Abstract
Bioindicators of wildlife health are useful tools for studying the viability of various organisms and populations, and can include a range of phenotypic variables, such as behavior, body size, and physiological parameters, such as circulating hormones and nutrients. Few studies have investigated the utility of total plasma protein as a predictor of environmental or nutritional variation among birds, as well as variation across different habitats and life-history stages. Here, we examined relationships between plasma protein and season, urbanization, sex, body condition, molt status, and disease state in House Finches (Haemorhous mexicanus). We sampled blood from House Finches across three seasons (winter, summer, and fall 2021) and measured plasma protein levels using a Bradford assay. We also collected data including condition, sex, and poxvirus infection state at capture, as well as fecal samples to assess gut parasitism (coccidiosis). During the fall season, we also estimated molt status, as number of actively growing feathers. We found a significant relationship between circulating protein levels and capture site, as well as novel links to molt state and pox presence, with urban birds, those infected with pox, and those in more intense molt having higher protein levels. Our results support the hypotheses that plasma protein concentration can be indicative of a bird’s body molt and degree of habitat urbanization, although future work is needed to determine why protein levels were higher in virus-infected birds.
Zusammenfassung
Variation des Plasmaproteinspiegels beim Hausgimpel ( Haemorhous mexicanus ): Auswirkungen von Jahreszeit, Krankheitszustand und Urbanisierung
Bioindikatoren für die Gesundheit von Wildtieren sind nützliche Instrumente zur Untersuchung der Überlebensfähigkeit verschiedener Organismen und Populationen und können eine Reihe von phänotypischen Parametern wie Verhalten, Körpergröße und physiologische Parameter, z. B. im Blut zirkulierende Hormone und Nährstoffe, umfassen. Nur wenige Studien haben den Nutzen des Gesamtplasmaproteinspiegels als Indikator für umwelt- und ernährungsbedingte Variation unter Vögeln sowie Unterschiede über verschiedene Lebensräume und -stadien hinweg untersucht. In dieser Studie haben wir die Beziehungen zwischen dem Plasmaproteinspiegel und Jahreszeit, Urbanisierung, Geschlecht, Körperkondition, Mauser- und Krankheitszustand beim Hausgimpel (Haemorhous mexicanus) beleuchtet. Es wurden Blutproben vom Hausgimpel über drei Jahreszeiten (Winter, Sommer, Herbst 2021) entnommen und der Plasmaproteinspiegel mithilfe eines Bradford-Tests gemessen. Weiterhin sammelten wir Daten zu Körperkondition, Geschlecht und Grad der Pockenvirusinfektion beim Fang sowie Kotproben, um den Befall mit Darmparasiten (Kokzidiose) zu beurteilen. Weiterhin schätzten wir während der Herbstsaison den Mauserzustand ein, d. h. die Anzahl an aktiv wachsenden Federn. Wir fanden eine signifikante Beziehung zwischen dem zirkulierenden Proteinspiegel und dem Fangort sowie neue Zusammenhänge zwischen dem Mauserzustand und vorhandenen Pockenviren, wobei Vögel in der Stadt, mit Pockenviren infizierte Vögel und Vögel, die sich in einer intensiveren Mauser befanden, einen höheren Proteinspiegel aufwiesen. Unsere Ergebnisse unterstützen die Hypothese, dass der Plasmaproteinspiegel einen Hinweis auf den Mauserstatus der Vögel und den Grad der Verstädterung ihres Habitats bieten kann, wenn auch noch zukünftige Untersuchungen benötigt werden, um festzustellen, warum der Proteinspiegel bei Vögeln mit einer Virusinfektion höher war.





Similar content being viewed by others
Data availability
The datasets generated during and analysed during the current study are available from the corresponding author on reasonable request.
