Abstract
Climate change is a key driver of biodiversity loss in multiple ecosystems, which can act at multiple stages of a species life- and annual cycle. Identifying where, when, and how these impacts may happen is key to understanding, and planning for, the population-level effects of climate change. This study assesses how climate change will impact the breeding and non-breeding areas of the European Bee-eater Merops apiaster, a long-distance migratory bird, by combining correlative species distribution models with recent tracking data to account for population connectivity between breeding and non-breeding sites. The three populations studied (Iberian, German, and Bulgarian) demonstrated different levels of exposure to climate change, as well as different patterns of connectivity. The Iberian breeding population showed high exposure to climate change in both its breeding and non-breeding sites, which may result in significant, interacting impacts on this population. In contrast, breeding populations in Germany are likely to benefit from climate change, both in their breeding and non-breeding range. Connectivity also varied; while most populations demonstrated extremely high connectivity between breeding and non-breeding sites, the Iberian population was substantially more behaviourally flexible, indicating there may be some adaptive capacity of this population to change non-breeding sites if some become less suitable due to climate change. Incorporating breeding and non-breeding areas into species distribution modelling highlights how the impacts of climate change may combine into multiplicative impacts across a species’ annual cycle, and how combining methodologies and data sources can provide greater insight into the impact of climate change on migratory species and, in turn, inform conservation action.
Zusammenfassung
Vernetzte Effekte: Die Kombination von Zugdaten mit der Modellierung der Artenverteilung zeigt die vielschichtigen möglichen Auswirkungen des Klimawandels auf den Bienenfresser.
Der Klimawandel ist eine der Hauptursachen für den Verlust der biologischen Vielfalt in vielen Ökosystemen und wirkt sich möglicherweise auf verschiedene Teile des Lebens- und Jahreszyklus einer Art aus. Festzustellen, wo, wann und wie diese Auswirkungen auftreten, ist der Schlüssel zum Verständnis – und Berücksichtigung – der Folgen des Klimawandels auf Populationsebene. In dieser Studie wird untersucht, wie sich der Klimawandel auf die Brut- und Nichtbrutgebiete des Bienenfressers Merops apiaster, eines Langstreckenziehers, auswirken wird. Hierfür werden entsprechende Verbreitungsmodelle für diese Art mit aktuellen Tracking-Daten kombiniert, um einen Zusammenhang der Populationen zwischen ihren Brut- und Nichtbrutgebieten zu untersuchen. Die drei untersuchten Populationen (eine iberische, eine deutsche und eine bulgarische) waren dem Klimawandel mit unterschiedlicher Intensität ausgesetzt und wiesen auch unterschiedliche Vernetzungsmuster auf. Die iberische Brutpopulation war dem Klimawandel sowohl an ihren Brut- als auch an ihren Nichtbrutplätzen stark ausgesetzt, was zu erheblichen Wechselwirkungen mit dieser Population führen könnte. Im Gegensatz dazu könnten die Brutpopulationen in Deutschland sowohl in ihrem Brutgebiet als auch in ihrem Nichtbrutgebiet möglicherweise vom Klimawandel profitieren. Auch die Vernetzung variierte, wobei die meisten Populationen eine extrem hohe Vernetzung zwischen Brut- und Nichtbrutplätzen aufwiesen. Die iberische Population war in ihrem Verhalten wesentlich flexibler, was darauf hindeutet, dass diese Population möglicherweise über eine gewisse Anpassungsfähigkeit verfügt, um von einem Nichtbrutplatz zu einem anderen zu wechseln, wenn dieser aufgrund des Klimawandels geeigneter würde. Die Einbeziehung von Brut- und Nichtbrutgebiete in die Verbreitungsmodelle einer Art kann verdeutlichen, wie sich die Auswirkungen des Klimawandels über den Jahreszyklus einer Art hinweg gegenseitig beeinflussen können und wie eine Kombination von Methoden und Datenquellen einen besseren Einblick in die Auswirkungen des Klimawandels auf Zugvogelarten geben kann, was wiederum die Informationen für Schutzmaßnahmen für die Arten liefert.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The impacts of anthropogenic climate change are widespread across the globe, and now in many areas are contributing to significant declines in biodiversity (IPCC 2023; NOAA National Centers for Environmental Information 2023). Climate change is a complex phenomenon and can impact populations both directly and indirectly at any point of their annual cycle, including during the breeding season, non-breeding season, and migration. However, the majority of climate change vulnerability assessments (CCVA), and associated frameworks, exclusively consider the breeding season (Marra et al. 2015), and this is especially true for assessments concerning migratory species (Small-Lorenz et al. 2013; Marra et al. 2015; Culp et al. 2017; Nagy et al. 2022; Zhu et al. 2022). This has led to several calls for full-annual cycle assessments (Marra et al. 2015; Culp et al. 2017) and for methods to improve our understanding of vulnerability across a species’ annual cycle (Small-Lorenz et al. 2013; Trierweiler et al. 2014). To make effective conservation decisions, there is a need for species- and ecosystem-level assessments that identify where, when, and how biodiversity is likely to be at risk from climate change. Identifying present and future threats can provide key information on where action is most needed to increase population resilience and recovery in the face of climate change or, conversely, to identify areas that may not be viable in the future and, therefore, be of a lower priority for conservation in the present day. In this study, we draw together distribution modelling and tracking data to create a breeding and non-breeding assessment of exposure to climate change for an Afro-Palearctic migrant species, the European bee-eater (Merops apiaster).
