Abstract
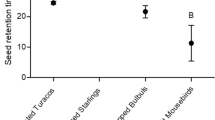
Figs (Ficus spp.) are a diverse taxon of woody plants that play keystone ecological roles. They provide nutritional and aseasonal fruit crops that are consumed by many frugivores, often in times of fruit scarcity. In a mutually beneficial relationship, the plants also benefit from seed dispersal by birds and mammals away from the parent plants enhancing germination and plant recruitment. Here, we assessed the effect of ingestion of Ficus fruit by avian frugivores, compared with manually de-pulped fruit and whole fruits, on seedling emergence and cumulative germination of three Ficus spp. (F. sur, F. lutea and F. natalensis). Fruits of Ficus spp. were fed to Dark-capped Bulbuls Pycnonotus tricolor, Knysna Turacos Tauraco corythaix, Purple-crested Turaco Gallirex porphyreolophus, Red-winged Starlings Onychognathus morio, Speckled Mousebirds Colius striatus and invasive Rose-ringed Parakeets Psittacula krameri. We recorded seed retention time as the time from the ingestion of seeds to the first appearance when excreted by the birds. Seeds removed from excreta, manually de-pulped seeds, and whole fruit were planted concurrently in soil trays in a greenhouse. Seedling emergence was recorded daily, and the proportion of seed germinated was calculated as germination success. Germination success of avian ingested seeds increased significantly compared with seeds in whole fruit and manually de-pulped seeds, except seed germination for F. sur seeds ingested by Dark-capped Bulbuls. Ingested seeds germinated the fastest, followed by seeds in whole fruits and lastly, de-pulped seeds. Ficus sur and F. lutea ingested seeds had higher germination percentages than F. natalensis. Our results showed that ingestion by birds typically improved Ficus seed germination as well as the additional positive effects of seed movement such as long-distance seed dispersal. These findings have important implications with the potential to improve Ficus seed dispersal, promote regeneration and sustain populations of the many vertebrates that depend on fig fruit.
Zusammenfassung
Die Rolle frugivorer Vögel bei Keimung und Verbreitung von Ficus -Arten mit fleischigen Früchten in KwaZulu-Natal, Südafrika
Feigen (Ficus spp.) sind ein diverses Taxon von Gehölzpflanzen, die ökologische Schlüsselrollen innehaben. Sie liefern ein nahrhaftes und jahreszeitübergreifendes Angebot an Früchten, die von zahlreichen Frugivoren verzehrt werden, häufig in Zeiten, in denen es sonst wenige Früchte gibt. Es ist ein Verhältnis zum gegenseitigen Vorteil, denn die Pflanzen profitieren von der Samenverbreitung durch Vögel und Säugetiere fort von den Elternpflanzen, wodurch Keimung und Entwicklung der Jungpflanzen begünstigt werden. Hier untersuchten wir den Effekt des Fruchtverzehrs durch frugivore Vögel auf Saatauflauf und die kumulative Keimrate dreier Ficus-Arten (F. sur, F. lutea und F. natalensis), verglichen mit von Hand entfernten Samen sowie mit ganzen Früchten. Die Früchte diverser Ficus-Arten wurden an Rußkopfbülbüls Pycnonotus tricolor, Helmturakos Tauraco corythaix, Glanzhaubenturakos Gallirex porphyreolophus, Rotschwingenstare Onychognathus morio, Braunflügel-Mausvögel Colius striatus sowie invasive Halsbandsittiche Psittacula krameri verfüttert. Wir erfassten die Darmpassagezeit, das heißt die Zeitspanne zwischen dem Verzehr der Samen bis zum ersten Wiedererscheinen beim Ausscheiden durch die Vögel. Die Samen aus den Ausscheidungen, von Hand vom Fruchtfleisch befreite Samen sowie ganze Früchte wurden zeitgleich in Pflanzschalen in einem Gewächshaus ausgebracht. Das Auflaufen von Keimlingen wurde täglich erfasst und der Anteil gekeimter Samen als Keimerfolg berechnet. Mit Ausnahme von Ficus sur-Samen, welche von Rußkopfbülbüls verzehrt worden waren, stieg der Keimerfolg bei den von Vögeln verzehrten Samen verglichen mit ganzen Früchten und den von Hand vom Fruchtfleisch befreiten Samen signifikant an. Verzehrte Samen keimten am schnellsten, gefolgt von den Samen aus ganzen Früchten und schließlich den aus dem Fruchtfleisch freigelegten Samen. Verzehrte Samen von Ficus sur und F. lutea wiesen höhere Keimraten auf als F. natalensis. Unsere Ergebnisse konnten zeigen, dass der Verzehr durch Vögel die Keimung von Ficus-Samen typischerweise verbesserte, abgesehen vom zusätzlichen Nutzen der Samenverbreitung über weitere Strecken. Diese Erkenntnisse sind von weitreichender Konsequenz durch ihr Potenzial zur Verbesserung der Verbreitung von Ficus-Samen sowie zur Förderung der Regeneration und des Erhalts von Populationen der zahlreichen Wirbeltiere, die von Feigenfrüchten abhängig sind.




