Abstract
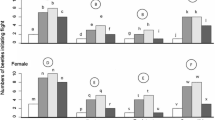
An understanding of how sublethal exposure to phosphine affects an organism’s respiration is important for predicting its effects on activities depending on a reliable energy source including movement. After treatment with the appropriate sublethal doses of phosphine (LC10), both resistant and susceptible Tribolium castaneum beetles suffered reductions in respiration rates, flight initiation rates and successful location (by walking) of aggregation pheromone lures. The susceptible beetles showed a significantly greater reduction in their respiration rate (53.5%) after exposure to sublethal doses of phosphine compared to the resistant beetles (37.5%), although the respiration rate was still higher in susceptible beetles than in resistant ones. Simultaneously, much larger decreases in the activities associated with both walking and flight were observed in the susceptible beetles, after exposure to sublethal doses of phosphine, compared to the decreases recorded in resistant beetles. The relatively greater reduction in the locomotory responses of susceptible beetles showed that these insects may withstand exposure to phosphine by remaining relatively still and reducing their respiration rate. They would thus minimize their exposure to phosphine, but perhaps only incidentally because the lack of movement may be the result of impairment. By contrast, phosphine-resistant beetles, when exposed to phosphine fumigation, continue to move and may even remove themselves from areas of phosphine exposure. An understanding of how the survivors of sublethal exposures behave is important for predicting the spread and local build-up of phosphine resistance genes and thus improving the control and resistance management of stored grain pests.


Similar content being viewed by others
References
Ahmad F, Walter GH, Raghu S (2012) Comparative performance of Tribolium castaneum (Herbst)(Coleoptera: Tenebrionidae) across populations, resource types and structural forms of those resources. J Stored Prod Res 48:73–80
Ahmedani MS, Shaheen N, Ahmedani MY, Aslam M (2007) Status of phosphine resistance in Khapra beetle, Trogoderma granarium (Everts) strains collected from remote villages of Rawalpindi district. Pak Entomol 29:95–102
Arnold PA, Cassey P, White CR (2016) Maturity matters for movement and metabolic rate: trait dynamics across the early adult life of red flour beetles. Anim Behav 111:181–188
Bell C (2000) Fumigation in the 21st century. Crop Prot 19:563–569
Benhalima H, Chaudhry M, Mills K, Price N (2004) Phosphine resistance in stored-product insects collected from various grain storage facilities in Morocco. J Stored Prod Res 40:241–249
Bond E, Upitis E (1973) Response of three insects to sublethal doses of phosphine. J Stored Prod Res 8:307–313
Cato AJ, Elliott B, Nayak MK, Phillips TW (2017) Geographic variation in phosphine resistance among North American populations of the red flour beetle (Coleoptera: Tenebrionidae). J Econ Entomol 110:1359–1365
Chaudhry MQ (2000) Phosphine resistance. Pestic Outlook 11:88–91
Chaudhry MQ, Price NR (1990) Insect mortality at doses of phosphine which produce equal uptake in susceptible and resistant strains of Rhyzopertha dominica (F.) (Coleoptera: Bostrychidae). J Stored Prod Res 26:101–107
Chaudhry MQ, Bell H, Savvidou N, MacNicoll A (2004) Effect of low temperatures on the rate of respiration and uptake of phosphine in different life stages of the cigarette beetle Lasioderma serricorne (F.). J Stored Prod Res 40:125–134
Chefurka W, Kashi K, Bond E (1976) The effect of phosphine on electron transport in mitochondria. Pest Biochem Physiol 6:65–84
Daglish GJ, Collins PJ, Pavic H, Kopittke KA (2002) Effects of time and concentration on mortality of phosphine-resistant Sitophilus oryzae (L.) fumigated with phosphine. Pest Manag Sci 58:1015–1021
