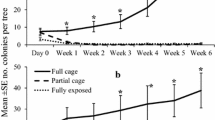
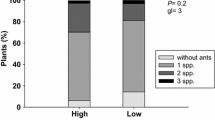
Abstract
Rich and abundant predator complexes are frequently associated with aphids in perennial agroecosystems. The ability of these predators to successfully suppress aphid populations is nevertheless highly variable. The development of operative conservation biological control strategies is mostly hindered by the lack of knowledge of the specific roles of the aphidophagous assemblage components, their intra-guild relationships and the predatory attributes that chiefly determine their effectiveness. The role of predation in the biological control of aphids in perennial agroecosystems was assessed through exclusion experiments in aphid infested citrus crops. Important predator attributes such as recruitment, aphid consumption rates, and foraging strategies were related to their efficacy. Predation greatly affected aphid colony phenology as well as size. Predators with lower aphid consumption rates (Micro-coccinellid species and Cecidomyiidae) were revealed to be the most efficient aphidophaga. These predators encountered aphid colonies at earlier colony stages and significantly reduced their population growth rates. Later more voracious aphidophaga groups (Chrysopidae and Macro-coccinellids) did not present effective biological control of the colonies. Contrarily to what was widely believed, the less voracious aphidophaga groups such as the Micro-coccinellids and Cecidomyiids are probably the groups who are mostly responsible for aphid suppression. Future conservation biological control studies in this crop should therefore chiefly focus on these groups.





Similar content being viewed by others
References
Anderson D, Burnham K (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, New York, p 488
Atlıhan R, Kaydan B, Özgökçe MS (2004) Feeding activity and life history characteristics of the generalist predator, Chrysoperla carnea (Neuroptera: Chrysopidae) at different prey densities. J Pest Sci 77:17–21. https://doi.org/10.1007/s10340-003-0021-6
Boulanger FX, Jandricic S, Bolckmans K, Wäckers F, Pekas A (2019) Optimizing aphid biocontrol with the predator Aphidoletes aphidimyza, based on biology and ecology. Pest Manag Sci 75:1479–1493. https://doi.org/10.1002/ps.5270
Bouvet JPR, Urbaneja A, Pérez-Hedo M, Monzó C (2019a) Contribution of predation to the biological control of a key herbivorous pest in citrus agroecosystems. J Anim Ecol 88:915–926. https://doi.org/10.1111/1365-2656.12982
Bouvet JPR, Urbaneja A, Monzó C (2019b) Effects of citrus overwintering predators, host plant phenology and environmental variables on aphid infestation dynamics in clementine citrus. J Econ Entomol 112:1587–1597. https://doi.org/10.1093/jee/toz101
Bouvet JPR, Urbaneja A, Monzo C (2019c) Life history traits of the coccinellids Scymnus subvillosus and S. interruptus on their prey Aphis spiraecola and A. gossypii: implications for biological control of aphids in clementine citrus. Biol Control 132:49–56. https://doi.org/10.1016/j.biocontrol.2019.02.002
Brewer MJ, Elliott NC (2004) Biological control of cereal aphids in North America and mediating effects of host plant and habitat manipulations. Annu Rev Entomol 49:219–242. https://doi.org/10.1146/annurev.ento.49.061802.123149
Bugg RL, Colfer RG, Chaney WE, Smith HA, Cannon J (2008) Flower flies (Syrphidae) and other biological control agents for aphids in vegetable crops. http://dx.doi.org/10.3733/ucanr.8285. Retrieved from https://escholarship.org/uc/item/37k3d5g8
Cambra M, Gorris MT, Marroquın C, Román MP, Olmos A, Martınez MC, Hermoso de Mendoza A, Lopez A, Navarro L (2000) Incidence and epidemiology of Citrus tristeza virus in the Valencian Community of Spain. Virus Res 71:85–95. https://doi.org/10.1016/S0168-1702(00)00190-8