European bee-eaters, hereafter bee-eaters, are long-distance migratory birds that breed in the Palearctic region, mainly along the Mediterranean, and spend the non-breeding season in sub-Saharan Africa (Fry 1984; Bastian and Bastian 2022). The species has recently expanded its breeding range northwards in central Europe (Schulze and Ortlieb 2010; Arbeiter et al. 2012), presumably benefiting from recent shifts in climate. In Europe, breeding occurs during late April to July, followed by autumn migration from August to September. Spring migration occurs from late March to late April (Hahn et al. 2020; Costa et al. 2021). Recent tracking data have shown bee-eaters follow several distinct migratory pathways from Europe to their non-breeding grounds in Africa. From western Europe, bee-eaters migrate across the western Sahara to the west coast and central Africa, and from eastern Europe, they migrate along the eastern coast of Africa to southern Africa (Dhanjal-Adams et al. 2018; Hahn et al. 2020; Costa et al. 2021). Bee-eaters’ core breeding and non-breeding areas are likely to undergo substantial changes because of climate change, including temperature and precipitation regimes (Coetzee et al. 2009; Stiels et al. 2021), and it is, therefore, possible that bee-eater populations may experience significant stress. Although the bee-eater is classed as Least Concern by the IUCN Red List (BirdLife International 2019), some southern Mediterranean populations are facing declines (e.g. in Iberia, Alonso et al. 2019; Escandell and Escudero 2019), leading to concerns about these populations’ viability under climate change. However, its vulnerability to climate change is likely to vary across its range and annual cycle; in addition, climate change may result in a variety of different and potentially interacting impacts. For example, climate change may result in an overall range reduction in one or several seasonal ranges; this is projected to be the case for many migratory species. Previous estimates have predicted that African Eurasian migratory waterbirds will suffer a 23% net reduction in breeding range by 2050 (Nagy et al. 2022). However, climate change can result in more complex changes to environmental conditions which can impact species across their annual cycle. For example, it can also cause shifts in phenology, and potential mismatches in resource availability, such as through mistimed arrival, breeding and departure from breeding ranges, and lack of access to optimum resources (Crick 2004; Both et al. 2006). In addition, impacts experienced in previous seasons are known to have “carry-over” effects on subsequent seasons, with breeding impacts happening outside the breeding range (Webster et al. 2002; Strandberg et al. 2010). The effects of climate change may be different at different non-breeding sites. Therefore, the effect on a given breeding population is determined not only by what is occurring in the breeding sites, but also by the conditions at the non-breeding sites that breeding populations rely on (Cresswell 2014). Frameworks that omit non-breeding ranges can therefore omit key sources of risk (Hostetler et al. 2015). Our study provides one example of how tracking data and modelling approaches can be used to link breeding and non-breeding areas, how climate change may affect populations at different stages of its annual cycle, and how these different impacts may interact. The bee-eater is a particularly suitable model species to explore this, as it presents high data coverage, available tracking data and current and past known range changes (Schulze and Ortlieb 2010; Arbeiter et al. 2012; Dhanjal-Adams et al. 2018; Hahn et al. 2020; Costa et al. 2021).
The overall aim of this study, using bee-eaters as a model species, is to develop a species distribution model (SDM) that includes the breeding and non-breeding range, and then overlay migratory connectivity (MC) information between different populations based on available tracking data. MC is a commonly used approach to analyse links between breeding and non-breeding areas (Webster et al. 2002). In this study, MC is defined as the likelihood of an individual to breed and winter at the same site as conspecifics from the breeding site (Webster et al. 2002), with the breeding and non-breeding areas defined prior to the analysis. Initially, a SDM was used to project the climatically suitable range for bee-eaters in 2081 to 2100 and compare the current and projected range to show where climate suitability is likely to decline, remain stable, or increase. Following this, existing data on connectivity between different breeding and non-breeding populations was used to supplement and verify the connectivity analysis based on raw tracking data, to identify how breeding populations may be impacted by changes in their non-breeding area and vice versa. In addition, populations with particularly high connectivity were highlighted, as these are likely particularly vulnerable to climate change, especially those that breed or winter in areas of high exposure. This study, therefore, provides an example framework to connect tracking data with correlative SDMs based on observation data to identify populations exposed to climate change throughout their life cycle.
Materials and methods
Selection and data preparation
Species occurrence data
Bee-eater species occurrence data were downloaded from the Global Biodiversity Information Facility (GBIF.org 2022). Records were obtained from the “Merops apiaster Linnaeus, 1758” occurrence records, and occurrences were limited to the years 2002 to 2022 (GBIF.org 2022). Data records were cleaned in R (for a full description of the data cleaning process see electronic supplementary material S1). Occurrence data were categorised into seasonal occurrences determined by the relevant months. This study defined May to August as the breeding period, hereafter breeding, and November to February as the non-breeding stationary period, hereafter non-breeding, as breeding and non-breeding months can vary between bee-eater populations because of different migration timings (Hahn et al. 2020; Costa et al. 2021), the selection of these months was intended to capture all relevant breeding and non-breeding areas. The breeding and non-breeding occurrence data were cropped to only include occurrences within the defined non-breeding and breeding regions. There were 70,015 occurrences used in the breeding range, and 9511 occurrences used in the non-breeding range after data cleaning.
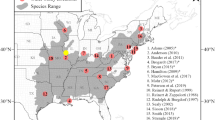
The breeding region was defined as northern Africa, and south and central Europe (Fig. 1). The non-breeding region was defined as sub-Saharan continental Africa (Fig. 1, Bastian and Bastian 2022). This restriction of the breeding and non-breeding range enabled the use of one best-fit model for both regions, resulting in higher accuracy of the projected models (Parding et al. 2020). These regions contain all major breeding and non-breeding sites in Europe and Africa (Dhanjal-Adams et al. 2018; Hahn et al. 2020; Costa et al. 2021). We verified that the occurrence data broadly matches the known distribution of breeding sites in Europe by comparing it to the European Breeding Bird Atlas 2 (Keller et al. 2020; EBCC 2022). To reduce sampling bias, as areas such as western and central Europe are more heavily monitored than areas like Eastern Europe, we conducted spatial filtering. The occurrence data were thinned to one occurrence per 0.1667-degree resolution, the same resolution used for climate data (10-min raster grids, Fick and Hijmans 2017), and within the same order of magnitude as the approximate home range size (Bastian et al. 2020). Records were not thinned to an equal record density across the breeding and non-breeding, as this would significantly reduce the data included in the model. However, by thinning to one occurrence per ten-minute grid, we counteract the high density of records in western Europe. While this does not fully compensate for sampling bias, it was judged to be preferable to losing a large portion of data to ensure equal sampling across areas. After occurrence data were thinned, the sample size was 8,171 for the breeding range, and 3,548 for the non-breeding range. All data processing, modelling and statistical analyses were conducted in R 4.2.2 (R Core Team 2022).
Overview of study regions and European Bee-eater distribution data used for analyses. Points in blue are occurrence data from global biodiversity information facility (GBIF) (after cropping and cleaning). The study regions were limited to continental Europe and Africa, with the breeding study region outlined in green, and the non-breeding study area outlined in orange
Geolocation tracking data
Geolocation tracking data were obtained by permission through Movebank (electronic supplementary material Table S1). Tracking data were collected from 2014 to 2017 across three different European breeding sites (n = 70) (Dhanjal-Adams et al. 2018; Hahn et al. 2020; Costa et al. 2021). These breeding sites were at the border of Portugal and Spain (Iberian Peninsula, n = 28), central Germany near Merseburg (n = 36), and Burgas, Bulgaria (n = 6). Bee-eaters were ringed and solar geolocators were attached to individuals (Dhanjal-Adams et al. 2018; Hahn et al. 2020; Costa et al. 2021).
