant difference between the intervention and control groups regarding development of SSI (12.0% vs. 23.0%; p = 0.068). The authors concluded that ciNPWT had no significant benefit in acute conflict-related wounds (level II), and they even questioned the application of costly treatments in resource-limited conflict settings (level II) [19]. The most recent RCT was published by Costa et al. in 2020 [20], randomly assigning 1548 adult patients with major limb trauma in the Wound Healing in Surgery for Trauma (WHIST) trial to either ciNPWT (n = 785) or standard wound care (n = 763). The authors did not discover a statistically significant difference in rates of deep SSI between ciNPWT-treated patients and controls at 30 days (5.8% vs. 6.7%) and 90 days (11.4% vs. 13.2%). Furthermore, there were no significant differences between the two groups regarding secondary outcome measures such as self-assessment of surgical scar, presence of chronic neuropathic pain and patient-reported outcome scores [20]. They concluded that ciNPWT had no protective effect on the development of deep SSI in patients with lower limb fractures following major trauma (level II) [20].
General surgery
SSI developed in up to 45.0% of patients undergoing colorectal surgery [21]. Besides causing significant financial burden, SSI following colorectal surgery often led to prolonged hospital stay, negatively affected patients’ quality of life and increased the risk of developing incisional hernias [22].
Two systematic reviews and one meta-analysis were identified analysing the effect of ciNPWT following laparotomy for general surgical procedures, colorectal surgery and gynaecological procedures (Table 2). In the systematic review by Fowler et al. [23], four prospective RTCs and four retrospective studies were analysed to assess the potential effect of ciNPWT on wound healing following open abdominal surgery in comparison to standard dressing. Overall, 454 patients were treated with ciNPWT, with a standard Vacuum Assisted Closure (VAC) system in four studies and a ciNPWT system in another four studies [23]. The remaining 1016 patients received standard dressings. In eight of the nine studies investigated, a significant reduction in SSI was observed in the ciNPWT group in comparison to the control group. The only study not reporting a statistically significant difference was based on a retrospective analysis of 208 patients undergoing laparotomy for oncologic gynaecology resections, of whom 22 were treated with a VAC system [23, 24]. Across all studies, between 2.9 and 12.5% of patients in the intervention groups developed SSI, as compared to 7.2 to 44.0% of patients in the control group [23]. The authors concluded that ciNPWT was a promising measure to lower the risk of SSI in patients with abdominal surgery, although its impact on other wound complications remained unanswered (level I) [23]. In the systematic review and meta-analysis by Sahebally et al. [25], three RTCs, two prospective and three retrospective studies assessing the impact of ciNPWT on SSI following laparotomy for colorectal and general surgery were analysed. Altogether, 704 patients were treated with standard dressings, whilst 485 patients received ciNPWT following laparotomy. In four studies a standard VAC system was used, whilst in five studies a ciNPWT system was applied [25]. The rate of SSI ranged between 25.5 and 48.0% for the control group and 0.0 to 20.5% for the intervention group. On random-effects analysis, ciNPWT was associated with a significantly lower SSI rate in comparison to standard dressings (OR: 0.25; 95%CI: 0.12–0.52; p < 0.001). Other wound complications like seroma (OR: 0.38; 95%CI: 0.12–1.23; p = 0.100) or dehiscence (OR: 2.03; 95%CI: 0.61–6.78; p = 0.25) showed no significantly different occurrence between intervention and control groups. In the subgroup analysis including patients after colorectal surgery only, ciNPWT was significantly associated with a reduced risk of SSI (OR: 0.16; 95%CI: 0.07–0.36; p < 0.001). Therefore, the authors concluded that ciNPWT was associated with reduced rates of SSI following laparotomy for colorectal and general surgery, although the rate of other wound complications was not affected [25]. In the meta-analysis by Tran et al. [26], summarising results of two RCTs and nine retrospective studies with altogether 1723 patients undergoing abdominal wall reconstruction, the use of ciNPWT (n = 681) was associated with a lower SSI rate (15.0% vs. 28.0%; p < 0.001) and wound dehiscence rate (8.0% vs. 15.0%; p < 0.001) in comparison to patients treated by conventional wound care (n = 1042). However, no statistically significant differences with regard to seroma or haematoma formation, reoperation or readmission-rate were observed (level I) [26].
We identified seven meta-analyses analysing the impact of ciNPWT used for various surgical procedures. The meta-analysis by Hyldig et al. [27] of 10 RTCs included 664 patients in the intervention group and 647 patients in the control group. Standard VAC systems were used in 4 studies for ciNPWT and specifically designed ciNPWT systems in 6 studies. The overall SSI rate was 4.7% for the intervention group and 8.9% for the control group. The use of ciNPWT was associated with a significant reduction of SSI (relative risk [RR]: 0.54; 95%CI: 0.33–0.89) and seroma formation (RR: 0.48; 95%CI: 0.27–0.84), whilst there was no influence on development of wound dehiscence [27]. However, due to the heterogeneity of the studies included, analysing the effect of ciNPWT in trauma surgery, orthopaedic surgery, general surgery and cardiac surgery, the authors concluded that no general recommendations for the use of ciNPWT were possible (level I) [27]. In the same year, another meta-analysis was published by De Vries et al. [28], investigating six RCTs and 15 observational studies with 562 patients in the RTC and 4560 patients in the observational studies. Thirteen of the studies used a conventional (wound-specific) negative-pressure wound therapy (NPWT) system and eight studies a ciNPWT system [28]. SSI rates ranged between 1.3 and 20.4% for the ciNPWT group, and 2.8 and 53.1% for the wound NPWT group [28]. Overall, there was a significant reduction in SSI rates when ciNPWT was used, both for RCTs (OR: 0.56; 95%CI: 0.32–0.96; p = 0.040) and observational studies (OR: 0.30; 95%CI: 0.22–0.42; p < 0.0001). These results remained statistically significant when stratifying for clean and clean-contaminated procedures, as well as for different types of surgery. In trauma and orthopaedic procedures, however, this effect of ciNPWT vs. conventional dressings was no longer measurable [28]. With an overall low evidence according to the GRADE methodology, the prophylactic use of ciNPWT seemed to significantly reduce SSI rates (level I) [28]. In the third meta-analysis, 10 RCTs, three prospective studies and three retrospective studies were analysed with regards to ciNPWT for several surgical indications. Overall, the rate of SSI was significantly lower in patients treated with ciNPWT in comparison to controls (5.2% vs. 12.5%). Significantly fewer wound healing deficits were also observed in the ciNPWT-treated group as compared to controls (12.8% vs. 17.4%). Of note, although the reduction in SSI was present irrespective of the surgical procedure performed (e.g. orthopaedic, colorectal), the number needed to treat was lower in the case of surgical interventions with high complication rates [29]. Moreover, patients in the intervention group had a shorter hospital length of stay vs. controls. The authors concluded that ciNPWT was a viable option for various surgical indications to reduce the rate of wound healing deficits, SSI and hospital length of stay (level I) [29]. In three other meta-analyses, a positive influence of ciNPWT on postoperative wound healing, reducing the rate of SSI and, to a lesser extent, wound dehiscence and seroma formation, was confirmed (level I) [30,31,32]. Of note, in the meta-analysis by Singh et al., two different ciNPWT systems were compared, namely one using a foamed dressing and one using a multilayer absorbent dressing. Whilst the ciNPWT device with foamed dressing significantly reduced the rate of SSI, the other absorbent one was not associated with a significant reduction in SSI rate [33].
