Abstract
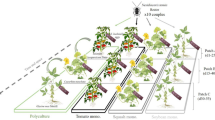
The plant matrix influences the performance of omnivorous mirids as biocontrol agents and increasing plant diversity has been hypothesised to enhance pest control. This research aimed to determine the effect of using calabash, Lagenaria siceraria, as a companion plant on the population dynamics and whitefly control efficacy of Dicyphus argensis in tomato greenhouses. The response of D. argensis was also compared with that of Nesidiocoris tenuis. Four treatments were assayed in a complete randomised block design with three replicates each: (1) Bemisia tabaci, (2) B. tabaci + D. argensis, (3) B. tabaci + D. argensis + calabash and (4) B. tabaci + N. tenuis. Calabash harboured high populations of D. argensis, but its abundance on tomato plants was significantly lower in the presence of calabash than in its absence, and in both treatments, it reached lower numbers than N. tenuis. Dicyphus argensis reduced the whitefly density on tomato plants relative to the compartments with no mirids, but the whitefly density was higher in the presence of companion plants, and N. tenuis was more effective in reducing whitefly populations. Calabash served as a host for the multiplication of whitefly and increased the pest density on tomato. In this research, increasing plant diversity in crops did not enhance pest control because: (1) the aggregation of D. argensis in calabash reduced its abundance in tomato plants; (2) the pest populations multiplied. This contrasts with the diversity hypothesis and confirms the importance of the plant context for predatory dicyphines.





Similar content being viewed by others
Data availability
Data and material will be available upon request.
References
Abbas S, Pérez-Hedo M, Colazza S, Urbaneja A (2014) The predatory mirid Dicyphus maroccanus as a new potential biological control agent in tomato crops. Biocontrol 59:565–574. https://doi.org/10.1007/s10526-014-9587-6
Alomar Ò, Goula M, Albajes R (2002) Colonisation of tomato fields by predatory mirid bugs (Hemiptera: Heteroptera) in northern Spain. Agric Ecosyst Environ 89:105–115. https://doi.org/10.1016/S0167-8809(01)00322-X
Altieri MA (1991) How best can we use biodiversity in agroecosystems. Outlook Agric 20:15–23. https://doi.org/10.1177/003072709102000105
Altieri MA, Letourneau DK (1982) Vegetation management and biological control in agroecosystems. Crop Prot 1:405–430. https://doi.org/10.1016/0261-2194(82)90023-0
Ardanuy A, Figueras M, Matas M, Arnó J, Agustí N, Alomar Ò, Albajes R, Gabarra R (2022) Banker plants and landscape composition influence colonisation precocity of tomato greenhouses by mirid predators. J Pest Sci 95:447–459. https://doi.org/10.1007/s10340-021-01387-y
Arnó J, Ariño J, Español R, Marta, M, Alomar, O (2000) Conservation of Macrolophus caliginosus Wagner (Het. Miridae) in commercial greenhouses during tomato crop-free periods. IOBC wprs Bull 23:241–246.
Balzan MV (2017) Flowering banker plants for the delivery of multiple agroecosystem services. Arthropod Plant Interact 11:743–754. https://doi.org/10.1007/s11829-017-9544-2
Balzan MV, Wäckers FL (2013) Flowers to selectively enhance the fitness of a host-feeding parasitoid: adult feeding by Tuta absoluta and its parasitoid Necremnus artynes. Biol Control 67:21–31. https://doi.org/10.1016/j.biocontrol.2013.06.006
Bianchi FJJ, Booij CJH, Tscharntke T (2006) Sustainable pest regulation in agricultural landscapes: a review on landscape composition, biodiversity and natural pest control. Proc Biol Sci 273:1715–1727. https://doi.org/10.1098/rspb.2006.3530
Biondi A, Zappalà L, Di Mauro A, Tropea-Garzia G, Russo A, Desneux N, Siscaro G (2016) Can alternative host plant and prey affect phytophagy and biological control by the zoophytophagous mirid Nesidiocoris tenuis? Biocontrol 61:79–90. https://doi.org/10.1007/s10526-015-9700-5