References
Ashraf M, Rea R (2017) Effect of dehydration on blood tests. Pract Diabetes 34:169–171. https://doi.org/10.1002/pdi.2111
Balan DG, Sianu DP, Stanescu II, Ionescu D, Stroescu Balcangiu AE, Raducu L, Stoicescu SM, Ceau AM, Tiliscan C, Nimigean VR, Tarmure V, Croitoru AG (2018) A comparative evaluation of serum and salivary total proteins and immunoglobulins in patients with hepatitis A and healthy subjects. Rev Chim 69:1125–1128. https://doi.org/10.37358/RC.18.5.6273
Bichet C, Scheifler R, Cœurdassier M, Julliard R, Sorci G, Loiseau C (2013) Urbanization, trace metal pollution, and malaria prevalence in the house sparrow. PLoS ONE. https://doi.org/10.1371/journal.pone.0053866
Bozdogan H (1987) Model selection and Akaike’s information criterion (AIC): the general theory and its analytical extensions. Psychometrika 52:345–370
Bradley CA, Altizer S (2007) Urbanization and the ecology of wildlife diseases. Trends Ecol Evol 22:95–102. https://doi.org/10.1016/j.tree.2006.11.001
Brock PA, Hall AJ, Goodman SJ, Cruz M, Acevedo-Whitehouse K (2013) Immune activity, body condition and human-associated environmental impacts in a wild marine mammal. PLoS ONE. https://doi.org/10.1371/journal.pone.0067132
Cazenave J, Bacchetta C, Parma MJ, Scarabotti PA, Wunderlin DA (2009) Multiple biomarkers responses in Prochilodus lineatus allowed assessing changes in the water quality of Salado River basin (Santa Fe, Argentina). Environ Pollut 157:3025–3033. https://doi.org/10.1016/j.envpol.2009.05.055
Chace JF, Walsh JJ (2006) Urban effects on native avifauna: a review. Landsc Urban Plan 74:46–69. https://doi.org/10.1016/j.landurbplan.2004.08.007
Chan SM, Rankin SM, Keeley LL (1988) Characterization of the molt stages in Penaeus vannamei: setogenesis and hemolymph levels of total protein, ecdysteroids, and glucose. Biol Bull 175:185–192
Chaousis S, Leusch FD, van de Merwe JP (2018) Charting a path towards non-destructive biomarkers in threatened wildlife: a systematic quantitative literature review. Environ Pollut 234:59–70. https://doi.org/10.1016/j.envpol.2017.11.044
Cherel Y, Charrassin JB, Challet E (1994) Energy and protein requirements for molt in the king penguin Aptenodytes patagonicus. Am J Physiol Regul Integr Comp Physiol 266:1182–1188. https://doi.org/10.1152/ajpregu.1994.266.4.R1182
Dawson RD, Bortolotti GR (1997) Total plasma protein level as an indicator of condition in wild American kestrels (Falco sparverius). Can J Zool 75:680–686. https://doi.org/10.1139/z97-088
Dolnik VR, Gavrilov VM (1979) Bioenergetics of molt in the chaffinch (Fringilla coelebs). Auk 96:253–264
Dominoni DM (2015) The effects of light pollution on biological rhythms of birds: an integrated, mechanistic perspective. J Ornithol 156:409–418. https://doi.org/10.1007/s10336-015-1196-3
Dominoni DM, Quetting M, Partecke J (2013) Long-term effects of chronic light pollution on seasonal functions of European blackbirds (Turdus merula). PLoS ONE. https://doi.org/10.1371/journal.pone.0085069
Funk A, Hutton P, Earl S, Deviche P, Sweazea K (2020) Levels of land use and land cover in Phoenix, Arizona are associated with elevated plasma triglycerides in the Gambel’s Quail, Callipepla gambelii. Comp Biochem Physiol Part A Mol Integr Physiol 247:110730. https://doi.org/10.1016/j.cbpa.2020.110730
Gavett AP, Wakeley JS (1986) Blood constituents and their relation to diet in urban and rural house sparrows. Condor 88:279–284. https://doi.org/10.2307/1368873
Giraudeau M, Mousel M, Earl S, McGraw K (2014) Parasites in the city: degree of urbanization predicts poxvirus and coccidian infections in house finches (Haemorhous mexicanus). PLoS ONE. https://doi.org/10.1371/journal.pone.0086747