Climate data
Climate data were obtained at a ten-minute resolution from WorldClim (electronic supplementary material Table S2, Fick and Hijmans 2017). Temperature, precipitation, and vegetation are important factors that define bee-eater distribution (Fry 1984; Arbeiter et al. 2016), and previous work has demonstrated precipitation, temperature and vegetation cover can strongly affect breeding success and, to a lesser extent, survival (Eckhoff 2012; Arbeiter et al. 2016; Gordo et al. 2021; Stiels et al. 2021; Bastian and Bastian 2022). As a result, several temperature and precipitation variables were selected that either relate directly to bee-eater physiological preferences, or that correlate broadly to vegetation indices and therefore prey availability. Several vegetation indices were trialled (such as NDVI) for inclusion, but were ultimately excluded due to a lack of projections that cover the study area. Instead, several bioclimatic variables were selected that have previously been found to act as NDVI proxies (Liu et al. 2015; Jennings and Harris 2017; Ma et al. 2022). From a long list of candidate variables, we filtered out variables that showed little relevance or correlation to bee-eater presence, as well as removing variables that showed high co-variance (see electronic supplementary material S2 for full details). “Annual” variables refer to climatic variables averaged over an entire year or a predefined season (e.g. the hottest quarter), and are not specific to the breeding or non-breeding season (Fick and Hijmans 2017). “Seasonal” variables refer to climatic variables averaged over defined months of the year, either the breeding or non-breeding months in a given area. Annual variables included in this study were: mean diurnal range, isothermality, temperature seasonality, mean temperature of the wettest quarter, mean temperature of the driest quarter, precipitation seasonality (Fick and Hijmans 2017). Seasonal variables included in this study were: seasonal maximum temperature and seasonal precipitation. Several sets of variables were trialled in addition to those listed above, but were excluded; for a full description of candidate variables and methodology, see electronic supplementary material S2.
The retained variables were considered potential predictors of bee-eater range due to one of the following reasons. Firstly, candidate variables may affect bee-eater distribution due to their direct effects during the breeding and non-breeding season, either related to their physiological relevance, or due to their impact on vegetation (Karnieli et al. 2019), and prey availability during the relevant season (Arbeiter et al. 2014, 2016); seasonal maximum temperature and seasonal precipitation were included under this rationale. Secondly, candidate variables may act as correlates of vegetation density and cover during the breeding and non-breeding season, and therefore insect abundance, due to their effects over the course of the year (Chamaille-Jammes et al. 2006; Touhami et al. 2022), including times when bee-eaters are not present. A number of variables have been found in previous studies to correlate to vegetation indices, including mean diurnal range, isothermality, temperature seasonality, and precipitation seasonality, mean temperature of the wettest quarter and mean temperature of the driest quarter (Liu et al. 2015; Jennings and Harris 2017; Ma et al. 2022), so these variables were retained for investigation.
Climate data were collated for the present day (averaged from 1970 to 2000) and for the future (averaged projections from 2081 to 2100) using the projected model CMIP6.MRI_ESM2_0. The projected year range was 2081 to 2100 and the selected Shared Socio-economic Pathway was 370 (SSP370); the “middle-road” carbon emission scenario (O’Neill et al. 2014, 2017; Riahi et al. 2017). Climate variables were normalised to a standardised scale with a mean of zero and a deviation of one, using the “scale” function in R. For a full description on how climate change models and SSP were selected and processed, please see electronic supplementary material S3.
Species distribution models
SDMs were constructed for breeding and non-breeding regions using the biomod2 package (Thuiller et al. 2022). To assess model sensitivity, five SDM algorithms were used, generalised linear models (GLM), generalised additive models (GAM), multivariate adaptive regression spline (MARS), random forests (RF) and generalised boosted models (GBM). Pseudo-absences were generated at a 1:1 ratio with the occurrence data, using the “disk” strategy to randomly generate pseudo-absences at a minimum distance of 10,000 m from another occurrence. In total, absence/pseudo-absence sets were generated five times. SDMs were filtered using the true skill statistic (TSS, Allouche et al. 2006) as the evaluation metric, where a model below a TSS threshold of 0.5 was discarded and not used further for ensemble building. All other model settings were the default option as defined by biomod2. SDM uncertainty was estimated using the coefficient of variation across predictions. Pearson’s correlation coefficient was used to assess variable importance across all algorithms, where each variable is dropped in turn and replaced with a “noise” variable to assess a variable’s overall importance on model fit and predictive capacity. Ten permutations were used to estimate variable importance for each SDM. Final ensemble SDMs were produced as weighted mean ensembles for the breeding and non-breeding regions and then projected to 2081 to 2100. The current and future weighted ensembles were used to calculate the change in climate suitability within the breeding and non-breeding regions.
Model validation was conducted using an 80% split SDM, where models are built with 80% of occurrence data and then tested with the remaining 20%. This validation approach was replicated five times. Overall model performance was tested using the metrics TSS, and the area under the curve (AUC, Fielding and Bell 1997), the latter found by examining the receiver operating characteristics (ROC). Over the five replicates, the validation metrics were produced as mean and standard deviation to give an overall metric of performance.
Migratory connectivity analysis
Breeding and non-breeding sites were obtained for MC analysis by isolating the first coordinates in July (deployment of geolocators at the breeding colonies) and the last coordinates before spring migration to give the non-breeding site (Hahn et al. 2019; Costa et al. 2021). Due to incomplete data records in February, the last coordinates in January were used. For MC analyses, boundaries for each separate population need to be defined. For the breeding season, we created polygons around the isolated coordinates, which were grouped based on previous knowledge of disjunct breeding populations (Dhanjal-Adams et al. 2018). Thus, breeding site areas were defined as Iberia, Germany, and Bulgaria. While these are not the only populations in Europe, these provide three examples of separate breeding populations to allow comparison of migratory routes. For the non-breeding site areas, polygons were created around the isolated coordinates separated by country. Polygons that lay in adjacent countries were then joined together to form the final set of non-breeding polygons. A few coordinates were classed as outliers, where the final position was defined as offshore. If these outliers lay within a 50 km buffer of a non-breeding region, they were included in the non-breeding region, otherwise they were discarded as potential errors. Non-breeding site areas were therefore defined as West Coast of Africa (hereafter West Coast), Côte d'Ivoire, Nigeria, Central Africa, and Southern Africa. For this analysis, the “origin points” were defined as the breeding regions, and the “target points” as the non-breeding regions. Average distances between breeding and non-breeding regions were calculated using the centre coordinates of the breeding and non-breeding regions.
MC analysis was conducted using the MigConnectivity package (Hostetler et al. 2021), following the methods developed by Cohen et al. (2018). Relative abundance was defined as even between the origin regions, as the tracking data are not likely to be proportional to the population size of each region. This introduces a potential source of bias, as in reality it is likely these populations are not of equal size. Therefore, an additional sensitivity analysis was conducted, in which the relative abundance was defined on the estimated population size from each breeding region according to the breeding occurrence data before thinning. The relative abundance used for this was 0.92 (Iberia), 0.041 (Germany), and 0.039 (Bulgaria). 100 bootstrap iterations were used to estimate the overall migration connectivity and the specific breeding transition probabilities. Overall MC was estimated as the mean of all results, and the transition probabilities (psi) were calculated for specific breeding populations (Cohen et al. 2018). Connectivity estimates were calculated with a small sample size correction, available in the MigConnectivity package (Hostetler et al. 2021).