Blitzer EJ, Dormann CF, Holzschuh A, Klein AM, Rand TA, Tscharntke T (2012) Spillover of functionally important organisms between managed and natural habitats. Agric Ecosyst Environ 146:34–43. https://doi.org/10.1016/j.agee.2011.09.005
Bresch C, Ottenwalder L, Poncet C, Parolin P (2014) Tobacco as banker plant for Macrolophus pygmaeus to control Trialeurodes vaporariorum in tomato crops. Univers J Agric Res 2:297–304. https://doi.org/10.13189/ujar.2014.020803
Calvo FJ, Urbaneja A (2004) Nesidiocoris tenuis un aliado para el control biológico de mosca blanca. Hortic Int 44:20–25
Calvo J, Bolckmans K, Stansly PA, Urbaneja A (2009) Predation by Nesidiocoris tenuis on Bemisia tabaci and injury to tomato. Biocontrol 54:237–246. https://doi.org/10.1007/s10526-008-9164-y
Calvo FJ, Bolckmans K, Belda JE (2012a) Release rate for a pre-plant application of Nesidiocoris tenuis for Bemisia tabaci control in tomato. Biocontrol 57:809–817. https://doi.org/10.1007/s10526-012-9455-1
Calvo FJ, Lorente MJ, Stansly PA, Belda JE (2012b) Preplant release of Nesidiocoris tenuis and supplementary tactics for control of Tuta absoluta and Bemisa tabaci in greenhouse tomato. Entomol Exp Appl 143:111–119. https://doi.org/10.1111/j.1570-7458.2012.01238.x
Calvo FJ, Torres-Ruiz A, Velázquez-González JC, Rodríguez-Leyva E, Lomeli-Flores JR (2016) Evaluation of Dicyphus hesperus for biological control of sweet potato whitefly and potato psyllid on greenhouse tomato. Biocontrol 61:415–424. https://doi.org/10.1007/s10526-016-9719-2
Cassis G, Schuh RT (2012) Systematics, biodiversity, biogeography, and host associations of the miridae (Insecta: Hemiptera: Heteroptera: Cimicomorpha). Annu Rev Entomol 57:377–404. https://doi.org/10.1146/annurev-ento-121510-133533
Castañé C, Alomar O, Goula M, Gabarra R (2004) Colonization of tomato greenhouses by the predatory mirid bugs Macrolophus caliginosus and Dicyphus tamaninii. Biol Control 30:591–597. https://doi.org/10.1016/j.biocontrol.2004.02.012
Castañé C, Arnó J, Gabarra R, Alomar O (2011) Plant damage to vegetable crops by zoophytophagous mirid predators. Biol Control 59:22–29. https://doi.org/10.1016/j.biocontrol.2011.03.007
Ceglarska EB (1999) Dicyphus hyalinipennis Burm. (Heteroptera: Miridae): a potential biological control agent for glasshouse pests in Hungary. IOBC wprs Bull 22:33–36
Eubanks MD, Styrsky JD, Denno RF (2003) The evolution of omnivory in heteropteran insects. Ecology 84:2549–2556. https://doi.org/10.1890/02-0396
Fischer S, Terrettaz C (2003) Release Strategies of the Mirid Macrolophus caliginosus in Protected Tomato Crops. Rev suisse Vitic Arboric Hortic 35:191–196
Frank SD, Shrewsbury PM, Denno RF (2011) Plant versus prey resources: Influence on omnivore behavior and herbivore suppression. Biol Control 57:229–235. https://doi.org/10.1016/j.biocontrol.2011.03.004
Gillespie DR, McGregor RR (2000) The functions of plant feeding in the omnivorous predator Dicyphus hesperus: water places limits on predation. Ecol Entomol 25:380–386. https://doi.org/10.1046/j.1365-2311.2000.00285.x
Gillespie D, McGregor R, Sanchez JA, VanLaerhoven S, Quiring D, Roitberg B, Foottit R, Schwartz M, Shipp L (2007) An endemic omnivorous predator for control of greenhouse pests. In: Vicent C, Goettel MS, Lazarovits G (eds) Biological control: a global perspective. CABI, Agassiz, pp 128–135. https://doi.org/10.1079/9781845932657.0128
Gillespie DR, Vanlaerhoven SL, McGregor RR, Chan S, Roitberg BD (2012) Plant feeding in an Omnivorous mirid Dicyphus hesperus: why plant context matters. Psyche. https://doi.org/10.1155/2012/495805
Gurr GM, Wratten SD, Landis DA, You M (2017) Habitat management to suppress pest populations: progress and prospects. Annu Rev Entomol 62:91–109. https://doi.org/10.1146/annurev-ento-031616-035050
Hothorn T, Hornik K, Zeileis A (2006) Unbiased recursive partitioning: a conditional inference framework. J Comput Graph Stat 15:651–674. https://doi.org/10.1198/106186006X133933