Giraudeau M, Toomey MB, Hutton P, McGraw KJ (2018) Expression of and choice for condition-dependent carotenoid-based color in an urbanizing context. Behav Ecol 29:1307–1315. https://doi.org/10.1093/beheco/ary093
Hashemnia M, Khodakaram-Tafti A, Razavi SM, Nazifi S (2014) Hematological and serum biochemical analyses in experimental caprine coccidiosis. J Parasit Dis 38:116–123. https://doi.org/10.1007/s12639-012-0205-1
Heitmeyer ME (1988) Protein costs of the prebasic molt of female mallards. Condor 90:263–266. https://doi.org/10.2307/1368465
Hill GE (2002) A red bird in a brown bag: the function and evolution of colorful plumage in the house finch. Oxford University Press, New York
Hope D, Gries C, Casagrande D, Redman CL, Grimm NB, Martin C (2006) Drivers of spatial variation in plant diversity across the Central Arizona-Phoenix ecosystem. Soc Nat Resour 19:101–116
Hutton P, McKenna J, McGraw KJ (2021) Urban links to molt schedule, body condition and carotenoid-based coloration in the house finch Haemorhous mexicanus. J Avian Biol. https://doi.org/10.1111/jav.02761
Isaksson C, Örnborg J, Stephensen E, Andersson S (2005) Plasma glutathione and carotenoid coloration as potential biomarkers of environmental stress in great tits. EcoHealth 2:138–146. https://doi.org/10.1007/s10393-005-3869-5
Job JR, Kohler SL, Gill SA (2016) Song adjustments by an open habitat bird to anthropogenic noise, urban structure, and vegetation. Behav Ecol 27:1734–1744. https://doi.org/10.1093/beheco/arw105
Koelmel JP, Ulmer CZ, Fogelson S, Jones CM, Botha H, Bangma JT, Guillette TC, Luus-Powell WJ, Sara JR, Smit WJ, Albert K, Miller HA, Guillette MP, Olsen BC, Cochran JA, Garrett TJ, Yost RA, Bowden JA (2019) Lipidomics for wildlife disease etiology and biomarker discovery: a case study of pansteatitis outbreak in South Africa. Metabolomics 15(3):1–11. https://doi.org/10.1007/s11306-019-1490-9
Lessells CM, Boag PT (1987) Unrepeatable repeatabilities: a common mistake. Auk 104:116–121. https://doi.org/10.2307/4087240
McEwan JC, Mason P, Baker RL, Clarke JN, Hickey SM, Turner K (1992) Effect of selection for productive traits on internal parasite resistance in sheep. Proc NZ Soc Anim Prod 52:53–56
McGraw KJ, Hill GE (2000) Differential effects of endoparasitism on the expression of carotenoid- and melanin-based ornamental coloration. Proc Roy Soc B 267:1525–1531. https://doi.org/10.1098/rspb.2000.1174
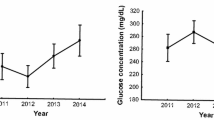
McGraw KJ, Chou K, Bridge A, McGraw HC, McGraw PR, Simpson RK (2020) Body condition and poxvirus infection predict circulating glucose levels in a colorful songbird that inhabits urban and rural environments. J Exp Zool A Ecol Integr Physiol 333:561–568. https://doi.org/10.1002/jez.2391
Mills GS, Dunning JB, Bates JM (1989) Effects of urbanization on breeding bird community structure in southwestern desert habitats. Condor 91:416–428. https://doi.org/10.2307/1368320
Mondal D, Chattopadhyay S, Batabyal S, Bera A, Bhattacharya D (2011) Plasma biochemical indices at various stages of infection with a field isolate of Eimeria tenella in broiler chicken. Vet World 4:404–409. https://doi.org/10.5455/vetworld.2011.404-409
Murphy ME, King JR (1992) Energy and nutrient use during moult by white-crowned sparrows Zonotrichia leucophrys gambelii. Ornis Scand 23:304–313. https://doi.org/10.2307/3676654
Murphy ME, Taruscio TG (1995) Sparrows increase their rates of tissue and whole-body protein synthesis during the annual molt. Comp Biochem Physiol Part A Physiol 111:385–396. https://doi.org/10.1016/0300-9629(95)00039-A
Norte AC, Ramos JA, Sousa JP, Sheldon BC (2009) Variation of adult great tit Parus major body condition and blood parameters in relation to sex, age, year and season. J Ornithol. https://doi.org/10.1007/s10336-009-0387-1