Results
Species distribution models
Validation statistics
Generally, the algorithms performed well, with non-breeding models performing better than breeding models on mean average. Aside from GAM, which was removed from ensemble models due to poor performance (Table 1, TSS = 0.323 and AUC = 0.624), all breeding algorithms showed TSS > 0.5 and AUC > 0.8 (Table 1), and were designated as well-performing models (using thresholds as set in Allouche et al. 2006; Fielding and Bell 1997). For non-breeding models, all models showed TSS > 0.5 and AUC > 0.8 (Table 1). RF was the strongest performing model in both the breeding (TSS = 0.667, and AUC = 0.910) and the non-breeding (TSS = 0.746, and AUC = 0.940). Final weighted mean ensembles for the current climate suitability performed well with TSS > 0.8, and AUC > 0.8 in breeding (TSS = 0.860 and AUC = 0.824) and non-breeding ensembles (TSS = 0.886 and AUC = 0.871).
Climate variable importance
Variables with high importance (mean Pearson’s coefficient ≥ 0.2 in multiple algorithms) in the breeding season were seasonal maximum temperature, seasonal precipitation and isothermality (Fig. 2a). Seasonal maximum temperature had a Pearson’s coefficient > 0.2 in all models except GAM (0.169), ranging from 0.203 (RF) to 0.426 (GBM), the optimum range being 25 °C to 30 °C (electronic supplementary material Fig. S1). Seasonal precipitation had a Pearson’s coefficient > 0.4 in MARS, GAM and GLM, ranging from 0.413 (MARS) to 0.673 (GAM), the optimum range being ≤ 75 mm (electronic supplementary material Fig. S1). Isothermality had a Pearson’s coefficient > 0.2 in GLM, GAM, and MARS, ranging from 0.209 (MARS) to 0.399 (GLM), with optimum range being 30% to 40% (electronic supplementary material Fig. S1). Precipitation of the warmest quarter had a high coefficient value in two of the five algorithms (GLM = 0.346 and GAM = 0.355) and a mean of 0.186. For full model results see electronic supplementary material (Fig. S2).
Importance of highest-ranking climate variables in final ensemble species distribution models (SDM). Note that only variables with a Pearson’s correlation of above 0.15 are shown here. A The four most important variables for the breeding season, namely isothermality, precipitation of the warmest quarter (PrecWQ), seasonal precipitation (SPrec) and seasonal maximum temperature (STmax). B The three most important variables for the non-breeding season, namely mean temperature of the driest quarter (MeanTDQ), mean temperature of the wettest quarter (MeanTWQ), and seasonal precipitation (SPrec). Variable importance was estimated using Pearson’s Correlation based on ensemble SDM outputs. Variable importance has been averaged from across the five model algorithms used (multivariate adaptive regression spline (MARS), generalised boosted models (GBM), generalised additive models (GAM), generalised linear models (GLM), and random forests (RF))
In the non-breeding region, the most important variables were mean temperature of the driest quarter, mean temperature of the wettest quarter, and seasonal precipitation (Fig. 2b). For mean temperature of the driest quarter, Pearson’s coefficient ranged from 0.249 (RF) to 0.786 (GLM), with the optimum range being 10 °C to 15 °C (electronic supplementary material Fig. S1). RF was an outlier compared to other algorithms, and its removal narrowed this range from 0.431 (MARS) to 0.786 (GLM). Mean temperature of the wettest quarter (ranging from 0.276–0.360) had high importance, but only in some models, namely in GLM, GAM, and MARS. Although seasonal precipitation had a mean of 0.190, it had a high importance in three of the five algorithms (GAM (0.246), MARS (0.227), and GBM (0.201)), with an optimum value of 100 mm (electronic supplementary material Fig. S1). For full model results see electronic supplementary material (Fig. S2).
Ensemble models and climate change projections
All individual models that demonstrated performance above the TSS threshold (TSS > 0.5) were incorporated into the final ensemble models, where models were weighted by performance. Model variance was checked visually for any areas with very high variance in predictions (see electronic supplementary material Fig. S3), and then models were projected to 2080 to 2100 climatic conditions. The future projections for both regions show significant shifts in suitability compared to the current ensemble models (Fig. 3), with some areas showing significant declines in suitability while others are likely to increase in suitability. There are projected overall declines across northwest Europe and southwest Europe, most notably within the Iberian Peninsula. Within the projected non-breeding ensemble, there is a decline in climate suitability around the southern cape of South Africa, and a prominent decline on the West Coast, and in the region of Senegal. Declines are also projected in Tanzania and Kenya region (Fig. 3f). Climate suitability is projected to increase within central Africa and along the West Coast in the Côte d’Ivoire region (Fig. 3f).
Summary maps of climate suitability for the present data and for 2080 to 2100, from the Shared Socio-economic Pathway 370 (SSP370), across the European Bee-eater breeding and non-breeding region based on SDM ensemble models. A Present-day climate suitability in the breeding region. B Projected climate suitability in the breeding region in 2080 to 2100. C Difference between current and future suitability in the breeding region. D Present-day climate suitability in the non-breeding region. E Projected climate suitability in the non-breeding region in 2080 to 2100. F Difference between current and future suitability in the non-breeding region
Migratory connectivity analysis
Mean MC across all populations was 0.923 (SD ± 0.001), based on a zero to one scale, demonstrating high overall MC between the breeding and non-breeding areas. Broadly, our results confirm other tracking studies (Dhanjal-Adams et al. 2018; Hahn et al. 2020; Costa et al. 2021). Iberian breeding populations mostly migrated to the West Coast area, the German population mostly migrated to central Africa, and the Bulgarian population mostly migrated to southern Africa (Fig. 4). However, the transition probabilities varied substantially. The breeding population in Bulgaria, that used South Africa as the non-breeding site, had the highest mean transition probability (psi) of 1 (SE ± 0.00), and the German breeding population that spent the non-breeding period in Central Africa was also extremely high (psi = 0.971, SE ± 0.029, Table 2). The Iberian breeding population had a psi = 0.571 (SE ± 0.091) in connection with the West Coast non-breeding site, but also showed weak connectivity between Côte d’Ivoire (psi = 0.236, SE ± 0.081) and Nigeria (psi = 0.193, SE ± 0.079). The mean MC for the sensitivity analysis was shown to be 0.835 (SD ± 0.032) and further results are presented in electronic supplementary material Table S3. The shift in mean MC is likely because the Iberian population is by far the most numerous in our dataset, and has the lowest MC. However, the mean MC is still high even with extremely biased weighting, and so as not to let the Iberian population dominate the weighting (which would introduce error as our sensitivity weighting is likely not to reflect real life population size), we used the initial equal weighting model.