Plastic and reconstructive surgery
Plastic surgery was one of the early adopters of ciNPWT, lending its use for complication-prone wounds or surgical sites submitted to increased tensile forces. For example, a high rate of postoperative seroma can be observed in surgical cases on certain large flap donor sites [34, 35], or following lymph node dissection (e.g. for malignant melanoma) [36]. Body contouring procedures, especially in the massive weight loss patient, are also prone to developing wound complications or SSI [37]. Several retrospective and prospective publications that investigated the effects of ciNPWT in plastic surgery are summarized in Table 3.
Starting with the retrospective publications, Jorgensen et al. [38] applied ciNPWT in 14 of 55 malignant melanoma patients after inguinal lymph node dissection, the remainder underwent standard postoperative wound care. Seroma formation was significantly reduced in the ciNPWT group in comparison to the control group (28.6% vs. 90.3%; p < 0.001). A statistically non-significant reduced trend for developing SSI favoured ciNPWT (42.9% vs. 65.9%; p = 0.130), while no effect on lymphedema formation was observed (35.7% vs. 51.2%; p = 0.330). The intervention group was more cost-effective than the conventional wound care ($ 911.2 vs. $ 2542.7; p < 0.05; level IV) [38]. In a study by Tauber et al. [39], ciNPWT was applied after inguinal lymph node dissection for penile or urethral cancer in 8 patients, whilst 16 patients underwent conventional wound care. Patients in the intervention group had a significantly reduced length of hospital stay, less formation of lymphedema (0.0% vs. 46.0%), lymphocele (20.0% v. 62.0%) and persistent lymphorrhea (7.0% vs. 45.0%; level IV) [39]. In a case series of 8 patients after surgical excision of pathological scars, ciNPWT was applied for 8 days postoperatively [40]. In all but one patient, an overall good compliance and complication-free follow-up was noted (in one case the ciNPWT system had to be removed prematurely after one day due to insufficient vacuum sealing; level IV) [40]. In a study by Abatangelo et al. [41], five of 11 cases of post-bariatric abdominoplasty received postoperative ciNPWT, whilst the remaining patients underwent conventional wound care. The authors observed a faster healing rate (10.8 ± 5.0 days vs. 23.0 ± 7.0 days) and reduced “minor local complication rate” (0% vs. 80%) in the intervention vs. control groups, respectively. The Vancouver Scar Scale at 90 days also demonstrated better results in the intervention group. The observations led the authors to conclude that ciNPWT reduced the risk for developing minor wound complications and improved surgical outcome (level IV) [41]. The influence of ciNPWT following two-stage expander/implant-based postmastectomy reconstruction [42] revealed wound complications in three of 25 breasts of 13 patients (12.0%) and one flap necrosis, which required reoperation (4.0%). On 3‑month follow-up, 24 of the 25 breasts were closed without any further deviation (96.0%; level IV) [42]. Two years thereafter, the same set of authors published a larger retrospective case–control series on almost the same topic [43]. They observed closed incisions in 665 breasts from 356 patients, of which 177 (331 breasts) were treated by ciNPWT and 179 (334 breasts) by conventional wound care. The overall complication rate was lower in the ciNPWT group as compared to the control group (8.5% vs. 15.9%; p = 0.009), as was the rate of SSI (2.1% vs. 4.5%; p = 0.023), of wound dehiscence (2.4% vs. 5.4%; p = 0.018), of seroma formation (1.8% vs. 5.7%; p = 0.011), of tissue necrosis (5.1% vs. 9.3%; p = 0.007), of time to drain removal (9.9 days vs. 13.1 days; p < 0.001) and, last but not least, surgical revision rate (2.4% vs. 5.4%; p = 0.050). The authors concluded that ciNPWT may be used for reducing postoperative complications following postmastectomy breast reconstruction (level IV) [43].
This paragraph highlights the prospective publications in plastic surgery: in 2016, an RCT investigated conventional wound therapy (n = 50) vs. ciNPWT applied for 5 days (n = 50) after harvesting superficial circumflex iliac artery perforator flaps [44]. Significantly lower amounts of fluid were collected in ciNPWT wounds in comparison to controls (23.3 ± 18.4 ml vs. 100.5 ± 140.7 ml; p < 0.001), resulting in a significantly earlier removal of suction drains (3.3 ± 1.3 days vs. 6.1 ± 5.0 days; p = 0.008). Interestingly, an increase in skin blood perfusion at 5 days was observed in the ciNPWT group. Overall, one wound dehiscence developed in the control group vs. none in the intervention group (level II) [44]. An RCT in 2017 including 40 patients with latissimus dorsi flap donor sites for breast reconstruction [45] detected in 3 of 20 patients with ciNPWT a seroma formation (15.0%) vs. in 14 of 20 patients (70.0%) in the control group with conventional wound care (OR: 0.07). The mean number of percutaneous seroma releases and the totally aspirated volume was lower in the ciNPWT group as compared to the control group. No statistically significant difference for duration or total volume of wound drainage was found (level II) [45]. An RCT—(DEhiscence PREvention Study) (DEPRES)—after autologous breast reconstruction with a deep internal epigastric perforator (DIEP) or profunda artery perforator (PAP) flap [46] included a total of 51 female patients. Twenty-five patients were treated with ciNPWT and 26 patients with adhesive strips for managing the donor site. The ciNPWT group had less wound dehiscence vs. control (8.0% vs. 34.6%; p = 0.038; level II) [46]. ciNPWT [47] was also applied in an RCT for 7 days following oncological breast surgery and included 37 patients with 47 breasts. In the ciNPWT group (n = 17; 25 breasts), complications were seen in 4.0%, as compared to 45.0% in the control group (n = 20; 22 breasts, conventional post-surgical dressings; p = 0.001; level III) [47]. Furthermore, two RCTs have been published on the use of ciNPWT following bilateral breast reductions [48, 49]. In the smaller study, Tanaydin et al. [48] used an intraindividual comparison study design in 32 patients, where the ciNPWT device was used on one breast and conventional adhesive strips on the other. Overall, the wound complication rate was lower in the intervention group (15.6% vs. 31.3%; p < 0.004). Statistically significant as well as improved pain scores, scar quality and subjectively rated aesthetic appearance were also observed in the ciNPWT-treated breasts (level II) [48]. In the second, larger study by Galiano et al. [49], 200 patients undergoing bilateral breast reduction surgery were prospectively enrolled in a multicentre trial. With the same intraindividual comparison design, one breast received ciNPWT and the other standard wound care postoperatively. The 21-day assessment was completed in 185 patients. At that time, 56.8% of ciNPWT-treated breasts (n = 113) had developed wound healing complications, in comparison to 61.8% (n = 123) in controls (p = 0.004). Interestingly, the rate of SSI demonstrated no statistically significant difference (2.0% vs. 3.0%). The authors concluded that wound healing complications following bilateral breast reduction surgery can be reduced by applying ciNPWT (level II) [49]. The prospective series of Abesamis et al. in 2019 [50] analysed the use of ciNPWT after abdominoplasty in 16 female patients. Nine patients received an ciNPWT and 7 patients served as control (standard postoperative wound care). There was a statistically significant reduction of fluid drainage in the ciNPWT group vs. control (p < 0.0001), as there was for time to drain removal (5.3 ± 1.6 days vs. 10.6 ± 2.9 days; p < 0.001; level III) [50].
