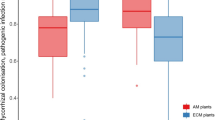
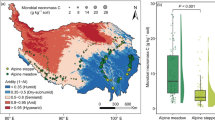
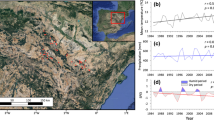
Abstract
Forests worldwide have been recently affected by severe decline and mortality, while our understanding about forest decline across spatial scale is still limited. In this work, we study how Quercus suber trees adjust their physiology, in terms of water use efficiency and secondary growth, to pathogen-induced oak decline at the whole-tree, local and landscape scales. This work was carried out in Mediterranean mixed forests where their dominant key species Q. suber is affected by a severe decline and mortality induced by the exotic soil-borne pathogen Phytophthora cinnamomi. Significant differences were not observed between defoliated and healthy trees, either in terms of water use efficiency or growth at the whole-tree scale. We found that limiting conditions, such as low soil depth and high pathogen abundance, induced trees to higher water use efficiency at local and landscape scales. Overall our findings point out that Q. suber trees subjected to soil drought and root pathogens increase water use efficiency to some extent, while this response might not be enough for the trees to overcome the physiological stress associated with the pathogen-induced dieback. We discuss the complex way by which adult Q. suber trees physiologically respond to P. cinnamomi-induced mortality, improving our understanding of the likely consequences of chronic oak decline in the future.




Similar content being viewed by others
References
Allen CD (2007) Interactions across spatial scales among forest dieback, fire, and erosion in northern New Mexico landscapes. Ecosystems 10:797–808
Allen CD, Breshears DD, McDowell NG (2015) On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere. doi:10.1890/ES15-00203.1
Anderegg WRL, Kane JM, Anderegg LDL (2013) Consequences of widespread tree mortality triggered by drought and temperature stress. Nat Clim Change 3:30–36
Ávila JM, Gallardo A, Ibáñez B, Gómez-Aparicio L (2016) Quercus suber dieback alters soil respiration and nutrient availability in Mediterranean forests. J Ecol 104:1441–1452
Balci Y, Long RP, Mansfield M, Balser D, MacDonald WL (2010) Involvement of Phytophthora species in white oak (Quercus alba) decline in southern Ohio. For Pathol 40:430–442. doi:10.1111/j.1439-0329.2009.00617.x
Barton K (2016) MuMIn: Multi-Model Inference. R package version 1.15.7. https://CRAN.R-project.org/package=MuMIn
Bendixsen DP, Hallgren SW, Frazier AE (2015) Stress factors associated with forest decline in xeric oak forests of south-central United States. For Ecol Manag 347:40–48. doi:10.1016/j.foreco.2015.03.015
Brasier CM (1992) Oak tree mortality in Iberia. Nature 360:539
Brasier CM (1996) Phytophthora cinnamomi and oak decline in southern Europe environmental constraints including climate change. Ann For Sci 53:347–358. doi:10.1051/forest:19960217
Brouwers NC, Mercer J, Lyons T, Poot P, Veneklaas E, Hardy G (2013) Climate and landscape drivers of tree decline in a Mediterranean ecoregion. Ecol Evol 3:67–79. doi:10.1002/ece3.437
Burnham KP, Anderson DR (2002) Model selection and multi-model inference: a practical information-theoretic approach, 2nd edn. Springer, Secaucus, NJ
Camilo-Alves CSP, da Clara MIE, Ribeiro NA (2013) Decline of Mediterranean oak trees and its association with Phytophthora cinnamomi: a review. Eur J For Res 132:411–432. doi:10.1007/s10342-013-0688-z
Chave J (2013) The problem of pattern and scale in ecology: what have we learned in 20 years? Ecol Lett 16:4–16. doi:10.1111/ele.12048