Summary of European Bee-eater migration routes from breeding to non-breeding sites. Individual tracking routes are given as points across the map, with the colours corresponding to the breeding location and non-breeding location. Routes containing four or more individuals, going from the same breeding to non-breeding locations have been averaged by month, starting from the breeding location to the non-breeding location and back to the breeding location. The averaged migration routes have been overlayed over the individual tracking routes. The only migration route this excludes is the Germany/Cote d’Ivoire route, which contained data from two individuals, and the tracking data from one of these individuals is incomplete. The averaged migration routes used from breeding sites in Iberia, Germany, and Bulgaria, coloured in green, to non-breeding sites in West Coast Africa, Côte d’Ivoire, Nigeria, Northern Angola, and Southern Africa (respectively), coloured in orange. All individuals were tracked with solar geolocators
Discussion
The potential risk of climate change to breeding bee-eater populations has been previously highlighted (Stiels et al. 2021), but this is the first time that both the breeding and non-breeding range of the bee-eater has been assessed in conjunction. Notably, we found that the Iberian population may be at risk from multiplicative effects of climate change, as both its breeding and main non-breeding sites (on the West Coast) are projected to decline in climatic suitability under the projected changes. Indeed, Iberia is flagged as a hotspot for climate change (IPCC 2023), where extreme events such as heat waves and droughts are becoming more frequent. Increased maximum temperatures have been shown to have a negative effect on arid-zone species’ breeding success (van de Ven et al. 2020; Bourne et al. 2021), with Costa et al. (2021) describing a decrease in Iberian bee-eater breeding success after a heat wave. Here, a decrease in climate suitability together with other factors such as agriculture intensification, pesticide use and disappearance or destruction of suitable nesting sites at local scales (Dellwisch et al. 2021; Gordo et al. 2021) may be potentially contributing to the current decreasing trend of the Iberian bee-eater population (Alonso et al. 2019; Escandell and Escudero 2019; Gordo et al. 2021). Based on the species’ past behaviour in colonising central Germany, it is possible that bee-eaters may respond to declines in climate suitability by shifting breeding and non-breeding sites to more suitable areas (Arbeiter et al. 2012). This shift may be accompanied by a shift in phenology, as arrival date of bee-eaters in central Europe is correlated to day length and environmental cues (Bastian and Bastian 2014), however, this behaviour is difficult to predict and bee-eater species outside of Europe have not shown similar shifts in range or behaviour (Gholamhosseini and Baharlu 2021). For the west African non-breeding populations, this may involve a shift to more suitable sites in Côte d’Ivoire and Central Africa, and/or corresponding shifts in breeding area to northern Iberia and southern France. However, shifting sites in this manner would increase migration length and, potentially, the risk of adult mortality during migration (Strandberg et al. 2010; Klaassen et al. 2014). It could also adversely affect individual fitness or change migration timings (e.g., arrival to the breeding site or non-breeding sites; Bussière et al. 2015) leading to delayed laying and/or lower breeding success (Costa et al. 2021), and ultimately, to severe changes in population demography. Therefore, climate change could have substantial negative impacts on the Iberian population of bee-eaters, even if the population has the capacity to shift its breeding or non-breeding ranges. As a result, this study projects on the SSP370, or “middle road” carbon emissions scenario, that the breeding and main non-breeding sites of the Iberian population is likely to decline in climate suitability.
Our results showed that some breeding sites will increase in suitability under climate change, such as in central and northern Europe, particularly the German population, which has also been suggested by previous projections (Dellwisch et al. 2021). Considering the positive effect that non-breeding conditions can have on breeding (Strandberg et al. 2010; Hostetler et al. 2015), the projected increase in non-breeding climatic suitability in central Africa could increase individual fitness, spring migration survival and breeding success (Strandberg et al. 2010; Paxton and Moore 2015), suggesting that the German population could remain stable or continue to increase provided that no other factors negatively affect this population. Our results highlight the importance of isothermality, seasonal precipitation and seasonal maximum temperature during the breeding season. Indeed, maximum temperature and precipitation have been highlighted to impact several breeding parameters (e.g. insect availability and brood size) of the German population (Arbeiter et al. 2016; Dellwisch et al. 2021), and the migration routes of bee-eaters around the Caspian Sea (Vilkov 2016). Considering the projected decline in climate suitability within southern Europe, and the bee-eater’s role as an ecological engineer, conservation of these areas would maintain a stable population of bee-eaters within central Europe, which would also benefit other species that could use old nests left from previous years, such as the Eurasian Hoopoe (Upupa epops), Little Owl (Athene noctua) and House Sparrow (Passer domesticus) (Casas-Crivillé and Valera 2005).
High MC implies that populations migrate from the same breeding sites to the same non-breeding sites, leading to little population cross over between different breeding sites. Species with low MC that lose one non-breeding site may see an overall decline in population, but this may be mitigated to some extent by the use of alternative non-breeding sites. In contrast, high MC is likely to result in an increased impact of climate change on breeding populations, as if one non-breeding site is heavily impacted, there is less likely to be capacity to shift to a secondary non-breeding site that can support the breeding population (Webster et al. 2002; Taylor and Stutchbury 2016). Given that bee-eaters are known to migrate with conspecifics from the same colony, displaying a fission–fusion dynamic throughout (Dhanjal-Adams et al. 2018), the observed high MC was expected. The relatively low mean transition probability of the Iberian breeding population, and its mean transition probabilities with the Côte d’Ivoire and Nigeria suggests that this population may be able to shift its non-breeding site or compensate to some extent for any changes to the climate suitability at its primary non-breeding site. Bee-eaters have been recorded colonising new breeding areas and shifting in response to accessibility to resources (Arbeiter et al. 2012). Whilst research demonstrates that there is a significant difference in the foraging range of bee-eaters during and post-breeding within the breeding range (Bastian et al. 2020), there is limited understanding regarding how bee-eaters utilise space in the non-breeding range. Evidence suggests that whilst non-breeding sites are more sparsely spread (Brooke and Herroelen 1988; Dhanjal-Adams et al. 2018), some bee-eaters do use multiple non-breeding sites (Hahn et al. 2020; Costa et al. 2021), but this is still not fully understood. The low mean transition probability of the Iberian population in comparison to the Bulgarian and German populations, suggests that the Iberian population may react adaptively to locate sites of higher suitability under climate change, which may be particularly relevant as the Iberian population is projected to experience the highest overall decrease in climatic suitability. However, caution should be used when applying climate change suitability changes to population-level projections. An observed or projected change in key climate variables may not necessarily result in proportional, or consistent, responses in abundance or range size across species or populations (Howard et al. 2023). In addition, while this analysis used as much tracking data as was available, it is still a small sample size relative to the total size of the population, and therefore the results shown here could be either an overestimate or underestimate of populations’ migratory adaptive capacity.