Cardiac surgery
Between 1 and 3% of patients risk developing SSI following cardiac surgery, with the rate strongly depending on the presence of obesity, diabetes, other peri- and postoperative complications, and coronary artery bypass grafting using both internal thoracic arteries [51]. Therefore, measurements to lower the risk of SSI, especially in high-risk multimorbid patients, are required. In this respect, ciNPWT in cardiac surgery has gained more attention over the past few years (Table 4).
Atkins et al. [52] published a retrospective study of 57 adult cardiac patients receiving ciNPWT dressing, who were at a high-risk of developing deep sternal wound infection and mediastinitis after sternotomy. They were compared to 213 patients with a low risk for sternal wound infection and who received standard gauze wound dressings. Treatment duration was 4 days. In the ciNPWT group, the authors observed no infected wound, while they “expected”—based on the preoperative risk factors—at least three wound infections. There was one wound infection in the control group of low-risk patients. This led the authors to conclude that ciNPWT reduced the incidence of sternal wound infections and dehiscence (level IV) [52]. Colli et al. [53] published a retrospective review of 10 high-risk patients for SSI following cardiac surgery. All patients received a ciNPWT system for 5 days and none of them subsequently developed an SSI. They concluded that ciNPWT systems were safe and easy to use and can be beneficial in healing of sternal wounds in high-risk patients (level IV) [53]. In the same year, Atkins et al. [54] published a study regarding peristernal perfusion evaluated by laser Doppler flowmetry in patients following myocardial revascularization via median sternotomy. In detail, the authors analysed the influence of left internal mammary artery (LIMA) harvest on postoperative peristernal perfusion and the potential impact of subsequently applied ciNPWT. Altogether, 20 patients were included in the study, of whom 10 received ciNPWT for 4 days (intervention group) and 10 a standard gauze dressing (control group) [54]. All patients underwent daily assessment of peristernal perfusion. Overall, there was no sternal wound infection. In the ciNPWT group, however, there was improved peristernal perfusion in comparison to the control group. LIMA harvest reduced peristernal perfusion rates by more than 25%, but a postoperatively applied ciNPWT device enhanced peristernal perfusion to 100%, thus actually compensating for the loss. Consequently, application of ciNPWT may improve peristernal perfusion in high-risk patients following median sternotomy, potentially reducing SSI (level IV) [54]. Grauhan et al. [55] published a prospective comparative study without randomization of 150 consecutive patients with BMI >30 who had undergone median sternotomy. Patients in the intervention group (n = 75) were treated with ciNPWT for 6–7 days, whilst patients in control group (n = 75) received standard gauze dressing. The primary endpoint was sternal wound infection developing within 90 days following surgery [55]. Sternal wound infection rate in the ciNPWT group was 4% vs. 16% in control group. Therefore, the authors concluded that application of ciNPWT decreases the postoperative wound infection rate in patients following median sternotomy (level III) [55]. The same authors published another study a year thereafter [56], prospectively evaluating 237 patients with median sternotomy and application of ciNPWT for 6–7 days. This group of patients was compared to a historic cohort of 3508 patients who had been treated with standard dressings [56]. Primary endpoint was sternal wound infection during the first 30 postoperative days. Patients in the ciNPWT group developed significantly fewer sternal wound infections (1.3% vs. 3.4%; p < 0.05; odds ratio: 2.74) in comparison to the control group, which is why the authors concluded that ciNPWT significantly reduces deep sternal wound infections following median sternotomy (level III) [56]. In the same year, the first randomized controlled trial was published by Witt-Majchrzak et al. [57], analysing 80 patients after sternotomy for open heart surgery. In the intervention group (n = 40), patients were treated with a ciNPWT system and a negative pressure of 80 mm Hg, whilst patients in the control group (n = 40) received standard dressings [57]. In this study, statistically fewer sternal wound infections were observed in patients with ciNPWT in comparison to patients in the control group (7.5% vs. 25.0%; p = 0.034; level II) [57]. Santarpino et al. [58] published a retrospective study on 129 patients undergoing myocardial revascularization with bilateral internal mammary artery harvest. In 21 of these patients, the authors applied ciNPWT for 5 days. The remaining patients received a standard gauze dressing [58]. Patients in the ciNPWT group were generally younger, the surgical priority less urgent and general risk for SSI lower, yielding a selection bias. In comparison to the control group with 5.6% sternal wound infections, the ciNPWT group had no infections, which was not statistically significant (p = 0.336). Sternal dehiscence rate was also lower (0.0% vs. 3.7%), but statistically not significant (p = 0.487). Of note, one patient in the ciNPWT group required premature removal of the ciNPWT due to bleeding (level IV) [58]. In 2016, Reddy et al. [59] published a retrospective review on 27 high-risk patients with mixed cardiac pathology receiving ciNPWT. The majority (78%) of these patients had intact incisions showing good re-approximation of the skin 30 days following surgery. Six of the 27 patients developed wound healing problems (22.2%), of whom one required re-admission to hospital for intravenous antibiotics (level IV) [59]. Similarly, Jennings et al. [60] published a retrospective study of 62 patients treated with ciNPWT after median sternotomy. They divided patients into a high- and a low-risk group for SSI and compared actual infection rates to predicted infection rates, according to the Fowler score [61]. Significantly lower infection rates were observed in both cohorts, with only one patient developing sternal wound infection. The authors concluded that ciNPWT should be considered in patients at a high risk for sternal wound infection, like in diabetics, elderly and obese patients (level IV) [60]. Phillip et al. [62] published a retrospective review of 10 patients after median sternotomy with mixed cardiac pathology. All incisions healed without complications. They suggested that the use of ciNPWT should be considered for sternotomy patients with comorbidities increasing the risk of SSI (level IV). More recently, Gatti et al. [63] published a prospective study of 53 high-risk patients undergoing coronary artery bypass surgery using both internal mammary arteries. They compared the actual rate of deep sternal wound infections to the predicted rate using two different scoring systems. The authors observed lower infection rates than predicted from the Gatti score [64] (3.4% vs. predicted 5.8%) and slightly higher rates than predicted by the Fowler score (3.4% vs. predicted 2.3%) [61]. The authors stated that application of ciNPWT may reduce the risk of deep sternal wound infections following bilateral internal mammary artery harvest (level III) [63]. Furthermore, Ruggieri et al. [65] published a prospective non-randomized study on patients undergoing isolated CABG using BIMA grafts, where ciNPWT was used in 161 patients and conventional sterile wound dressing was used in 266 patients. Propensity score matching was performed to allow statistical “pseudo-randomisation”, resulting in 128 matched pairs with similar characteristics. The rate of SSI was similar in both groups, whereas the rate of deep SSI and mediastinitis was lower in the ciNPWT group as compared to the control group, without reaching statistical significance (level III).