Corcobado T, Cubera E, Moreno G, Solla A (2013a) Quercus ilex forests are influenced by annual variations in water table, soil water deficit and fine root loss caused by Phytophthora cinnamomi. Agric For Meteorol 169:92–99. doi:10.1016/j.agrformet.2012.09.017
Corcobado T, Solla A, Madeira M, Moreno G (2013b) Combined effects of soil properties and Phytophthora cinnamomi infections on Quercus ilex decline. Plant Soil 373:403–413. doi:10.1007/s11104-013-1804-z
Corcobado T, Cubera E, Juárez E, Moreno G, Solla A (2014) Drought events determine performance of Quercus ilex seedlings and increase their susceptibility to Phytophthora cinnamomi. Agric For Meteorol 192–193:1–8. doi:10.1016/j.agrformet.2014.02.007
Costa A, Madeira M, Oliveira ÂC (2008) The relationship between cork oak growth patterns and soil, slope and drainage in a cork oak woodland in Southern Portugal. For Ecol Manag 255:1525–1535
Costa A, Pereira H, Madeira M (2010) Analysis of spatial patterns of oak decline in cork oak woodlands in Mediterranean conditions. Ann For Sci 67:204p201–204p210. doi:10.1051/forest/2009097
Damesin C, Rambal S, Joffre R (1998) Co-occurrence of trees with different leaf habit: a functional approach on Mediterranean oaks. Acta Oecol 19:195–204
Edburg SL et al (2012) Cascading impacts of bark beetle-caused tree mortality on coupled biogeophysical and biogeochemical processes. Front Ecol Environ 10:416–424. doi:10.1890/110173
Ellison AM et al (2005) Loss of foundation species: consequences for the structure and dynamics of forested ecosystems. Front Ecol Environ 3:479–486. doi:10.2307/3868635
Erwin DC, Ribeiro OK (1996) Phytophthora diseases worldwide. APS Press, St Paul
Farquhar G, Richards R (1984) Isotopic composition of plant carbon correlates with water-use efficiency of wheat genotypes. Funct Plant Biol 11:539–552. doi:10.1071/PP9840539
Farquhar G, O’Leary M, Berry J (1982) On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Funct Plant Biol 9:121–137. doi:10.1071/PP9820121
Ferrio JP, Florit A, Vega A, Serrano L, Voltas J (2003) Δ13C and tree-ring width reflect different drought responses in Quercus ilex and Pinus halepensis. Oecologia 137:512–518. doi:10.1007/s00442-003-1372-7
Fisher R, Binkley D (2000) Ecology and management of forest soils, 3rd edn. Wiley, New York
Fleischmann F, Schneider D, Matyssek R, Oßwald W (2002) Investigations on net CO2 assimilation, transpiration and root growth of Fagus sylvatica infested with four different Phytophthora species. Plant Biol 4:144–152
Galiano L, Martínez-Vilalta J, Lloret F (2010) Drought-induced multifactor decline of scots pine in the pyrenees and potential vegetation change by the expansion of co-occurring oak species. Ecosystems 13:978–991. doi:10.1007/s10021-010-9368-8
Galiano L, Martinez-Vilalta J, Sabate S, Lloret F (2012) Determinants of drought effects on crown condition and their relationship with depletion of carbon reserves in a Mediterranean holm oak forest. Tree Physiol 32:478–489. doi:10.1093/treephys/tps025
García LV et al (2011) Protected wading bird species threaten relict centenarian cork oaks in a Mediterranean Biosphere Reserve: a conservation management conflict. Biol Conserv 144:764–771. doi:10.1016/j.biocon.2010.11.007
Gee GW, Bauder JW (1986) Particle-size analysis. SSSA Book Series. In: Klute A (ed) Methods of soil analysis: part 1—physical and mineralogical methods, vol 5.1. Soil Science Society of America, American Society of Agronomy, Madison. doi:10.2136/sssabookser5.1.2ed.c15