There are a number of limitations to the approach used here that should be considered during interpretation. We exclusively used climatic variables, which typically describe species ranges well at a broad scale, but did not include other potential key factors such as habitat substrate, land use, or vegetation factors. Thus, it is possible that key drivers of habitat suitability in bee-eaters were missed. For example, normalised difference vegetation index (NDVI) is likely a key factor that influences bee-eater distribution, especially during the non-breeding season, and was not included in this analysis. This was due to data availability issues, and we compensated by including bioclimatic variables that were selected to act as proxy variables of NDVI, such as mean diurnal range, isothermality, temperature seasonality, and precipitation seasonality (Ma et al. 2022). In addition, we may not have captured the full climatic tolerances of bee-eaters as only continental Europe and Africa were considered as breeding and non-breeding areas, respectively, in this study. This was due to data availability constraints, 94.9% of the occurrences recorded in GBIF (GBIF.org 2022) are within these areas, as well as to broadly matching the available tracking data. It should be noted bee-eaters have a very large range, and in the past three decades have been recorded expanding this range into Scandinavia and the UK, alongside a known non-breeding population in southern India. It is possible therefore that their climatic tolerance extends further than was captured in this study. In addition, the overall ability of bee-eaters to move between sites during the non-breeding season is poorly understood, and to assume populations are sedentary once they arrive, as we did in our models, may be in error. However, as there is little data available for the non-breeding season, and due to low temporal resolution and precision in tracking data, we therefore assumed that the last coordinates were representative of the overall non-breeding area.
Identifying ecosystems and species most at risk from climate change is a key conservation priority, and a number of techniques have been developed to identify at-risk species, areas or ecosystems. SDMs are one such commonly used tool, to spatially and temporally quantify exposure to climate change. In correlative SDMs, this is done by identifying the environmental conditions that correlate strongly with the probability of a given species presence or abundance, and then by using climate change projections to predict where areas may become less (or more) suitable in the future. These metrics of exposure are frequently used as a key component of conservation assessments, such as in CCVA (Lankford et al. 2014; Culp et al. 2017; Rempel and Hornseth 2017), which can provide a broad overview of the likely exposure, sensitivity, and potential for adaptation for the target species or ecosystems. However, several contexts are challenging when using conventional SDM approaches, in particular for data deficient species, areas and ecosystems (van Proosdij et al. 2016; Guillaumot et al. 2019) and for species that have distinct life stages or migratory behaviour, for which dynamic methods are called for (Runge et al. 2016; Zuckerberg et al. 2016). These deficiencies undermine vulnerability assessments for a variety of species but are particularly pressing for migratory birds (Robinson et al. 2009; Albers et al. 2023). Migratory species comprise 16% of known bird species (Robinson et al. 2009), and can be particularly vulnerable to climate change (Robinson et al. 2009; Small-Lorenz et al. 2013; Marra et al. 2015). This gap in available frameworks and methodology fundamentally hinders efforts to identify and conserve migratory birds in the face of climate change. Therefore, assessments of vulnerability need to consider these connections and interacting impacts, using novel approaches and multiple sources of information; our approach of combining tracking data and conventional correlative models provide one method of doing so using established datasets and methods. By including data on bee-eaters’ non-breeding ranges, and by using associated tracking data, we have highlighted potential risk to some populations that would have otherwise been missed if we had focussed just on breeding range. This study therefore provides information on year-round exposure of bee-eaters to climate change and reinforces the identified need of assessments that include both the breeding and non-breeding ranges of migratory species.
Data availability
All data used in this study are available within online repositories. Climate data analysed during the current study are available from WorldClim, and can be accessed from https://www.worldclim.org/data/worldclim21.html and https://www.worldclim.org/data/cmip6/cmip6_clim10m.html. All M. apiaster occurrence data used in this study are available from GBIF, the specific occurrence download dataset can be accessed at https://doi.org/10.15468/dl.juskee. The tracking data analysed during the current study are available upon request from the Movebank online database, (https://www.movebank.org/, Movebank IDs: 502110670, 759031657, 753257610, 1416270261, and 1416275401).
References
Albers HJ, Lee KD, Martínez-Salinas A et al (2023) How ecological characteristics of seasonal migratory species influence conservation threats and policy needs. Rev Environ Econ Policy 17:91–110. https://doi.org/10.1086/723886
Allouche O, Tsoar A, Kadmon R (2006) Assessing the accuracy of species distribution models: prevalence, kappa and the true skill statistic (TSS). J Appl Ecol 43:1223–1232. https://doi.org/10.1111/j.1365-2664.2006.01214.x
Alonso H, Coelho R, Costa J et al (2019) Relatório do Censo de Aves Comuns 2004–2018. Sociedade Portuguesa para o Estudo das Aves, Lisboa
Arbeiter S, Schulze M, Todte I, Hahn S (2012) Das Zugverhalten und die Ausbreitung von in Sachsen-Anhalt brütenden Bienenfressern (Merops apiaster). Ber Vogelwarte Hiddensee 21:33–40
Arbeiter S, Schnepel H, Uhlenhaut K et al (2014) Seasonal shift in the diet composition of European Bee-Eaters Merops apiaster at the Northern edge of distribution. Ardeola 61:161–170. https://doi.org/10.13157/arla.61.1.2014.161
Arbeiter S, Schulze M, Tamm P, Hahn S (2016) Strong cascading effect of weather conditions on prey availability and annual breeding performance in European Bee-eaters Merops apiaster. J Ornithol 157:155–163. https://doi.org/10.1007/s10336-015-1262-x
Bastian H-V, Bastian A (2014) Maiwitterung bestimmt Erstankunft des Bienenfressers (Merops apiaster) in einer rheinland-pfälzischen Brutkolonie. Vogelwarte 52:169–174