Vascular surgery
Following vascular surgery, superficial SSIs were predicted to develop in up to 76.0% of patients, deep SSIs in up to 24.0% and SSIs with graft exposure were observed in up to 6.1% of patients [66]. In these often multimorbid patients requiring vascular procedures, reduction of SSI risk is needed to prevent consequences such as prolonged hospital length of stay, ICU admission, increased morbidity and mortality [67]. Different retrospective and prospective studies have been published in recent years, investigating the potential positive effect of ciNPWT on postoperative outcomes of patients undergoing vascular surgery (Table 5).
Matatov et al. [68] retrospectively analysed 90 patients with altogether 115 femoral cutdown procedures for vascular surgery. Patients were divided into two groups according to the type of wound dressing (ciNPWT: n = 52; standard wound dressing: n = 63). Wounds were evaluated 7 and 30 days after surgery and the Szilagy classification used to assess grade of SSI [69]. The ciNPWT group developed significantly fewer groin infections in comparison to the control group (30.0% vs. 6.0%). The authors concluded that ciNPWT significantly decreased the incidence of groin wound infections (level IV) [68]. Weir et al. [70] performed a prospective intraindividual case–control study in 8 patients with bilateral groin incisions. ciNPWT was placed on one side and standard dressing on the other side. No significant wound complications occurred in cases where ciNPWT was used (one small superficial haematoma, one small superficial wound necrosis, both treated conservatively), as compared to three significant complications in wounds treated with standard dressings (two seromas and one deep wound necrosis, all of them requiring revision surgery) [70]. A potential reduction in wound complications without an increase in haemorrhage rate was observed in high-risk patients undergoing vascular surgery. This article, however, was based on preliminary results of an ongoing study. Therefore, the authors stated that more investigation was required to prove this observation (level III) [70].
Furthermore, we found five randomised controlled trials and one meta-analysis published in the literature analysing the use of ciNPWT in vascular surgery. Lee K et al. [71] performed a single-centre randomised controlled trial including 192 high-risk patients for SSI who underwent surgical exposure of the femoral artery for different vascular procedures. Surgical wounds were closed primarily and covered either with standard gauze dressings (n = 49) or ciNPWT (n = 53). Primary outcome was the 30-day SSI incidence in the groin wound. Lower but non-significant wound infection rates in the ciNPWT group were observed in comparison to the control group (11% vs. 19%, p = 0.240) [71]. However, there was a significantly shorter length of stay (6.4 vs. 8.9 days, p = 0.01) in the ciNPWT group (level II) [71]. Pleger et al. [72] reported results of a prospective randomized single-centre study. They analysed 100 patients with 129 groin incisions for vascular procedures. Patients were divided into two groups according to the wound dressing used. The intervention group received a ciNPWT system (n = 58) and the control group (n = 71) conventional adhesive dressings. The wounds were followed up for 30 days. There were significantly fewer wound healing complications in the ciNPWT group than in the control group (8.6% vs. 4.3%; p < 0.005) [72]. The same results were reported for subgroups evaluated at 5 to 7 days (0.0% vs. 21.1%; p < 0.005) and 30 days (8.6% vs. 21.1%; p = 0.230) postoperatively, and for revision surgeries within the first 30 postoperative days (1.7% vs. 14.1%; p = 0.022). The authors concluded that ciNPWT significantly reduced the incidence of SSI and revision procedures after vascular surgery (level II) [72]. Engelhardt et al. [73] investigated 132 consecutive patients who received longitudinal femoral incisions for different vascular procedures. Patients were randomized into two groups: in the intervention group (n = 64), ciNPWT was applied for 5 days, and in the control group (n = 68), conventional absorbent dressing was used [73]. Wounds were evaluated after 5 and 42 days. The infection rate was lower in the ciNPWT group as compared to the control group without reaching statistical power (14.0% vs. 28.0%; p = 0.055). The same trend was observed for early infections (6.0% vs. 15.0%; p = 0.125). Although the authors saw encouraging effects with ciNPWT, the study failed to demonstrate significant efficacy to reduce groin infections (level II) [73]. Gombert et al. [74] reported results of the AIMS trial. This was a randomized prospective trial evaluating 188 patients of two study centres who had longitudinal femoral incisions for vascular procedures. 45% of these procedures were reoperations. There were significantly less SSI in the intervention group with ciNPWT (n = 98) as compared to the control group (n = 90) using standard wound dressing (13.2% vs. 33.3%; p = 0.002) [74]. This RCT confirmed a benefit of ciNPWT devices in reducing the occurrence of groin infections in vascular surgery (level II) [74]. Kwon et al. [75] published a prospective randomized controlled single-institution trial on 123 groin incisions in high-risk patients for various vascular procedures. Patients were randomized into two groups depending on the wound dressing used, being either a standard gauze dressing in the control group (n = 60) or a ciNPWT device in the intervention group (n = 59). There was another group of patients at a low risk for SSI (n = 21) who were not randomized but served as an additional low-risk control. The primary endpoint was the incidence of a major groin complication within 30 days. The low-risk group had a 4.8% wound complication rate. There was a significant increase in the infection rate in the high-risk control group as compared to the ciNPWT group (25.0% vs. 8.5%; p < 0.001) [75]. Patients in the high-risk control group also had higher reoperation (18.3% vs. 8.5%; p < 0.05) and readmission rates (16.7% vs. 6.8%; p < 0.05). However, there was no significant difference in the length of stay (10.6 days vs. 10.6 days) or average costs associated with the treatment assignment ($ 36,537 vs. $ 30,492; p = 0.11) between the patient groups. This study supported the use of ciNPWT in vascular surgery, especially in patients at a high risk for SSI (level II) [75]. Svensson-Björk et al. [76] published a meta-analysis on ciNPWT use in groin incisions for arterial surgery, including seven RCTs with 1049 incisions (intervention group: n = 512; control group: n = 537). According to their analysis, a significant reduction in SSI with ciNPWT (OR: 0.35; 95%CI: 0.24–0.50; p < 0.001) was observed in comparison to the control group. The heterogeneity between the included studies was low and the quality of evidence was moderate. In a subgroup analysis of 363 incisions from three studies of patients with lower limb revascularization, a similar reduction in SSI (OR: 0.37; 95%CI: 0.22–0.63; p < 0.001) could be observed [76]. The authors concluded that ciNPWT use in groin incisions after arterial procedures significantly reduced the incidence of SSI as compared to standard wound dressings. However, they also stressed the need for more high-quality RCTs with cost-effectiveness analyses (level I) [76]. Benrashid et al. [77] published a review of prospectively collected data in 504 patients, of whom 225 