Chen TY, Liu TX (2001) Relative consumption of three aphid species by the lacewing, Chrysoperla rufilabris, and effects on its development and survival. Biocontrol 46:481–491. https://doi.org/10.1023/A:1014115906698
DeBach P, Rosen D (1991) Biological control by natural enemies. Cambridge University Press, Cambridge, p 456
Gómez-Marco F, Urbaneja A, Jaques JA, Rugman-Jones PF, Stouthamer R, Tena A (2015) Untangling the aphid-parasitoid food web in citrus: can hyperparasitoids disrupt biological control? Biol Control 81:111–121. https://doi.org/10.1007/s10340-015-0668-9
Gómez-Marco F, Tena A, Jaques JA, Urbaneja A (2016) Early arrival of predators controls Aphis spiraecola colonies in citrus clementines. J Pest Sci 89:69–79
Harwood JD, Obrycki JJ (2005) Quantifying aphid predation rates of generalist predators in the field. Eur J Entomol 102:335–350
Hassell MP (1978) The dynamics of arthropod predator-prey systems. Princeton University Press, Princeton, p 237
Hassell MP, May RM (1986) Generalist and specialist natural enemies in insect predator-prey interactions. J Anim Ecol 55:923–940. https://doi.org/10.2307/4425
Hermoso de Mendoza A, Ballester-Olmos JF, Pina JA (1984) Transmission of citrus tristeza virus by aphids (Homoptera, Aphididae) in Spain. In: International organization of citrus virologists conference proceedings 1957–2010:1984
Hermoso de Mendoza A, Arouni R, Belliure B, Carbonell EA, Pérez-Panadés J (2006) Intervention thresholds for Aphis spiraecola (Hemiptera: Aphididae) on Citrus clementina. J Econ Entomol 99:1273–1279. https://doi.org/10.1603/0022-0493-99.4.1273
Hodek I, Honêk A (2013) Ecology of coccinellidae. Springer, Berlin, p 463
Kindlmann P, Dixon AFG (1999) Strategies of aphidophagous predators: lessons for modelling insect predator–prey dynamics. J Appl Entomol 123:397–399. https://doi.org/10.1046/j.1439-0418.1999.00390.x
Komazaki S (1982) Effects of constant temperatures on population growth of three aphid species, Toxoptera citricidus (Kirkaldy), Aphis citricola van der Goot and Aphis gossypii Glover (Homoptera: Aphididae) on citrus. Appl Entomol Zool 17:75–81. https://doi.org/10.1303/aez.17.75
Lester PJ, Harmsen R (2002) Functional and numerical responses do not always indicate the most effective predator for biological control: an analysis of two predators in a two-prey system. J Appl Ecol 39:455–468
Liu L, Forman S, Barton B (2009) Fitting cox model using PROC PHREG and beyond in SAS. In: SAS global forum 2009, pp 22–25. https://pdfs.semanticscholar.org/1302/e5605f669c6141660984ddda4a94d04f94f0.pdf
Lucas É (2005) Intraguild predation among aphidophagous predators. Eur J Entomol 102:351–364. https://doi.org/10.14411/eje.2005.052
Lucas É, Coderre D, Brodeur J (1998) Intraguild predation among aphid predators: characterization and influence of extraguild prey density. Ecology 79:1084–1092. https://doi.org/10.1890/0012-9658(1998)079%5b1084:IPAAPC%5d2.0.CO;2
Michaud JP (1999) Sources of mortality in colonies of brown citrus aphid, Toxoptera citricida. Biocontrol 44:347–367. https://doi.org/10.1023/A:100995581
Monzó C, Qureshi JA, Stansly PA (2014) Insecticide sprays, natural enemy assemblages, and predation on Asian citrus psyllid, Diaphorina citri (Hemiptera: Psyllidae). B Entomol Res 104:576–585. https://doi.org/10.1017/S0007485314000315
Monzó C, Catalán J, Laurín MC, Montoro M, Torné M, Abad R, Urbaneja A (2019) Evaluación de los efectos secundarios del Sulfoxaflor sobre la fauna útil de cítricos. Levante Agrícola 448:189–194
Pinol J, Espadaler X, Cañellas N, Pérez N (2009) Effects of the concurrent exclusion of ants and earwigs on aphid abundance in an organic citrus grove. BioControl 54:515–527. https://doi.org/10.1007/s10526-008-9203-8
