Abstract
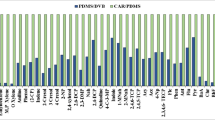
This study reports on the development of a fast and efficient method based on headspace solid-phase microextraction (HS-SPME) coupled to gas chromatography–tandem mass spectrometry (GC–MS/MS) for simultaneous analysis of 128 volatile or semi-volatile pesticide residues belonging to nine classes of pesticides. The important factors related to HS-SPME performance were optimized; these factors include fiber types, water volume, ion strength, extraction temperature, and extraction time. The best extraction conditions include a PDMS/DVB fiber, and analytes were extracted at 90 °C for 60 min from 1 g of tea added to 5 mL of 0.2 g mL−1 NaCl solution. The methodology was validated using tea samples spiked with pesticides at three concentration levels (10, 50, and 100 μg kg−1). In green tea, oolong tea, black tea, and puer tea, 82.8, 88.3, 79.7, and 84.3% of the targeted pesticides meet recoveries ranging from 70 to 120% with a relative standard deviation of ≤ 20%, respectively, when spiked at a level of 10 μg kg−1. Limits of quantification in this method for most of the pesticides were 1 or 5 μg kg−1, which are far below their maximum residue limits prescribed by EU. The optimized method was employed to analyze 30 commercial samples obtained from local markets; 17 pesticide residues were detected at concentrations of 2–452 μg kg−1. Chlorpyrifos was the most detected pesticide in 80% of the samples, and the highest concentration of dicofol (452 μg kg−1) was found in a puer tea. This is the first time to find that the optimized extraction temperature for pesticide residues is 90 °C, which is much higher than other reported HS-SPME extraction conditions in tea samples. This developed method could be used to screen over one hundred volatile or semi-volatile pesticide residues which belong to multiple classes in tea samples, and it is an accurate and reliable technique.






Similar content being viewed by others
References
Li YF, Quyang SH, Chang YQ, Wang TM, Li WX, Tian HY, Cao H, Kurihara H, He RR (2016) Food Chem 216:282–288
Darvesh AS, Bishayee A (2013) Nutr Cancer 65:329–344
Kraujalytė V, Pelvan E, Alasalvar C (2015) Food Chem 194:864–872
E U Commission, Regulation (EC) No. 396/2005 of the European Parliament and of the Council of 23 February 2005 on Maximum Residue Levels of Pesticides in Products of Plant and Animal Origin
Huo F, Tang H, Wu X, Chen D, Zhao T, Liu P, Li L (2016) J Chromatogr B 1023:44–54
Duan Y, Guan N, Li P, Li J, Luo J (2016) Food Contr 59:250–255
Wang Z, Chang Q, Kang J, Cao Y, Ge N, Fan C, Pang G (2015) Anal Methods 7:6385–6402
Kadir HA, Abas F, Zakaria O, Ismail IS, Lajis NH (2015) Anal Methods 7:3141–3147
Celeiro M, Llompart M, Lamas JP, Lores M, Garcia-Jares C, Dagnac T (2014) J Chromatogr A 1343:18–25
Chiesa LM, Labella GF, Giorgi A, Panseri S, Pavlovic R, Bonacci S, Arioli F (2016) Chemosphere 154:482–490
Pano-Farias NS, Ceballos-Magana SG, Gonzalez J, Jurado JM, Muniz-Valencia R (2015) J Sep Sci 38:1240–1247
Tan Q, Fan J, Gao R, He R, Wang T, Zhang Y, Zhang W (2017) Talanta 164:362–367
Yoshida T, Itou A, Yamamoto R, Tobino T, Murakawa H, Toda K (2013) Anal Sci 29:919–922
Steinborna A, Alder L, Spitzkeb M, Doerk D, Anastassiades M (2017) J Agric Food Chem 65:1296–1305
Han Y, Song L, Zou N, Chen R, Qin Y, Pan C (2016) J Chromatogr B 1031:99–108
González-Curbelo MÁ, Lehotay SJ, Hernández-Borges J, Rodríguez-Delgado MÁ (2014) J Chromatogr A 1358:75–84
He Z, Wang L, Peng Y, Luo M, Wang W, Liu X (2015) Food Chem 169:372–380
Dawidowicz AL, Szewczyk J, Dybowski MP (2016) Anal Chim Acta 935:1–8
Du L, Li J, Li W, Li Y, Li T, Xiao D (2014) Food Res Int 57:61–70
Gutiérrez-Serpa A, Rocío-Bautista P, Pino V, Jiménez-Moreno F, Jiménez-Abizanda AI (2017) J Sep Sci 40:2009–2021
Wu M, Wang L, Zeng B, Zhao F (2016) J Chromatogr A 1444:42–49
Souza-Silva ÉA, Lopez-Avilab V, Pawliszyn J (2013) J Chromatogr A 1313:139–146
Zhang S, Yang Q, Yang X, Wang W, Li Z, Zhang L, Wang C, Wang Z (2017) Talanta 166:46–53
Wu F, Lu W, Chen J, Liu W, Zhang L (2010) Talanta 82:1038–1043
Juan PM, Carrillo JD, Tena MT (2007) J Chromatogr A 1139:27–35
Tat L, Comuzzo P, Stolfo I, Battistutta F (2005) Food Chem 93:361–369
Bianco G, Novario G, Ziann IR, Cataldi TR (2009) Anal Bioanal Chem 393:2019–2027
Torrens J, Riu-Aumatell M, López-Tamames E, Buxaderas S (2004) J Chromatogr Sci 42:310–316
Menezes Filho A, dos Santos FN, de Paula Pereira PA (2010) Talanta 81:348–354
Verzera A, Ziino M, Condurso C, Romeo V, Zappala M (2004) Anal Bioanal Chem 380:930–936
Xu X, Yu C, Han J, Li J, El-Sepai F, Zhu Y, Huang B, Cai Z, Wu H, Ren Y (2011) J Sep Sci 34:210–216
Feng J, Tang H, Chen D, Wang G, Li L (2012) Anal Methods 4:4198–4203
Hayward DG, Wong JW, Park HY (2015) J Agric Food Chem 63:8116–8124
Acknowledgements
The authors acknowledge the financial support of the Key Basic Research Program (NO. 2015FY111200) of the Ministry of Science and Technology, P. R. China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest with this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, J., Zhang, Z., Sun, M. et al. Use of a Headspace Solid-Phase Microextraction-Based Methodology Followed by Gas Chromatography–Tandem Mass Spectrometry for Pesticide Multiresidue Determination in Teas. Chromatographia 81, 809–821 (2018). https://doi.org/10.1007/s10337-018-3499-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-018-3499-z