Gómez-Aparicio L et al (2012) Spatial patterns of soil pathogens in declining Mediterranean forests: implications for tree species regeneration. New Phytol 194:1014–1024. doi:10.1111/j.1469-8137.2012.04108.x
Gouveia AC, Freitas H (2009) Modulation of leaf attributes and water use efficiency in Quercus suber along a rainfall gradient. Trees 23:267–275
Haavik LJ, Billings SA, Guldin JM, Stephen FM (2015) Emergent insects, pathogens and drought shape changing patterns in oak decline in North America and Europe. For Ecol Manag 354:190–205. doi:10.1016/j.foreco.2015.06.019
Hüberli D, Tommerup IC, Hardy GESJ (2000) False-negative isolations or absence of lesions may cause mis-diagnosis of diseased plants infected with Phytophthora cinnamomi. Australas Plant Pathol 29:164–169. doi:10.1071/ap00029
Ibáñez B, Gómez-Aparicio L, Ávila JM, Pérez-Ramos IM, García LV, Marañón T (2015a) Impact of tree decline on spatial patterns of seedling-mycorrhiza interactions: implications for regeneration dynamics in Mediterranean forests. For Ecol Manag 353:1–9. doi:10.1016/j.foreco.2015.05.014
Ibáñez B, Gómez-Aparicio L, Stoll P, Ávila JM, Pérez-Ramos IM, Marañón T (2015b) A neighborhood analysis of the consequences of Quercus suber decline for regeneration dynamics in Mediterranean forests. PLoS ONE 10:e0117827. doi:10.1371/journal.pone.0117827
Jung T et al (2016) Widespread Phytophthora infestations in European nurseries put forest, semi-natural and horticultural ecosystems at high risk of Phytophthora diseases. For Pathol 46:134–163. doi:10.1111/efp.12239
Levin SA (1992) The problem of pattern and scale in ecology. Ecology 73:1943–1967. doi:10.2307/1941447
Lloret F, Escudero A, Iriondo JM, Martínez-Vilalta J, Valladares F (2012) Extreme climatic events and vegetation: the role of stabilizing processes. Global Change Biol 18:797–805. doi:10.1111/j.1365-2486.2011.02624.x
Lowe S, Browne M, Boudjelas S, De Poorter M (2000) 100 of the world’s worst invasive alien species: a selection from the global invasive species database. Invasive Species Specialist Group Species Survival Commission, World Conservation Union (IUCN), Auckland
Luque J, Cohen M, Save R, Biel C, Alvarez IF (1999) Effects of three fungal pathogens on water relations, chlorophyll fluorescence and growth of Quercus suber L. Ann For Sci 56:19–26. doi:10.1051/forest:19990103
Marañón T, Ajbilou R, Ojeda F, Arroyo J (1999) Biodiversity of woody species in oak woodlands of southern Spain and northern Morocco. For Ecol Manag 115:147–156. doi:10.1016/S0378-1127(98)00395-8
Marañón T, Ibáñez B, Anaya-Romero M, Muñoz-Rojas M, Pérez-Ramos I (2012) Oak trees and woodlands providing ecosystem services in southern Spain. In: Rotherham ID, Handley C, Agnoletti M, Samojlik T (eds) Trees beyond the wood. Wildtrack Publishing, Sheffield, pp 369–378
Martínez-Vilalta J, Prat E, Oliveras I, Piñol J (2002) Xylem hydraulic properties of roots and stems of nine Mediterranean woody species. Oecologia 133:19–29
Maseyk K, Hemming D, Angert A, Leavitt SW, Yakir D (2011) Increase in water-use efficiency and underlying processes in pine forests across a precipitation gradient in the dry Mediterranean region over the past 30 years. Oecologia 167:573–585
Maurel M, Robin C, Capron G, Desprez-Loustau ML (2001) Effects of root damage associated with Phytophthora cinnamomi on water relations, biomass accumulation, mineral nutrition and vulnerability to water deficit of five oak and chestnut species. For Pathol 31:353–369. doi:10.1046/j.1439-0329.2001.00258.x