were treated with ciNPWT. Patients had mixed vascular procedures with infrainguinal incisions. They observed significantly fewer SSI with ciNPWT (9.8% vs. 19.0% with standard dressings; p < 0.01) along with decreased perioperative mortality (5.8% vs 11.2%; p = 0.04). They concluded that the use of ciNPWT devices decreased the incidence of infrainguinal wound infections (level III). This observation could be confirmed in the Inguinal Vascular Surgical Wound Protection by Incisional Negative Pressure Wound Therapy (INVIPS) prospective randomised study by Hasselman et al. [78], investigating the use of ciNPWT following inguinal incisions for vascular surgery. A lower incidence of SSI in the ciNPWT group compared to the control group was observed (level II; Table 5). In line with this, Antoniou et al. [79] published a meta-analysis of six randomized controlled trials reporting on 733 groin incisions, discovering that patients with ciNPWT had a lower risk of SSI and revision surgery, and shorter hospital stays (level I). Wee et al. [80] published a meta-analysis of five randomized controlled trials including 662 patients. The overall risk of infection was lower for the ciNPWT group. Specifically, the risk of Szilagyi grade I and III infections was significantly reduced in the ciNPWT group (level I), whilst there was no significant difference with regards to Szilagyi grade II infections. Similarly, Sexton et al. [81] published a meta-analysis of seven RCTs, concluding that ciNPWT may reduce the incidence of groin wound complications following vascular surgery (level I). According to the meta-analysis comprising six RCTs by Gombert et al. [82], ciNPWT use was associated with a significant positive effect on postoperative wound healing (I).
Great saphenous vein (GSV) harvests have been one of the most common complication-prone interventions for coronary artery bypass grafting and contributed significantly to postoperative patient morbidity. For this review only one published feasibility study by Lee AJ et al. [83]—a single-centre randomized controlled trial with 64 patients—was found. Primary endpoint was feasibility of ciNPWT on GSV harvest sites in 35 patients (intervention group), potential complications and rate of system failure. Secondary endpoints were SSI rates, leg complications, hospital length of stay and quality of life at 6 weeks post-surgery in the intervention group in comparison to the control group (n = 29) [83]. The authors observed no serious complications associated with ciNPWT, except for the device requiring discontinuation in two cases and developing an allergic contact dermatitis in one. In comparison to the control group, ciNPWT patients had an earlier date of discharge (6 vs. 10 days, p = 0.008), increased ability for self-care (p = 0.023) and quality of life (p = 0.039) at initial assessment, and increased mobility at initial and follow-up assessment (p = 0.0117 and 0.012) [83]. The authors concluded that ciNPWT was safe to use, improved postoperative recovery and had a prolonged impact on mobility at 6 weeks post-surgery [83]. Nevertheless, they also pointed out that larger studies were needed to assess ciNPWT effects in patients with GSV harvest (level II) [83].
Discussion
Published literature on the use of ciNPWT for various surgical indications expresses positive views with regard to clinical experiences. Improved postoperative wound healing, reduced rate of SSI and shortened hospital length of stay were among the reported benefits. 42 of 59 analysed studies (71.2%) discovered a statistically significant beneficial effect of ciNPWT application for incision management in comparison to conventional wound care, 10.2% failed to identify any differences (n = 6) and in 18.6% (n = 11) results were inconclusive. Depending on the clinical speciality, the number of publications and associated level of evidence (i.e. I or II) varied widely (Fig. 2). In cardiac surgery, the majority had an either retrospective or non-randomised prospective design, whereas several RCTs on this topic in vascular or plastic surgery have been published. The focus of studies for ciNPWT application was centred on specific surgical procedures involving high-risk patients with distinct comorbidities. Patients undergoing vascular surgery were by far more likely to present with multiple comorbidities as compared to patients undergoing primary arthroplasty or even a body lift procedure. It seemed as if a “high-risk collective” (e.g. for developing wound complications) experienced benefits from ciNPWT to a greater extent than a “low-risk collective”. This needs to consider the selection bias of all studies, since the application of a ciNPWT system would have required a huge number to treat in the case of “complication-free” or “less complication-prone” interventions. Consequently, none of the reviewed studies focused on this. The cited favourable results for ciNPWT application may, as a result, not necessarily be extrapolated to other surgical interventions. The reviewed reports also contained company-funded research (e.g. by provision of the analysed ciNPWT systems), and we cannot rule out potential conflicts of interest (COI) possibly affecting study results. In fact, 35.6% of studies (n = 21) disclosed COI or financial support (FS) related to their study, whilst none was disclosed in 52.5% (n = 31). No COI-/FS-related information was available in 11.9% (n = 7).
Conclusion and recommendations
Recommendations for indications of closed-incision negative-pressure wound therapy (ciNPWT) in the different surgical specialties, their level of evidence and outcomes are listed in Table 6. Given the focus of this review, patients with complication-prone interventions and/or presurgical risk factors can benefit from ciNPWT, whereas this treatment modality—also in the absence of statistical models to prove this—could not be scientifically proven to outbalance them. Surgical incisions with anticipated increased postoperative tension on wound edges and complicated wound regions most likely benefit from ciNPWT, since it “buys the wounds/incisions some time to stabilise” before stretching and tensile forces act again upon removal of the systems. In the light of the current evidence, we rather refrain from listing any surgical procedures suitable for ciNPWT, but encourage future application settings to be tried and tested by identifying suitable patients benefitting from this therapy. Each case should be planned individually, taking together all intrinsic and extrinsic risk factors prior to application of ciNPWT. Three components for future analysis (and current lack thereof) were revealed by this review: 1) non-inferiority study designs comparing traditional negative-pressurewound therapy (NPWT) systems vs. ciNPWT, 2) cost–benefit calculations for healthcare providers and system specific-outcome markers and 3) quality research (up to RCT level) with unique outcome measures like the Clavien–Dindo classification (complication grading based on intervention type for complications, and not on clinical assessment alone) and patient-reported outcome measures (PROMS; e.g. evaluating patient experience in relation to the use of the system) [84].
We encourage future research to address these points.
References
Ban KA, Minei JP, Laronga C, Harbrecht BG, Jensen EH, Fry DE, et al. American College of Surgeons and Surgical Infection Society: Surgical Site Infection Guidelines, 2016 Update. J Am Coll Surg. 2017;224(1):59–74.