Bastian H-V, Bastian A, Essel S, Tietze DT (2020) Space use and daily movement patterns of the European Bee-Eater Merops apiaster during breeding and post-breeding. Ardea 107:321–327. https://doi.org/10.5253/arde.v107i3.a6
Both C, Bouwhuis S, Lessells CM, Visser ME (2006) Climate change and population declines in a long-distance migratory bird. Nature 441:81–83. https://doi.org/10.1038/nature04539
Bourne AR, Ridley AR, Spottiswoode CN, Cunningham SJ (2021) Direct and indirect effects of high temperatures on fledging in a cooperatively breeding bird. Behav Ecol 32:1212–1223. https://doi.org/10.1093/beheco/arab087
Brooke RK, Herroelen P (1988) The nonbreeding range of southern African bred European Bee-eaters Merops apiaster. Ostrich 59:63–66. https://doi.org/10.1080/00306525.1988.9633698
Bussière EMS, Underhill LG, Altwegg R (2015) Patterns of bird migration phenology in South Africa suggest northern hemisphere climate as the most consistent driver of change. Glob Chang Biol 21:2179–2190. https://doi.org/10.1111/gcb.12857
Cahill AE, Aiello-Lammens ME, Fisher-Reid MC et al (2013) How does climate change cause extinction? Proc Royal Soc B 280:20121890. https://doi.org/10.1098/rspb.2012.1890
Casas-Crivillé A, Valera F (2005) The European bee-eater (Merops apiaster) as an ecosystem engineer in arid environments. J Arid Environ 60:227–238. https://doi.org/10.1016/j.jaridenv.2004.03.012
Chamaille-Jammes S, Fritz H, Murindagomo F (2006) Spatial patterns of the NDVI–rainfall relationship at the seasonal and interannual time scales in an African savanna. Int J Remote Sens 27:5185–5200. https://doi.org/10.1080/01431160600702392
Coetzee BWT, Robertson MP, Erasmus BFN et al (2009) Ensemble models predict important bird areas in southern Africa will become less effective for conserving endemic birds under climate change. Glob Ecol Biogeogr 18:701–710. https://doi.org/10.1111/j.1466-8238.2009.00485.x
Cohen EB, Hostetler JA, Hallworth MT et al (2018) Quantifying the strength of migratory connectivity. Methods Ecol Evol 9:513–524. https://doi.org/10.1111/2041-210X.12916
Costa JS, Hahn S, Araújo PM et al (2021) Linking migratory performance to breeding phenology and productivity in an Afro-Palearctic long-distance migrant. Sci Rep 11:23258. https://doi.org/10.1038/s41598-021-01734-0
Cresswell W (2014) Migratory connectivity of Palaearctic-African migratory birds and their responses to environmental change: the serial residency hypothesis. Ibis 156:493–510. https://doi.org/10.1111/ibi.12168
Crick HQP (2004) The impact of climate change on birds. Ibis 146:48–56. https://doi.org/10.1111/j.1474-919X.2004.00327.x
Culp LA, Cohen EB, Scarpignato AL et al (2017) Full annual cycle climate change vulnerability assessment for migratory birds. Ecosphere 8:e01565. https://doi.org/10.1002/ecs2.1565
Dellwisch B, Bastian A, Bastian H-V et al (2021) Bienenfresser Merops apiaster in Deutschland – woher sie kamen, wohin sie gehen. Vogelwarte 59:197–206
Dhanjal-Adams KL, Bauer S, Emmenegger T et al (2018) Spatiotemporal group dynamics in a long-distance migratory bird. Curr Biol 28:2824-2830.e3. https://doi.org/10.1016/j.cub.2018.06.054
Eckhoff P (2012) Wiederholte Brutansiedlungen des Bienenfressers Merops apiaster im Landkreis Oberhavel. Otis 20:77–81
Escandell V, Escudero E (2019) Tendencia de las aves en primavera. Programas de seguimiento y grupos de trabajo de SEO, London
Fick SE, Hijmans RJ (2017) WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. J Climatol 37:4302–4315. https://doi.org/10.1002/joc.5086
Fielding AH, Bell JF (1997) A review of methods for the assessment of prediction errors in conservation presence/absence models. Environ Conserv 24:38–49. https://doi.org/10.1017/S0376892997000088
Fry H (1984) The Bee-eaters. Poyser, Epsom
Gholamhosseini A, Baharlu F (2021) Assessment of climate change impacts on geographic distribution of Merops orientalis in southern Iran: a case study in Fars province. J Anim Res 34:98–109
Gordo O, Aymí R, Molina B (2021) Der Bienenfresser Merops apiaster in Spanien: seine Verbreitung, Häufigkeit und Bedrohung. Vogelwarte 59:235–245
Guillaumot C, Artois J, Saucède T et al (2019) Broad-scale species distribution models applied to data-poor areas. Prog Oceanogr 175:198–207. https://doi.org/10.1016/j.pocean.2019.04.007
Hahn S, Alves JA, Bedev K et al (2020) Range-wide migration corridors and non-breeding areas of a northward expanding Afro-Palaearctic migrant, the European Bee-eater Merops apiaster. Ibis 162:345–355. https://doi.org/10.1111/ibi.12752
Hostetler JA, Sillett TS, Marra PP (2015) Full-annual-cycle population models for migratory birds. Auk 132:433–449. https://doi.org/10.1642/AUK-14-211.1
Howard C, Marjakangas EL, Morán-Ordóñez A et al (2023) Local colonisations and extinctions of European birds are poorly explained by changes in climate suitability. Nat Commun 14:4304. https://doi.org/10.1038/s41467-023-39093-1
IPCC (2023) A report of the intergovernmental panel on climate change. Contribution of working groups I, II and III to the sixth assessment report of the intergovernmental panel on climate change. IPCC, Geneva
Jennings MD, Harris GM (2017) Climate change and ecosystem composition across large landscapes. Landsc Ecol 32:195–207. https://doi.org/10.1007/s10980-016-0435-1
Karnieli A, Ohana-Levi N, Silver M et al (2019) Spatial and seasonal patterns in vegetation growth-limiting factors over Europe. Remote Sens 11:2406. https://doi.org/10.3390/rs11202406
Keller V, Herrando S, Voříšek P et al (2020) European breeding bird atlas 2: distribution, abundance and change. European Bird Census Council & Lynx Edicions, Barcelona
Klaassen RHG, Hake M, Strandberg R et al (2014) When and where does mortality occur in migratory birds? Direct evidence from long-term satellite tracking of raptors. J Anim Ecol 83:176–184. https://doi.org/10.1111/1365-2656.12135
Lankford AJ, Svancara LK, Lawler JJ, Vierling K (2014) Comparison of climate change vulnerability assessments for wildlife. Wildl Soc Bull 38:386–394. https://doi.org/10.1002/wsb.399
Liu Y, Li Y, Li S, Motesharrei S (2015) Spatial and temporal patterns of global NDVI trends: correlations with climate and human factors. Remote Sens 7:13233–13250. https://doi.org/10.3390/rs71013233
Ma B, Zeng W, Hu G et al (2022) Normalized difference vegetation index prediction based on the delta downscaling method and back-propagation artificial neural network under climate change in the Sanjiangyuan region. China Ecol Inform 72:101883. https://doi.org/10.1016/j.ecoinf.2022.101883
Marra PP, Cohen EB, Loss SR et al (2015) A call for full annual cycle research in animal ecology. Biol Lett 11:20150552. https://doi.org/10.1098/rsbl.2015.0552
Nagy S, Breiner FT, Anand M et al (2022) Climate change exposure of waterbird species in the African-Eurasian flyways. Bird Conserv Int 32:1–26. https://doi.org/10.1017/S0959270921000150