McCarroll D, Loader NJ (2004) Stable isotopes in tree rings. Quatern Sci Rev 23:771–801
McDowell NG (2011) Mechanisms linking drought, hydraulics, carbon metabolism, and vegetation mortality. Plant Physiol 155:1051–1059
McDowell N et al (2008) Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought? New Phytol 178:719–739
Ninyerola M, Pons X, Roure JM (2005) Atlas climático digital de la Península Ibérica: metodología y aplicaciones en bioclimatología y geobotánica. Universidad Autónoma de Barcelona, Bellaterra
Ogaya R, Penuelas J (2003) Comparative field study of Quercus ilex and Phillyrea latifolia: photosynthetic response to experimental drought conditions. Environ Exp Bot 50:137–148. doi:10.1016/s0098-8472(03)00019-4
Ojeda F, Marañón T, Arroyo J (2000) Plant diversity patterns in the Aljibe Mountains (S. Spain): a comprehensive account. Biodivers Conserv 9:1323–1343
Olea L, San Miguel-Ayanz A (2006) The Spanish dehesa. A traditional Mediterranean silvopastoral system linking production and nature conservation. In: Grassland Science in Europe, vol 11, pp. 3–13
Osswald W et al (2014) Strategies of attack and defence in woody plant-Phytophthora interactions. For Pathol 44:169–190. doi:10.1111/efp.12096
Otieno D, Schmidt M, Kurz-Besson C, Do Vale RL, Pereira J, Tenhunen J (2007) Regulation of transpirational water loss in Quercus suber trees in a Mediterranean-type ecosystem. Tree Physiol 27:1179–1187
Peñuelas J, Filella I, Lloret F, Piñol J, Siscart D (2000) Effects of a severe drought on water and nitrogen use by Quercus ilex and Phyllyrea latifolia. Biol Plant 43:47–53. doi:10.1023/A:1026546828466
Pérez-Sierra A, López-García C, León M, García-Jiménez J, Abad-Campos P, Jung T, Belbahri L (2013) Previously unrecorded low-temperature Phytophthoras pecies associated with Quercus decline in a Mediterranean forest in eastern Spain. For Pathol 43:331–339. doi:10.1111/efp.12037
Peters DP, Bestelmeyer BT, Turner MG (2007) Cross–scale interactions and changing pattern–process relationships: consequences for system dynamics. Ecosystems 10:790–796
R Core Team (2014) R: a language and environment for statistical computing, 3.1.1 edn. R Foundation for Statistical Computing, Vienna
Rawls W, Pachepsky YA, Ritchie J, Sobecki T, Bloodworth H (2003) Effect of soil organic carbon on soil water retention. Geoderma 116:61–76
Robin C, Capron G, Desprez-Loustau ML (2001) Root infection by Phytophthora cinnamomi in seedlings of three oak species. Plant Pathol 50:708–716. doi:10.1046/j.1365-3059.2001.00643.x
Romero MA, Sanchez JE, Jimenez JJ, Belbahri L, Trapero A, Lefort F, Sanchez ME (2007) New Pythium taxa causing root rot on Mediterranean Quercus species in South-West Spain and Portugal. J Phytopathol 155:289–295. doi:10.1111/j.1439-0434.2007.01230.x
Rossi L, Sebastiani L, Tognetti R, d’Andria R, Morelli G, Cherubini P (2013) Tree-ring wood anatomy and stable isotopes show structural and functional adjustments in olive trees under different water availability. Plant Soil 372:567–579. doi:10.1007/s11104-013-1759-0
Sánchez ME, Caetano P, Ferraz J, Trapero A (2002) Phytophthora disease of Quercus ilex in south-western Spain. For Pathol 32:5–18. doi:10.1046/j.1439-0329.2002.00261.x
Sangüesa-Barreda G, Linares JC, Julio Camarero J (2013) Drought and mistletoe reduce growth and water-use efficiency of Scots pine. For Ecol Manag 296:64–73. doi:10.1016/j.foreco.2013.01.028
Saurer M, Siegwolf RT, Schweingruber FH (2004) Carbon isotope discrimination indicates improving water-use efficiency of trees in northern Eurasia over the last 100 years. Global Change Biol 10:2109–2120