Anderson DJ, Podgorny K, Berrios-Torres SI, Bratzler DW, Dellinger EP, Greene L, et al. Strategies to prevent surgical site infections in acute care hospitals: 2014 update. Infect Control Hosp Epidemiol. 2014;35(Suppl 2):S66–S88.
Nam D, Sershon RA, Levine BR, Della Valle CJ. The use of closed incision negative-pressure wound therapy in orthopaedic surgery. J Am Acad Orthop Surg. 2018;26(9):295–302.
Cooper HJ, Bas MA. Closed-incision negative-pressure therapy versus antimicrobial dressings after revision hip and knee surgery: a comparative study. J Arthroplasty. 2016;31(5):1047–52.
Krug E, Berg L, Lee C, Hudson D, Birke-Sorensen H, Depoorter M, et al. Evidence-based recommendations for the use of negative pressure wound therapy in traumatic wounds and reconstructive surgery: steps towards an international consensus. Injury. 2011;42(Suppl 1):S1–S12.
Kilpadi DV, Lessing C, Derrick K. Healed porcine incisions previously treated with a surgical incision management system: mechanical, histomorphometric, and gene expression properties. Aesthetic Plast Surg. 2014;38(4):767–78.
Group OLoEW. The Oxford 2011 Levels of Evidence. Oxford: Oxford Centre for Evidence-Based Medicine; 2011.
Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780–5.
Kim JH, Kim HJ, Lee DH. Comparison of the efficacy between closed Incisional negative-pressure wound therapy and conventional wound management after total hip and knee arthroplasties: a systematic review and meta-analysis. J Arthroplasty. 2019;34(11):2804–14.
Koval KJ, Helfet DL. Tibial plateau fractures: evaluation and treatment. J Am Acad Orthop Surg. 1995;3(2):86–94.
Teeny SM, Wiss DA. Open reduction and internal fixation of tibial plafond fractures. Variables contributing to poor results and complications. Clin Orthop Relat Res. 1993;292:108–17.
Achten J, Vadher K, Bruce J, Nanchahal J, Spoors L, Masters JP, et al. Standard wound management versus negative-pressure wound therapy in the treatment of adult patients having surgical incisions for major trauma to the lower limb—a two-arm parallel group superiority randomised controlled trial: protocol for Wound Healing in Surgery for Trauma (WHIST). BMJ Open. 2018;8(6):e22115.
Cooper HJ, Roc GC, Bas MA, Berliner ZP, Hepinstall MS, Rodriguez JA, et al. Closed incision negative pressure therapy decreases complications after periprosthetic fracture surgery around the hip and knee. Injury. 2018;49(2):386–91.
Stenqvist CP, Nielsen CT, Napolitano GM, Larsen BM, Flies MJ, Brander DC, et al. Does closed incision negative wound pressure therapy in non-traumatic major lower-extremity amputations improve survival rates? Int Wound J. 2019;16(5):1171–7.
Zayan NE, West JM, Schulz SA, Jordan SW, Valerio IL. Incisional negative pressure wound therapy: an effective tool for major limb amputation and amputation revision site closure. Adv Wound Care. 2019;8(8):368–73.
Newman JM, Siqueira MBP, Klika AK, Molloy RM, Barsoum WK, Higuera CA. Use of closed Incisional negative pressure wound therapy after revision total hip and knee arthroplasty in patients at high risk for infection: a prospective, randomized clinical trial. J Arthroplasty. 2019;34(3):554–9. e1.
Redfern RE, Cameron-Ruetz C, O’Drobinak SK, Chen JT, Beer KJ. Closed incision negative pressure therapy effects on postoperative infection and surgical site complication after total hip and knee arthroplasty. J Arthroplasty. 2017;32(11):3333–9.
Keeney JA, Cook JL, Clawson SW, Aggarwal A, Stannard JP. Incisional negative pressure wound therapy devices improve short-term wound complications, but not long-term infection rate following hip and knee arthroplasty. J Arthroplasty. 2019;34(4):723–8.
Alga A, Haweizy R, Bashaireh K, Wong S, Lundgren KC, von Schreeb J, et al. Negative pressure wound therapy versus standard treatment in patients with acute conflict-related extremity wounds: a pragmatic, multisite, randomised controlled trial. Lancet Glob Health. 2020;8(3):e423–e9.
Costa ML, Achten J, Knight R, Bruce J, Dutton SJ, Madan J, et al. Effect of Incisional negative pressure wound therapy vs standard wound dressing on deep surgical site infection after surgery for lower limb fractures associated with major trauma: the WHIST randomized clinical trial. JAMA. 2020;323(6):519–26.
Wick EC, Vogel JD, Church JM, Remzi F, Fazio VW. Surgical site infections in a “high outlier” institution: Are colorectal surgeons to blame? Dis Colon Rectum. 2009;52(3):374–9.
Gheorghe A, Moran G, Duffy H, Roberts T, Pinkney T, Calvert M. Health utility values associated with surgical site infection: a systematic review. Value Health. 2015;18(8):1126–37.
Fowler AL, Barry MK. Closed incision negative pressure therapy: results of recent trials and recommendations for clinical practice. Surgeon. 2019;18(4):241–50. https://doi.org/10.1016/j.surge.2019.10.007.
Lynam S, Mark KS, Temkin SM. Primary placement of Incisional negative pressure wound therapy at time of laparotomy for gynecologic malignancies. Int J Gynecol Cancer. 2016;26(8):1525–9.
Sahebally SM, McKevitt K, Stephens I, Fitzpatrick F, Deasy J, Burke JP, et al. Negative pressure wound therapy for closed laparotomy incisions in general and colorectal surgery: a systematic review and meta-analysis. JAMA Surg. 2018;153(11):e183467.
Tran BNN, Johnson AR, Shen C, Lee BT, Lee ES. Closed-incision negative-pressure therapy efficacy in abdominal wall reconstruction in high-risk patients: a meta-analysis. J Surg Res. 2019;241:63–71.
Hyldig N, Birke-Sorensen H, Kruse M, Vinter C, Joergensen JS, Sorensen JA, et al. Meta-analysis of negative-pressure wound therapy for closed surgical incisions. Br J Surg. 2016;103(5):477–86.
De Vries FE, Wallert ED, Solomkin JS, Allegranzi B, Egger M, Dellinger EP, et al. A systematic review and meta-analysis including GRADE qualification of the risk of surgical site infections after prophylactic negative pressure wound therapy compared with conventional dressings in clean and contaminated surgery. Medicine. 2016;95(36):e4673.
Strugala V, Martin R. meta-analysis of comparative trials evaluating a prophylactic single-use negative pressure wound therapy system for the prevention of surgical site complications. Surg Infect. 2017;18(7):810–9.