NOAA National Centers for Environmental Information (2023) Monthly global climate report for annual 2022. NOAA, Washington, DC
O’Neill BC, Kriegler E, Riahi K et al (2014) A new scenario framework for climate change research: the concept of shared socioeconomic pathways. Clim Change 122:387–400. https://doi.org/10.1007/s10584-013-0905-2
O’Neill BC, Kriegler E, Ebi KL et al (2017) The roads ahead: narratives for shared socioeconomic pathways describing world futures in the 21st century. Glob Environ Change 42:169–180. https://doi.org/10.1016/j.gloenvcha.2015.01.004
Parding KM, Dobler A, McSweeney CF et al (2020) GCMeval – An interactive tool for evaluation and selection of climate model ensembles. Clim Serv 18:100167. https://doi.org/10.1016/j.cliser.2020.100167
Paxton KL, Moore FR (2015) Carry-over effects of winter habitat quality on en route timing and condition of a migratory passerine during spring migration. J Avian Biol 46:495–506. https://doi.org/10.1111/jav.00614
R Core Team (2022) R: A language and environment for statistical computing. R Core Team, Geneva
Rempel RS, Hornseth ML (2017) Binational climate change vulnerability assessment of migratory birds in the Great Lakes Basins: tools and impediments. PLoS ONE 12:e0172668. https://doi.org/10.1371/journal.pone.0172668
Riahi K, van Vuuren DP, Kriegler E et al (2017) The shared socioeconomic pathways and their energy, land use, and greenhouse gas emissions implications: an overview. Glob Environ Change 42:153–168. https://doi.org/10.1016/j.gloenvcha.2016.05.009
Robinson R, Crick H, Learmonth J et al (2009) Travelling through a warming world: climate change and migratory species. Endanger Species Res 7:87–99. https://doi.org/10.3354/esr00095
Runge CA, Tulloch AIT, Possingham HP et al (2016) Incorporating dynamic distributions into spatial prioritization. Divers Distrib 22:332–343. https://doi.org/10.1111/ddi.12395
Schulze M, Ortlieb R (2010) Bestand, Schutz und Gefährdung des Bienenfressers (Merops apiaster) in Sachsen-Anhalt. Naturschutz Im Land Sachsen-Anhalt 47:3–15
Small-Lorenz SL, Culp LA, Ryder TB et al (2013) A blind spot in climate change vulnerability assessments. Nat Clim Chang 3:91–93. https://doi.org/10.1038/nclimate1810
Stiels D, Bastian H-V, Bastian A et al (2021) An iconic messenger of climate change? Predicting the range dynamics of the European Bee-eater (Merops apiaster). J Ornithol 162:631–644. https://doi.org/10.1007/s10336-021-01867-z
Strandberg R, Klaassen RHG, Hake M, Alerstam T (2010) How hazardous is the Sahara Desert crossing for migratory birds? Indications from satellite tracking of raptors. Biol Lett 6:297–300. https://doi.org/10.1098/rsbl.2009.0785
Taylor CM, Stutchbury BJM (2016) Effects of breeding versus winter habitat loss and fragmentation on the population dynamics of a migratory songbird. Ecol Appl 26:424–437. https://doi.org/10.1890/14-1410
Thuiller W, Georges D, Gueguen M et al (2022) biomod2: Ensemble platform for species distribution modelling. Wilfried Thuiller, Grenoble
Touhami I, Moutahir H, Assoul D et al (2022) Multi-year monitoring land surface phenology in relation to climatic variables using MODIS-NDVI time-series in Mediterranean forest. Northeast Tunisia Acta Oecologica 114:103804. https://doi.org/10.1016/j.actao.2021.103804
Trierweiler C, Klaassen RHG, Drent RH et al (2014) Migratory connectivity and population-specific migration routes in a long-distance migratory bird. Proc Royal Soc B 281:20132897. https://doi.org/10.1098/rspb.2013.2897
van de Ven TMFN, McKechnie AE, Er S, Cunningham SJ (2020) High temperatures are associated with substantial reductions in breeding success and offspring quality in an arid-zone bird. Oecologia 193:225–235. https://doi.org/10.1007/s00442-020-04644-6
van Proosdij ASJ, Sosef MSM, Wieringa JJ, Raes N (2016) Minimum required number of specimen records to develop accurate species distribution models. Ecography 39:542–552. https://doi.org/10.1111/ecog.01509
Vilkov EV (2016) Characteristics of ecology of Bee-eaters (Merops apiaster, M. superciliosus) in Daghestan as a region of intensive migrations. South of Russia: Ecology, Development 11:90–105. https://doi.org/10.18470/1992-1098-2016-3-90-105
Webster MS, Marra PP, Haig SM et al (2002) Links between worlds: unraveling migratory connectivity. Trends Ecol Evol 17:76–83. https://doi.org/10.1016/S0169-5347(01)02380-1
Zhu B, Verhoeven MA, Velasco N et al (2022) Current breeding distributions and predicted range shifts under climate change in two subspecies of Black-tailed Godwits in Asia. Glob Chang Biol 28:5416–5426. https://doi.org/10.1111/gcb.16308
Zuckerberg B, Fink D, La Sorte FA et al (2016) Novel seasonal land cover associations for eastern North American forest birds identified through dynamic species distribution modelling. Divers Distrib 22:717–730. https://doi.org/10.1111/ddi.12428
Bastian H-V, Bastian A (2022) European Bee-eater (Merops apiaster). In: Billerman SM, Keeney BK (eds) Birds of the world, 2.0. Cornell Lab of Ornithology, Ithaca, NY, USA. https://doi.org/10.2173/bow.eubeat1.03
BirdLife International (2019) Merops apiaster (amended version of 2016 assessment). In: The IUCN Red List of Threatened Species 2019. https://www.iucnredlist.org/species/22683756/155512816. Accessed 5 May 2023
EBCC (2022) European breeding bird atlas 2 website. In: European bird census council. https://ebba2.info/maps/. Accessed 16 Jan 2024
GBIF.org (2022) GBIF Occurrence Download. https://doi.org/10.15468/dl.juskee. Accessed 23 Nov 2022
Hostetler JA, Hallworth MT, Rushing CS, Cohen EB (2021) MigConnectivity: Estimate Migratory Connectivity for Migratory Animals
Acknowledgements
Many thanks to Dr Steffan Hahn, Dr Kiran Dhanjal-Adams, and Dr José Alves, for contributing the use of the European Bee-eater solar-geolocator tracking data within this study.
Funding
CAW was supported by the Sam Hignett Travelling Scholarship provided by the RVC Animal Care Trust. JSC was supported by Fundação para a Ciência e a Tecnologia (FCT) grant SFRH/BD/113580/2015, the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 847635 and by financial support from national funds provided by FCT/MCTES (Ministério da Ciência, Tecnologia e Ensino Superior) to CESAM (UIDP/50017/2020 + UIDB/50017/2020 + LA/P/0094/2020).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No competing interests between the authors.
Additional information
Communicated by F. Bairlein.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdul-Wahab, C., Costa, J.S., D’Mello, F. et al. Connected impacts: combining migration tracking data with species distribution models reveals the complex potential impacts of climate change on European bee-eaters. J Ornithol (2024). https://doi.org/10.1007/s10336-024-02190-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10336-024-02190-z