Daglish GJ, Nayak MK, Pavic H, Smith LW (2015) Prevalence and potential fitness cost of weak phosphine resistance in Tribolium castaneum (Herbst) in eastern Australia. J Stored Prod Res 61:54–58
FAO (1975) Recommended methods for the detection and measurements of agricultural pests to pesticides. 16: tentative method for adults of some stored cereals, with methyl bromide and phosphine. FAO Plant Prot Bull 23:12–25
Glazier DS (2005) Beyond the ‘3/4-power law’: variation in the intra- and interspecific scaling of metabolic rate in animals. Biol Rev 80:611–662
Halstead D (1963) External sex differences in stored-products Coleoptera. Bull Entomol Res 54:119–134
Ho S, Winks R (1995) The response of Liposcelis bostrychophila Badonnel and L. entomophila (Enderlein) (Psocoptera) to phosphine. J Stored Prod Res 31:191–197
Hobbs S, Bond E (1989) Response of Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae) to sublethal treatments with phosphine. J Stored Prod Res 25:137–146
Hole BD, Bell C, Mills K, Goodship G (1976) The toxicity of phosphine to all developmental stages of thirteen species of stored product beetles. J Stored Prod Res 12:235–244
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363
Jagadeesan R, Collins PJ, Daglish GJ, Ebert PR, Schlipalius DI (2012) Phosphine resistance in the rust red flour beetle, Tribolium castaneum (Coleoptera: Tenebrionidae): inheritance, gene interactions and fitness costs. PLoS ONE 7:1–12
Kaur R, Nayak MK (2015) Developing effective fumigation protocols to manage strongly phosphine-resistant Cryptolestes ferrugineus (Stephens) (Coleoptera: Laemophloeidae). Pest Manag Sci 71:1297–1302
Konemann CE, Hubhachen Z, Opit GP, Gautam S, Bajracharya NS (2017) Phosphine resistance in Cryptolestes ferrugineus (Coleoptera: Laemophloeidae) collected from grain storage facilities in Oklahoma, USA. J Econ Entomol 110:1377–1383
Lighton JRB (2008) Measuring metabolic rates: a manual for scientists. Oxford University Press, Oxford
Lorini I, Collins PJ, Daglish GJ, Nayak MK, Pavic H (2007) Detection and characterisation of strong resistance to phosphine in Brazilian Rhyzopertha dominica (F.) (Coleoptera: Bostrychidae). Pest Manag Sci 63:358–364
Malekpour R, Rafter MA, Daglish GJ, Walter GH (2016) Influence of phosphine resistance genes on flight propensity and resource location in Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae): the landscape for selection. Biol J Linn Soc 119:348–358
Malekpour R, Rafter MA, Daglish GJ, Walter GH (2018) The movement abilities and resource location behaviour of Tribolium castaneum: phosphine resistance and its genetic influences. J Pest Sci 91:739–749
Mills K (1983) Resistance to the fumigant hydrogen phosphide in some stored-product species associated with repeated inadequate treatments. Mitt Dtsch Ges Allg Angew Entomol 4:98–101
Nakakita H, Kuroda J (1986) Differences in phosphine uptake between susceptible and resistant strains of insects. J Pestic Sci 11:21–26
Nath NS, Bhattacharya I, Tuck AG, Schlipalius DI, Ebert PR (2011) Mechanisms of phosphine toxicity. J Toxic 2011:1–9
Nayak MK, Collins PJ (2008) Influence of concentration, temperature and humidity on the toxicity of phosphine to the strongly phosphine resistant psocid Liposcelis bostrychophila Badonnel (Psocoptera: Liposcelididae). Pest Manag Sci 64:971–976
Nayak MK, Collins PJ, Pavic H, Kopittke RA (2003) Inhibition of egg development by phosphine in the cosmopolitan pest of stored products Liposcelis bostrychophila (Psocoptera: Liposcelididae). Pest Manag Sci 59:1191–1196
Nayak MK, Holloway JC, Emery RN, Pavic H, Bartlet J, Collins PJ (2013) Strong resistance to phosphine in the rusty grain beetle, Cryptoestes ferrugineus (Stephens) (Coleoptera: Laemophloeidae): its characterisation, a rapid assay for diagnosis and its distribution in Australia. Pest Manag Sci 69:48–53