Oladele SB, Nok AJ, Esievo KAN, Abdu P, Useh NM (2005) Haemagglutination inhibition antibodies, rectal temperature and total protein of chickens infected with a local nigerian isolate of velogenic Newcastle disease virus. Vet Res Commun 29:171–179. https://doi.org/10.1023/B:VERC.0000047495.03341.2b
Ortiz-Santaliestra ME, Tauler-Ametller H, Lacorte S, Hernández-Matías A, Real J, Mateo R (2019) Accumulation of pollutants in nestlings of an endangered avian scavenger related to territory urbanization and physiological biomarkers. Environ Pollut B 252:1801–1809. https://doi.org/10.1016/j.envpol.2019.06.101
Patil R, Raghuwanshi U (2009) Serum protein, albumin, globulin levels, and A/G ratio in HIV positive patients. Biomed Pharmacol J 2:321–325
Peneaux C, Grainger R, Lermite F, Machovsky-Capuska GE, Gaston T, Griffin AS (2021) Detrimental effects of urbanization on the diet, health, and signal coloration of an ecologically successful alien bird. Sci Total Environ 796:148828. https://doi.org/10.1016/j.scitotenv.2021.148828
Pyle P (2008) Identification guide to North American Birds. Slate Creek Press, Point Reyes Station
Roman Y, Bomsel-Demontoy MC, Levrier J, Ordonneau D, Chaste-Duvernoy D, Saint Jalme M (2009) Influence of molt on plasma protein electrophoretic patterns in bar-headed geese (Anser indicus). J Wildl Dis 45:661–671. https://doi.org/10.7589/0090-3558-45.3.661
Ruff MD, Augustine PC (1982) Effects of coccidiosis on the electrophoretic pattern of serum proteins in chickens. J Parasitol Res 68:107–111. https://doi.org/10.2307/3281331
Saghazadeh A, Rezaei N (2022) Poxviruses and the immune system: implications for Monkeypox virus. Int Immunopharmacol 113:109364. https://doi.org/10.1016/j.intimp.2022.109364
Salmón P, Nilsson JF, Nord A, Bensch S, Isaksson C (2016) Urban environment shortens telomere length in nestling great tits, Parus major. Biol Lett 12:254–260. https://doi.org/10.1098/rsbl.2016.0155
Schaller J, Gerber S, Kaempfer U, Lejon S, Trachsel C (2008) Human blood plasma proteins: structure and function. Wiley, Hoboken
Serrano E, González FJ, Granados JE, Moço G, Fandos P, Soriguer RC, Pérez JM (2008) The use of total serum proteins and triglycerides for monitoring body condition in the Iberian wild goat (Capra pyrenaica). J Zoo Wildl Med 39:646–649. https://doi.org/10.1638/2007-0088.1
Acknowledgements
This work was supported by the National Science Foundation DEB-1832016, Central Arizona-Phoenix Long-Term Ecological Research Program (CAP LTER), and Barrett, The Honors College. We thank Dr. Karen Sweazea and Danny Jackson for input on the manuscript. We would also like to thank the South Mountain Environmental Education Center for graciously allowing us to conduct this research at their facilities. Finally, we would like to thank all of the undergraduate students who helped in this effort, Kathryn DePinto, Ian Sheedy, George Amacher, Elise Crawford-Paz Soldán, Preston Moskal, C. J. Writer, Jake Mitrius, Danielle Pais, Amanda Wrona, Cassie Rueda, Madison Hatcher, and Lauren West, as well as graduate students Victor Penha and Jamie Casseus. The experiments conducted comply with the current law of the United States of America, in which they were performed.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors are not aware of any competing interests related to this work.
Additional information
Communicated by I. Moore.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Drake, D.J., McGraw, K.J. Variation in plasma protein levels in House Finches (Haemorhous mexicanus): effects of season, disease state, and urbanization. J Ornithol 164, 629–638 (2023). https://doi.org/10.1007/s10336-023-02062-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-023-02062-y