Zwanenburg PR, Tol BT, Obdeijn MC, Lapid O, Gans SL, Boermeester MA. Meta-analysis, meta-regression, and GRADE assessment of randomized and nonrandomized studies of incisional negative pressure wound therapy versus control dressings for the prevention of postoperative wound complications. Ann Surg. 2020;272(1):81–91. https://doi.org/10.1097/SLA.0000000000003644.
Shiroky J, Lillie E, Muaddi H, Sevigny M, Choi WJ, Karanicolas PJ. The impact of negative pressure wound therapy for closed surgical incisions on surgical site infection: a systematic review and meta-analysis. Surgery. 2020;167(6):1001–9. https://doi.org/10.1016/j.surg.2020.01.018.
Singh DP, Gabriel A, Parvizi J, Gardner MJ, D’Agostino R Jr.. Meta-analysis of comparative trials evaluating a single-use closed-incision negative-pressure therapy system. Plast Reconstr Surg. 2019;143(1S):41S–6S.
Singh DP, Gabriel A, Silverman RP, Griffin LP, McGowan LD, D’Agostino RB Jr.. Meta-analysis comparing outcomes of two different negative pressure therapy systems in closed incision management. Plast Reconstr Surg Glob Open. 2019;7(6):e2259.
Menke H, Erkens M, Olbrisch RR. Evolving concepts in breast reconstruction with latissimus dorsi flaps: results and follow-up of 121 consecutive patients. Ann Plast Surg. 2001;47(2):107–14.
Daltrey I, Thomson H, Hussien M, Krishna K, Rayter Z, Winters ZE. Randomized clinical trial of the effect of quilting latissimus dorsi flap donor site on seroma formation. Br J Surg. 2006;93(7):825–30.
Chang SB, Askew RL, Xing Y, Weaver S, Gershenwald JE, Lee JE, et al. Prospective assessment of postoperative complications and associated costs following inguinal lymph node dissection (ILND) in melanoma patients. Ann Surg Oncol. 2010;17(10):2764–72.
Winocour J, Gupta V, Ramirez JR, Shack RB, Grotting JC, Higdon KK. Abdominoplasty: risk factors, complication rates, and safety of combined procedures. Plast Reconstr Surg. 2015;136(5):597e–606e.
Jorgensen MG, Toyserkani NM, Thomsen JB, Sorensen JA. Prophylactic incisional negative pressure wound therapy shows promising results in prevention of wound complications following inguinal lymph node dissection for melanoma: a retrospective case-control series. J Plast Reconstr Aesthet Surg. 2019;72(7):1178–83.
Tauber R, Schmid S, Horn T, Thalgott M, Heck M, Haller B, et al. Inguinal lymph node dissection: epidermal vacuum therapy for prevention of wound complications. J Plast Reconstr Aesthet Surg. 2013;66(3):390–6.
Bollero D, Malvasio V, Catalano F, Stella M. Negative pressure surgical management after pathological scar surgical excision: a first report. Int Wound J. 2015;12(1):17–21.
Abatangelo S, Saporiti E, Giatsidis G. Closed incision negative-pressure therapy (ciNPT) reduces minor local complications in post-bariatric abdominoplasty body contouring: a retrospective case-control series. OBES SURG. 2018;28(7):2096–104.
Gabriel A, Sigalove SR, Maxwell GP. Initial experience using closed incision negative pressure therapy after immediate postmastectomy breast reconstruction. Plast Reconstr Surg Glob Open. 2016;4(7):e819.
Gabriel A, Sigalove S, Sigalove N, Storm-Dickerson T, Rice J, Maxwell P, et al. The impact of closed incision negative pressure therapy on postoperative breast reconstruction outcomes. Plast Reconstr Surg Glob Open. 2018;6(8):e1880.
Peter Suh HS, Hong JP. Effects of incisional negative-pressure wound therapy on primary closed defects after superficial circumflex Iliac artery perforator flap harvest: randomized controlled study. Plast Reconstr Surg. 2016;138(6):1333–40.
Angspatt A, Laopiyasakul T, Pungrasmi P, Suwajo P. The role of negative-pressure wound therapy in latissimus dorsi flap donor site seroma prevention: a cohort study. Arch Plast Surg. 2017;44(4):308–12.
Muller-Sloof E, de Laat HEW, Hummelink SLM, Peters JWB, Ulrich DJO. The effect of postoperative closed incision negative pressure therapy on the incidence of donor site wound dehiscence in breast reconstruction patients: DEhiscence PREvention Study (DEPRES), pilot randomized controlled trial. J Tissue Viability. 2018;27(4):262–6.
Ferrando PM, Ala A, Bussone R, Bergamasco L, Actis Perinetti F, Malan F. Closed incision negative pressure therapy in oncological breast surgery: comparison with standard care dressings. Plast Reconstr Surg Glob Open. 2018;6(6):e1732.
Tanaydin V, Beugels J, Andriessen A, Sawor JH, van der Hulst R. Randomized controlled study comparing disposable negative-pressure wound therapy with standard care in bilateral breast reduction mammoplasty evaluating surgical site complications and scar quality. Aesthetic Plast Surg. 2018;42(4):927–35.
Galiano RD, Hudson D, Shin J, van der Hulst R, Tanaydin V, Djohan R, et al. Incisional negative pressure wound therapy for prevention of wound healing complications following reduction mammaplasty. Plast Reconstr Surg Glob Open. 2018;6(1):e1560.
Abesamis GM, Chopra S, Vickery K, Deva AK. A comparative trial of incisional negative-pressure wound therapy in abdominoplasty. Plast Reconstr Surg Glob Open. 2019;7(5):e2141.
Lemaignen A, Birgand G, Ghodhbane W, Alkhoder S, Lolom I, Belorgey S, et al. Sternal wound infection after cardiac surgery: incidence and risk factors according to clinical presentation. Clin Microbiol Infect. 2015;21(7):674. e11–e18.
Atkins BZ, Wooten MK, Kistler J, Hurley K, Hughes GC, Wolfe WG. Does negative pressure wound therapy have a role in preventing poststernotomy wound complications? Surg Innov. 2009;16(2):140–6.
Colli A, Camara ML. First experience with a new negative pressure incision management system on surgical incisions after cardiac surgery in high risk patients. J Cardiothorac Surg. 2011;6:160.
Atkins BZ, Tetterton JK, Petersen RP, Hurley K, Wolfe WG. Laser Doppler flowmetry assessment of peristernal perfusion after cardiac surgery: beneficial effect of negative pressure therapy. Int Wound J. 2011;8(1):56–62.
Grauhan O, Navasardyan A, Hofmann M, Muller P, Stein J, Hetzer R. Prevention of poststernotomy wound infections in obese patients by negative pressure wound therapy. J Thorac Cardiovasc Surg. 2013;145(5):1387–92.
Grauhan O, Navasardyan A, Tutkun B, Hennig F, Muller P, Hummel M, et al. Effect of surgical incision management on wound infections in a poststernotomy patient population. Int Wound J. 2014;11(Suppl 1):6–9.