Raki A, Verheggen FJ, Haubruge É (2009) Searching and oviposition behavior of aphidophagous hoverflies (Diptera: Syrphidae): a review. Biotechnol Agron Soc Environ 13:467–481. https://doi.org/10.14411/eje.2003.001
SAS/STAT 15.1. User’s Guide. The NLIN Procedure. http://documentation.sas.com/?docsetId=statug&docsetTarget=statug_nlin_syntax.htm&docsetVersion=15.1&locale=es. Last access: January 2020
Schabenberger O, Pierce FJ (2001) Contemporary statistical models for the plant and soil sciences. CRC Press, Boca Raton, p 730
Seagraves MP (2009) Lady beetle oviposition behavior in response to the trophic environment. Biol Control 51:313–322. https://doi.org/10.1016/j.biocontrol.2009.05.015
Sobhani M, Madadi H, Gharali B (2013) Host plant effect on functional response and consumption rate of Episyrphus balteatus (Diptera: Syrphidae) feeding on different densities of Aphis gossypii (Hemiptera: Aphididae) of Aphis gossypii (Hemiptera: Aphididae). J Crop Prot 2:375–385
Symondson WOC (2002) Molecular identification of prey in predator diets. Mol Ecol 11:627–641
Tena A, Pekas A, Cano D, Wäckers FL, Urbaneja A (2015) Sugar provisioning maximizes the biocontrol service of parasitoids. J Appl Ecol 52:795–804. https://doi.org/10.1111/1365-2664.12426
Van Driesche RG, Carruthers RI, Center T, Hoddle MS, Hough-Goldstein J, Morin L et al (2010) Classical biological control for the protection of natural ecosystems. Biol Control 54:S2–S33. https://doi.org/10.1016/j.biocontrol.2010.03.003
Van Emden HF, Harrington R (2007) Aphids as crop pests. CABI Publishing, London, p 717
Wolfinger R, O’connell M (1993) Generalized linear mixed models a pseudo-likelihood approach. J Stat Comput Simul 48:233–243. https://doi.org/10.1080/00949659308811554
Xia JY, Rabbinge R, Van Der Werf W (2003) Multistage functional responses in a ladybeetle-aphid system: scaling up from the laboratory to the field. Environ Entomol 32:151–162. https://doi.org/10.1603/0046-225X-32.1.151
Yahiaoui D, Addante R, Djelouah K, D’Onghia AM (2009) Preliminary monitoring of Citrus tristeza virus (CTV) vectors in Apulia region. Options Méditerranéennes B 65:173–175
Zarpas KD, Margaritopoulos JT, Tsitsipis JA (2007) Life histories of generalist predatory species, control agents of the cotton aphid Aphis gossypii (Hemiptera: Aphididae). Entomol Gen 30:85–102. https://doi.org/10.1127/entom.gen/30/2007/85
Zollner U, Poehling HM (1994) Influence of different aphid species on the efficiency of gall midge larvae (Aphidoletes aphidimyza)(Rond.)(Diptera, Cecidomyiidae). Mededelingen-Faculteit Landbouwkundige en Toegepaste Biologische Wetenschappen Universiteit Gent (Belgium) 59:281–286
Acknowledgements
JPRB was the recipient of a research fellowship from INTA-Argentina (INTA 1029_2012). CM was the recipient of a Juan de la Cierva program Grant (Juan e la Cierva 2014) from the Ministerio de Economía y Competitividad, Spain. We would like to thank Antonio Vicent Civera (IVIA) for his guidance in the statistical analyses.
Funding
IVIA research project 52004.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Juan Pedro Bouvet declares that he has no conflict of interest. Alberto Urbaneja declares that he has no conflict of interest. Cesar Monzo declares that he has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by: Michael Traugott.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bouvet, J.P.R., Urbaneja, A. & Monzó, C. Aphid predators in citrus crops: the least voracious predators are the most effective. J Pest Sci 94, 321–333 (2021). https://doi.org/10.1007/s10340-020-01265-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-020-01265-z