Similar content being viewed by others
Data availability
Data for this study are available on reasonable request from the authors but belong to the University of KwaZulu-Natal.
References
Barnea A, Yom-Tov Y, Friedman J (1990) Differential germination of two closely related species of Solanum in response to bird ingestion. Oikos 57:222–228. https://www.jstor.org/stable/3565943
Barnea A, Yom-Tov Y, Friedman J (1991) Does ingestion by birds affect seed germination? Funct Ecol 5:394–402. https://www.jstor.org/stable/2389811
Chama L, Berens DG, Downs CT, Farwig N (2013) Do frugivores enhance germination success of plant species? An experimental approach. S Afr J Bot 88:23–27. https://doi.org/10.1016/j.sajb.2013.05.003
Charalambidou I, Santamaria L, Langevoord O (2003) Effect of ingestion by five avian dispersers on the retention time, retrieval and germination of Ruppia maritima seeds. Funct Ecol 17:747–753. https://www.jstor.org/stable/3599246
Chimera CG, Drake DR (2010) Effects of pulp removal on seed germination of five invasive plants in Hawaii. Plant Protect Quart 25:137
Compton SG, Greeff JM (2020) Few figs for frugivores: Riparian fig trees in Zimbabwe may not be a dry season keystone resource. Afr J Ecol 58:778–785. https://doi.org/10.1111/aje.12773
Cottee-Jones HEW, Bajpai O, Chaudhary LB, Whittaker RJ (2016) The importance of Ficus (Moraceae) trees for tropical forest restoration. Biotropica 48:413–419. https://doi.org/10.1111/btp.12304
Czarnecka J, Orłowski G, Karg J (2012) Endozoochorous dispersal of alien and native plants by two Palearctic avian frugivores with special emphasis on invasive giant goldenrod Solidago gigantea. Centre Euro J Biol 7:895–901. https://doi.org/10.2478/s11535-012-0081-9
David JP, Manakadan R, Ganesh T (2015) Frugivory and seed dispersal by birds and mammals in the coastal tropical dry evergreen forests of southern India: a review. Trop Ecol 56:41–55
D’Avila G, Gomes-Jr A, Canary AC (2010) The role of avian frugivores on germination and potential seed dispersal of the Brazilian pepper Schinus terebinthifolius. Biota Neotrop 10:45–51
Debussche M, Isenmann P (1994) Bird-dispersed seed rain and seedling establishment in patchy Mediterranean vegetation. Oikos 69:414–426
Díaz Vélez MC, Sérsic AN, Traveset A, Paiaro V (2018) The role of frugivorous birds in fruit removal and seed germination of the invasive alien Cotoneaster franchetii in central Argentina. Austral Ecol 43:558–566. https://doi.org/10.1111/aec.12592
Dlamini P, Zachariades C, Downs CT (2018) The effect of frugivorous birds on seed dispersal and germination of the invasive Brazilian pepper tree (Schinus terebinthifolius) and Indian laurel (Litsea glutinosa). S Afr J Bot 114:61–68. https://doi.org/10.1016/j.sajb.2017.10.009
Fedriani JM, Zywiec M, Delibes M (2011) Thieves or mutualists? Pulp feeders enhance endozoochore local recruitment. Ecology 93:575–587. https://doi.org/10.1890/11-0429.1
Figueroa JA, Castro SA (2002) Effects of bird ingestion on seed germination of four woody species of the temperate rainforest of Chiloé island, Chile. Plant Ecol 160:17–23. https://doi.org/10.1023/A:1015889017812
Fricke EC, Simon MJ, Reagan KM, Levey DJ, Riffell JA, Carlo TA, Tewksbury JJ (2013) When condition trumps location: seed consumption by fruit-eating birds removes pathogens and predator attractants. Ecol Letters 16:1031–1036. https://doi.org/10.1111/ele.12134
Fukui A (2005) Relationship between seed retention time in bird’s gut and fruit characteristics. Ornith Sci 2:41–48. https://doi.org/10.2326/osj.2.41
Gosper CR, Stansbury CD, Vivian-Smith G (2005) Seed dispersal of fleshy-fruited invasive plants by birds: contributing factors and management options. Divers Distrib 11:549–558. https://doi.org/10.1111/j.1366-9516.2005.00195.x
Guerrero AM, Tye A (2009) Darwin’s Finches as seed predators and dispersers. Wilson J Ornith 121:752–764. https://doi.org/10.1676/09-035.1
Hammer Ø, Harper DA, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeo Elect 4:1–9
Heer K, Albrecht L, Kalko EK (2010) Effects of ingestion by neotropical bats on germination parameters of native free-standing and strangler figs (Ficus sp., Moraceae). Oecologia 163:425–435. https://www.jstor.org/stable/40606600
Herrera CM (1995) Plant-vertebrate seed dispersal systems in the Mediterranean: ecological, evolutionary, and historical determinants. Ann Rev Ecol System 26:705–727. https://www.jstor.org/stable/2097225