Niitepõld K, Smith AD, Osborne JL, Don RR, Carreck NL, Martin AP, Marden JH, Ovaskainen O, Hanski I (2009) Flight metabolic rate and Pgi genotype influence butterfly dispersal rate in the field. Ecology 90:2223–2232
Perez-Mendoza J, Campbell JF, Throne JE (2011) Influence of age, mating status, sex, quantity of food, and long-term food deprivation on red flour beetle (Coleoptera: Tenebrionidae) flight initiation. J Econ Entomol 104:2078–2086
Pimentel MAG, Faroni LRDA, Tótola MR, Guedes RNC (2007) Phosphine resistance, respiration rate and fitness consequences in stored-product insects. Pest Manag Sci 63:876–881
Pimentel MAG, Faroni LRDA, Corrêa AS, Guedes RNC (2012) Phosphine-induced walking response of the lesser grain borer (Rhyzopertha dominica). Pest Manag Sci 68:1368–1373
Price NR, Dance SJ (1983) Some biochemical aspects of phosphine action and resistance in three species of stored product beetles. Comp Biochem Physiol Part C: Comp Pharmacol 76:277–281
R Development Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Raymond B, Wright DJ, Bonsall MB (2011) Effects of host plant and genetic background on the fitness costs of resistance to Bacillus thuringiensis. Heredity 106:281–288
Rees D (2004) Insects of stored products. Manson Publishing, London
Ridley AW, Schlipalius DI, Daglish GJ (2012a) Reproduction of phosphine resistant Rhyzopertha dominica (F.) following sublethal exposure to phosphine. J Stored Prod Res 48:106–110
Ridley AW, Magabe S, Schlipalius DI, Rafter MA, Collins PJ (2012b) Sublethal exposure to phosphine decreases offspring production in strongly phosphine resistant female red flour beetles, Tribolium castaneum (Herbst). PLoS ONE 7:e53356
Schlipalius DI, Valmas N, Tuck AG, Jagadeesan R, Ma L, Kaur R, Goldinger A, Anderson C, Kuang J, Zuryn S, Mau YS, Cheng Q, Collins PJ, Nayak MK, Schirra HJ, Hilliard MA, Ebert PR (2012) A core metabolic enzyme mediates resistance to phosphine gas. Science 338:807–810
Suzuki T (1980) 4,8-Dimethyldecanal: the aggregation pheromone of the flour beetles, Tribolium castaneum and T. confusum (Coleoptera: Tenebrionidae). Agric Biol Chem 44:2519–2520
Valmas N, Zuryn S, Ebert PR (2008) Mitochondrial uncouplers act synergistically with the fumigant phosphine to disrupt mitochondrial membrane potential and cause cell death. Toxicology 252:33–39
White CR, Kearney MR (2014) Metabolic scaling in animals: methods, empirical results, and theoretical explanations. Comp Physiol 4:231–256
Winks RG, Waterford CJ (1986) The relationship between concentration and time in the toxicity of phosphine to adults of a resistant strain of Tribolium castaneum (Herbst). J Stored Prod Res 22:85–92
Acknowledgements
We acknowledge Prof. Craig White for provision of equipment for measuring respiration rate. We also acknowledge the Australian Government’s Australia–India Strategic Research Fund. R.M. is thankful for the funding provided by an International Scholarship from the University of Queensland towards her PhD studies.
Funding
This work was supported by the Australian Government’s Australia–India Strategic Research Fund (GCF010006) for the project “Ensuring food security: harnessing science to protect our grain harvest from insect threats” under which the research and analysis were completed.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by C.G. Athanassiou.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Malekpour, R., Arnold, P.A., Rafter, M.A. et al. Effects of sublethal phosphine exposure on respiration rate and dispersal propensity of adult females of Tribolium castaneum. J Pest Sci 93, 149–157 (2020). https://doi.org/10.1007/s10340-019-01133-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-019-01133-5