Witt-Majchrzak A, Zelazny P, Snarska J. Preliminary outcome of treatment of postoperative primarily closed sternotomy wounds treated using negative pressure wound therapy. Pol Przegl Chir. 2015;86(10):456–65.
Santarpino G, Gazdag L, Sirch J, Vogt F, Ledwon M, Fischlein T, et al. A Retrospective Study to Evaluate Use of Negative Pressure Wound Therapy in Patients Undergoing Bilateral Internal Thoracic Artery Grafting. Ostomy Wound Manage. 2015;61(12):26–30.
Reddy VS. Use of closed incision management with negative pressure therapy for complex cardiac patients. Cureus. 2016;8(2):e506.
Jennings S, Vahaviolos J, Chan J, Worthington MG, Stuklis RG. Prevention of sternal wound infections by use of a surgical incision management system: first reported Australian case series. Heart Lung Circ. 2016;25(1):89–93.
Fowler VG Jr., O’Brien SM, Muhlbaier LH, Corey GR, Ferguson TB, Peterson ED. Clinical predictors of major infections after cardiac surgery. Circulation. 2005;112(9 Suppl):I-358–I-65.
Philip B, McCluskey P, Hinchion J. Experience using closed incision negative pressure wound therapy in sternotomy patients. J Wound Care. 2017;26(8):491–5.
Gatti G, Ledwon M, Gazdag L, Cuomo F, Pappalardo A, Fischlein T, et al. Management of closed sternal incision after bilateral internal thoracic artery grafting with a single-use negative pressure system. Updates Surg. 2018;70(4):545–52.
Gatti G, Dell’Angela L, Barbati G, Benussi B, Forti G, Gabrielli M, et al. A predictive scoring system for deep sternal wound infection after bilateral internal thoracic artery grafting. Eur J Cardiothorac Surg. 2016;49(3):910–7.
Ruggieri VG, Olivier ME, Aludaat C, Rosato S, Marticho P, Saade YA, et al. Negative pressure versus conventional sternal wound dressing in coronary surgery using bilateral internal mammary artery grafts. Heart Surg Forum. 2019;22(2):E92–E6.
Acosta S, Bjorck M, Wanhainen A. Negative-pressure wound therapy for prevention and treatment of surgical-site infections after vascular surgery. Br J Surg. 2017;104(2):e75–e84.
Coomer NM, Kandilov AM. Impact of hospital-acquired conditions on financial liabilities for Medicare patients. Am J Infect Control. 2016;44(11):1326–34.
Matatov T, Reddy KN, Doucet LD, Zhao CX, Zhang WW. Experience with a new negative pressure incision management system in prevention of groin wound infection in vascular surgery patients. J Vasc Surg. 2013;57(3):791–5.
Szilagyi DE, Smith RF, Elliott JP, Vrandecic MP. Infection in arterial reconstruction with synthetic grafts. Ann Surg. 1972;176(3):321–33.
Weir G. The use of a surgical incision management system on vascular surgery incisions: a pilot study. Int Wound J. 2014;11(Suppl 1):10–2.
Lee K, Murphy PB, Ingves MV, Duncan A, DeRose G, Dubois L, et al. Randomized clinical trial of negative pressure wound therapy for high-risk groin wounds in lower extremity revascularization. J Vasc Surg. 2017;66(6):1814–9.
Pleger SP, Nink N, Elzien M, Kunold A, Koshty A, Boning A. Reduction of groin wound complications in vascular surgery patients using closed incision negative pressure therapy (ciNPT): a prospective, randomised, single-institution study. Int Wound J. 2018;15(1):75–83.
Engelhardt M, Rashad NA, Willy C, Muller C, Bauer C, Debus S, et al. Closed-incision negative pressure therapy to reduce groin wound infections in vascular surgery: a randomised controlled trial. Int Wound J. 2018;15(3):327–32.
Gombert A, Babilon M, Barbati ME, Keszei A, von Trotha KT, Jalaie H, et al. Closed incision negative pressure therapy reduces surgical site infections in vascular surgery: a prospective randomised trial (AIMS trial). Eur J Vasc Endovasc Surg. 2018;56(3):442–8.
Kwon J, Staley C, McCullough M, Goss S, Arosemena M, Abai B, et al. A randomized clinical trial evaluating negative pressure therapy to decrease vascular groin incision complications. J Vasc Surg. 2018;68(6):1744–52.
Svensson-Bjork R, Zarrouk M, Asciutto G, Hasselmann J, Acosta S. Meta-analysis of negative pressure wound therapy of closed groin incisions in arterial surgery. Br J Surg. 2019;106(4):310–8.
Benrashid E, Youngwirth LM, Guest K, Cox MW, Shortell CK, Dillavou ED. Negative pressure wound therapy reduces surgical site infections. J Vasc Surg. 2020;71(3):896–904.
Hasselmann J, Bjork J, Svensson-Bjork R, Acosta S. Inguinal vascular surgical wound protection by Incisional negative pressure wound therapy: a randomized controlled trial-INVIPS trial. Ann Surg. 2020;271(1):48–53.
Antoniou GA, Onwuka CC, Antoniou SA, Russell D. Meta-analysis and trial sequential analysis of prophylactic negative pressure therapy for groin wounds in vascular surgery. J Vasc Surg. 2019;70(5):1700–10. e6.
Wee IJY, Syn N, Choong A. Closed incision negative pressure wound therapy in vascular surgery: a systematic review and meta-analysis. Eur J Vasc Endovasc Surg. 2019;58(3):446–54.
Sexton F, Healy D, Keelan S, Alazzawi M, Naughton P. A systematic review and meta-analysis comparing the effectiveness of negative-pressure wound therapy to standard therapy in the prevention of complications after vascular surgery. Int J Surg. 2020;76:94–100.
Gombert A, Van Vuuren T, Barbati ME, Toonder IM, Van Laanen JHH, Wittens C, et al. Closed incisional negative pressure therapy may reduce surgical site infection rate following endophlebectomy with complementary polytetrafluoroethylene arteriovenous fistula of the common femoral vein. J Vasc Surg Venous Lymphat Disord. 2020;8(1):89–94.
Lee AJ, Sheppard CE, Kent WD, Mewhort H, Sikdar KC, Fedak PW. Safety and efficacy of prophylactic negative pressure wound therapy following open saphenous vein harvest in cardiac surgery: a feasibility study. Interact Cardiovasc Thorac Surg. 2017;24(3):324–8.
Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250(2):187–96.
Funding
Open access funding provided by Medical University of Graz.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M. Smolle, S.P. Nischwitz, M. Hutan, P. Trunk, D. Lumenta and G.A. Bernhardt declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Smolle, M.A., Nischwitz, S.P., Hutan, M. et al. Closed-incision negative-pressure wound management in surgery—literature review and recommendations. Eur Surg 52, 249–267 (2020). https://doi.org/10.1007/s10353-020-00657-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10353-020-00657-w