Saxton K, Rawls WJ, Romberger J, Papendick R (1986) Estimating generalized soil-water characteristics from texture. Soil Sci Soc Am J 50:1031–1036
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Serrano L, Peñuelas J (2005) Contribution of physiological and morphological adjustments to drought resistance in two Mediterranean tree species. Biol Plant 49:551–559. doi:10.1007/s10535-005-0049-y
Sghaier-Hammami B, Valero-Galvan J, Romero-Rodriguez MC, Navarro-Cerrillo RM, Abdelly C, Jorrin-Novo J (2013) Physiological and proteomics analyses of Holm oak (Quercus ilex subsp ballota Desf. Samp.) responses to Phytophthora cinnamomi. Plant Physiol Biochem 71:191–202. doi:10.1016/j.plaphy.2013.06.030
Shestakova TA, Aguilera M, Ferrio JP, Gutierrez E, Voltas J (2014) Unravelling spatiotemporal tree-ring signals in Mediterranean oaks: a variance-covariance modelling approach of carbon and oxygen isotope ratios. Tree Physiol 34:819–838. doi:10.1093/treephys/tpu037
Solla A, García L, Pérez A, Cordero A, Cubera E, Moreno G (2009) Evaluating potassium phosphonate injections for the control of Quercus ilex decline in SW Spain: implications of low soil contamination by Phytophthora cinnamomi and low soil water content on the effectiveness of treatments. Phytoparasitica 37:303–316. doi:10.1007/s12600-009-0042-7
Sparks DL et al (1996) Methods of soil analysis. Part 3. Chemical methods. Soil Science Society of America Inc., Madison
Sperry J, Hacke U (2002) Desert shrub water relations with respect to soil characteristics and plant functional type. Funct Ecol 16:367–378
Tenhunen J, Serra AS, Harley P, Dougherty R, Reynolds JF (1990) Factors influencing carbon fixation and water use by Mediterranean sclerophyll shrubs during summer drought. Oecologia 82:381–393
Thomas F, Blank R, Hartmann G (2002) Abiotic and biotic factors and their interactions as causes of oak decline in Central Europe. For Pathol 32:277–307
Urbieta IR, Zavala MA, Marañón T (2008) Human and non-human determinants of forest composition in southern Spain: evidence of shifts towards cork oak dominance as a result of management over the past century. J Biogeogr 35:1688–1700
van Mantgem PJ et al (2009) Widespread increase of tree mortality rates in the western United States. Science 323:521–524. doi:10.1126/science.1165000
Weste G, Marks GC (1987) The biology of Phytophthora cinnamomi in australasian forests. Annu Rev Phytopathol 25:207–229. doi:10.1146/annurev.py.25.090187.001231
Zuur A, Ieno EN, Walker N, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, Berlin
Acknowledgements
We thank the director and technicians of Los Alcornocales Natural Park for facilities and support to carry out the field work. We specially thank to Rocío Cortés, Jesús Márquez, María S. Serrano and Paolo de Vita for invaluable laboratory and field assistance. This research was supported by the Ministerio de Ciencia e Innovación (MICINN) projects CGL2011-26877 (RETROBOS) and CGL2014-56739-R (INTERCAPA). J.M.A. was supported by a Formación de Profesorado Universitario FPU-Ministerio de Educación y Ciencia grant (AP2010-0229).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Agustín Merino.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Avila, J.M., Linares, J.C., García-Nogales, A. et al. Across-scale patterning of plant–soil–pathogen interactions in Quercus suber decline. Eur J Forest Res 136, 677–688 (2017). https://doi.org/10.1007/s10342-017-1064-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-017-1064-1