Ingegno BL, Pansa MG, Tavella L (2011) Plant preference in the zoophytophagous generalist predator Macrolophus pygmaeus (Heteroptera: Miridae). Biol Control 58:174–181. https://doi.org/10.1016/j.biocontrol.2011.06.003
Ingegno BL, Ferracini C, Gallinotti D, Alma A, Tavella L (2013) Evaluation of the effectiveness of Dicyphus errans (Wolff) as predator of Tuta absoluta (Meyrick). Biol Control 67:246–252. https://doi.org/10.1016/j.biocontrol.2013.08.002
Karp DS, Chaplin-Kramer R, Meehan TD, Martin EA, DeClerck F, Grab H et al (2018) Crop pests and predators exhibit inconsistent responses to surrounding landscape composition. Proc Natl Acad Sci U S A 115:E7863–E7870. https://doi.org/10.1073/pnas.1800042115
Kishaba AN, Castle S, McCreight JD, Desjardins PR (1992) Resistance of white-flowered gourd to sweetpotato whitefly. HortScience 11:1217–1221. https://doi.org/10.21273/HORTSCI.27.11.1217
Lambion J (2014) Flower strips as winter shelters for predatory miridae bugs. Acta Hortic 1041:149–156. https://doi.org/10.17660/ActaHortic.2014.1041.16
Landis DA, Wratten SD, Gurr GM (2000) Habitat management to conserve natural enemies of arthropod pests in agriculture. Annu Rev Entomol 45:175–201. https://doi.org/10.1146/annurev.ento.45.1.175
Letourneau DK, Armbrecht I, Rivera BS, Lerma J, Carmona EJ, Daza MC et al (2011) Does plant diversity benefit agroecosystems? a synthetic review. Ecol Appl 21:9–21. https://doi.org/10.1890/09-2026.1
Lichtenberg EM, Kennedy CM, Kremen C, Batáry P, Berendse F, Bommarco R et al (2017) A global synthesis of the effects of diversified farming systems on arthropod diversity within fields and across agricultural landscapes. Glob Chang Biol 23:4946–4957. https://doi.org/10.1111/gcb.13714
López-Gallego E, Cerezuela-Serrano J, Ramírez-Soria M, Pérez-Marcos M, Sanchez JA (2019) Pumpkin as a barrier-trap plant to reduce whitefly immigration into tomato greenhouses. IOBC-WPRS Bull 147: 125–127
Perdikis DC, Favas C, Lykouressis DP, Fantinou A (2007) Ecological relationships between non-cultivated plants and insect predators in agroecosystems: the case of Dittrichia viscosa (Asteraceae) and Macrolophus melanotoma (Hemiptera: Miridae). Acta Oecologica 31:299–306. https://doi.org/10.1016/j.actao.2006.12.005
Perdikis DC, Kapaxidi E, Papadoulis G (2008) Biological control of insect and mite pests in greenhouse solanaceous crops. Eur J Plant Sci Biotechnol 2:125–144
R-Development-Core-Team (2017) A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Riudavets J, Castañe C (1998) Identification and evaluation of native predators of Frankliniella occidentalis (Thysanoptera: Thripidae) in the mediterranean. Environ Entomol 27:86–93. https://doi.org/10.1093/ee/27.1.86
Root RB (1973) Organization of a plant-arthropod association in simple and diverse habitats: the fauna of collards (Brassica oleracea). Ecol Monogr 1:95–124. https://doi.org/10.2307/1942161
Russell EP (1989) Enemies hypothesis: a Review of the Effect of vegetational diversity on predatory insects and parasitoids. Environ Entomol 18:590–599. https://doi.org/10.1093/ee/18.4.590
Sanchez JA (2008b) Zoophytophagy in the plantbug Nesidiocoris tenuis. Agric For Entomol 10:75–80. https://doi.org/10.1111/j.1461-9563.2007.00357.x
Sanchez JA (2009) Density thresholds for Nesidiocoris tenuis (Heteroptera: Miridae) in tomato crops. Biol Control 51:493–498. https://doi.org/10.1016/j.biocontrol.2009.09.006
Sanchez JA, Cassis G (2018) Towards solving the taxonomic impasse of the biocontrol plant bug subgenus Dicyphus (Dicyphus) (Insecta : Heteroptera : Miridae) using molecular, morphometric and morphological partitions. Zool J Linn Soc 184:330–406. https://doi.org/10.1093/zoolinnean/zly005/5003105
Sanchez JA, Gillespie DR, McGregor RR (2003) The effects of mullein (Verbascum thapsus ) on the population dynamics of Dycyphus hesperus (Heteroptera: Miridae) in tomato greenhouses. Biol Control 28:313–319. https://doi.org/10.1016/S1049-9644(03)00116-6
Sanchez JA, Gillespie DR, McGregor RR (2004) Plant preference in relation to life history traits in the zoophytophagous predator Dicyphus hesperus. Entomol Exp Appl 112:7–19. https://doi.org/10.1111/j.0013-8703.2004.00174.x
Sanchez JA, La-Spina M, Lacasa A (2014) Numerical response of Nesidiocoris tenuis (Hemiptera: Miridae) preying on Tuta absoluta (Lepidoptera: Gelechiidae) in tomato crops. Eur J Entomol 111:387–395. https://doi.org/10.14411/eje.2014.041
Sanchez JA, López-Gallego E, Pérez-Marcos M, Perera-Fernández L (2021) The effect of banker plants and pre-plant release on the establishment and pest control of Macrolophus pygmaeus in tomato greenhouses. J Pest Sci 94:297–307. https://doi.org/10.1007/s10340-020-01257-z
Sanchez JA (2008a) Population dynamics constraints in an omnivore dicyphine. In: Mason PG, Gillespie DR, Vincent C (eds) Procceedings of the third international symposium on biological control of arthropods. Christchurch, New Zealand. pp. 268–271. https://doi.org/10.1073/pnas.2002557117
Tillman PG, Northfield TD, Mizell RF, Riddle TC (2009) Spatiotemporal patterns and dispersal of stink bugs (Heteroptera: Pentatomidae) in peanut-cotton farmscapes. Environ Entomol 38:1038–1052. https://doi.org/10.1603/022.038.0411
Tscharntke T, Karp DS, Chaplin-Kramer R et al (2016) When natural habitat fails to enhance biological pest control—Five hypotheses. Biol Conserv 204:449–458. https://doi.org/10.1016/j.biocon.2016.10.001
Urbaneja A, Gonzalez-Cabrera J, Arno J, Gabarra R (2012) Prospects for the biological control of Tuta absoluta in tomatoes of the Mediterranean basin. Pest Manag Sci 68:1215–1222. https://doi.org/10.1002/ps.3344
van Lenteren JC (2012) The state of commercial augmentative biological control: plenty of natural enemies, but a frustrating lack of uptake. Biocontrol 57:1–20. https://doi.org/10.1007/s10526-011-9395-1
van Rijn PCJ, Van Houten YM, Sabelis MW (2002) How plants benefit from providing food to predators even when it is also edible to herbivores. Ecology 83:2664–2679. https://doi.org/10.1890/0012-9658(2002)083[2664:HPBFPF]2.0.CO;2
VanLaerhoven SL, Gillespie DR, Roitberg BD (2006) Patch retention time in an omnivore, Dicyphus hesperus is dependent on both host plant and prey type. J Insect Behav 19:613–621. https://doi.org/10.1007/s10905-006-9047-y
Venables WN, Ripley BD (2002) Statistic and computing. Modern applied statistic with S. Springer-Verlag, New York Inc, New York. https://doi.org/10.1007/978-0-387-21706-2
Voigt D, Gorb E, Gorb S (2007) Plant surface-bug interactions: Dicyphus errans stalking along trichomes. Arthropod Plant Interact 1:221–243. https://doi.org/10.1007/s11829-007-9021-4
Wäckers FL, van Rijn PCJ, Bruin J (eds) (2005) Plant-Provided Food for Carnivorous Insects. Cambridge University Press, Cambridge. https://doi.org/10.1017/CBO9780511542220
Wheeler AG (2001) Biology of the plant bugs (Hemiptera: Miridae). Cornell University Press, Ithaca, New York
Zhao J, Ayers G, Grafius E, Stehr F (1992) Effects of neighboring nectar-producing plants on populations of pest Lepidoptera and their parasitoids in broccoli plantings. Gt Lakes Entomol 25:253–258
Acknowledgements
This work was funded by research project AGL2003-07532-C03-03.
Funding
Project AGL2003-07532-C03-03.
Author information
Authors and Affiliations
Contributions
JAS, MdP and FJC conceived and designed the research; JAS analysed the data and wrote the manuscript. MdP and FJC conducted the experiments as well as revised and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by Alberto Urbaneja.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sanchez, J.A., del Pino, M. & Calvo, F.J. Increasing plant diversity does not always enhance the efficacy of omnivorous mirids as biocontrol agents. J Pest Sci 95, 1557–1566 (2022). https://doi.org/10.1007/s10340-022-01526-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-022-01526-z