Herrera CM (2003) Seed dispersal by vertebrates. In: Herrera CM, Pellmyr O (eds) Plant-animal interactions: an evolutionary approach. Blackwell Publishing, Oxford, pp 185–208
Holbrook KM (2011) Home range and movement patterns of toucans:implications for seed dispersal. Biotropica 43:357–364
Janzen DH (1981) Ficus ovalis seed predation by an orange-chinnedparakeet (Brotogeris jugularis) in Costa Rica. Auk 98:841–844
Jeevanandam N, Corlett RT (2013) Fig wasp dispersal in urban Singapore. Raffles Bull Zool 61:343–347
Jordaan LA, Johnson SD, Downs CT (2011) The role of avian frugivores in germination of seeds of fleshy-fruited invasive alien plants. Biol Invasions 13:1917–1930. https://doi.org/10.1007/s10530-011-0013-z
Jordaan LA, Johnson SD, Downs CT (2012) Wahlberg’s epauletted fruit bat (Epomophorus wahlbergi) as a potential dispersal agent for fleshy-fruited invasive alien plants: effects of handling behaviour on seed germination. Biol Invasions 14:959–968. https://doi.org/10.1007/s10530-011-0131-7
Jordano P (2000) Seeds: the ecology of regeneration in plant communities. CAB International, Wallingford, pp 125–166
Jordano P (1983) Fig-seed predation and dispersal by birds. Biotropica 15:38–41. https://www.jstor.org/stable/2387996
Kalko EK, Herre EA, Handley CO (1996) Relation of fig fruit characteristics to fruit-eating bats in the new and old world tropics. J Biogeog 23:565–576. https://doi.org/10.1111/j.1365-2699.1996.tb00018.x
Kattan GH, Valenzuela LA (2013) Phenology, abundance and consumers of figs (Ficus spp.) in a tropical cloud forest: evaluation of a potential keystone resource. J Tropical Ecol 29:401–407. https://www.jstor.org/stable/43831666
Kirika JM, Bleher B, Böhning-Gaese K, Chira R, Farwig N (2008) Fragmentation and local disturbance of forests reduce frugivore diversity and fruit removal in Ficus thonningii trees. Basic Appl Ecol 9:663–672. https://doi.org/10.1016/j.baae.2007.07.002
Korine C, Kalko EK, Herre EA (2000) Fruit characteristics and factors affecting fruit removal in a Panamanian community of strangler figs. Oecologia 123:560–568. http://www.jstor.com/stable/4222655
LaRosa AM, Smith CW, Gardner DE (1985) Role of alien and native birds in the dissemination of fire tree (Myricafaya Ait.-Myriacaceae) and associated plants in Hawaii. Pacific Sci 39:372–437
Meyer G, Witmer M (1998) Influence of seed processing by frugivorous birds on germination success of three North American shrubs. Am Midland Nat 140:129–139. https://www.jstor.org/stable/2426996
Mokotjomela TM, Musil CF, Esler KJ (2013) Frugivorous birds visit fruits of emerging alien shrub species more frequently than those of native shrub species in the South African Mediterranean climate region. S Afr J Bot 86:73–78. https://doi.org/10.1016/j.sajb.2013.02.004
Molefe KL, Tedder MJ, Thabethe V, Rushworth Downs CT (2019) Role of native avian frugivores in germination facilitation and potential dispersal of invasive American bramble (Rubus cuneifolius) in South Africa. Biol Invasion 22:1109–1120. https://doi.org/10.1007/s10530-019-02164-w
Moles AT, Warton DI, Westoby M (2003) Do small-seeded species have higher survival through seed predation than large-seeded species? Ecology 84:3148–3161. https://doi.org/10.1890/02-0662
Moore PD (2001) The guts of seed dispersal. Nature 414:406–407. https://doi.org/10.1038/35106677
Murray KG, Russell S, Picone CM, Winnettmurray K, SherwoodW Kuhlmann ML (1994) Fruit laxatives and seedpassage rates in frugivores consequences for plant reproductivesuccess. Ecology 75:989–994
Noble JC (1975) The effects of emu (Dromaius novaehollandiae Latham) on the distribution of the nitre bush (Nitraria billardier DC). J Ecol 63:979–984. https://www.jstor.org/stable/2258615
Nogales M, Nieves C, Illera JC, Padilla DP, Traveset A (2005) Effect of native and alien vertebrate frugivores on seed viability and germination patterns of Rubia fruticosa (Rubiaceae) in the eastern Canary Islands. Funct Ecol 19:429–436. https://www.jstor.org/stable/3599136
Panetta FD (2001) Seedling emergence and seed longevity of the tree weeds Celtis sinensis and Cinnamomum camphora. Weed Res 41:83–95
Paulsen TR, Hogstedt G (2002) Passage through bird guts increases germination rate and seedling growth in Sorbus aucuparia. Funct Ecol 16:608–616. https://www.jstor.org/stable/826743
R Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, https://www.r-project.org/
Righini N, Serio-Silva JC, Rico-Gray V, Martínez-Mota R (2004) Effect of different primate species on germination of Ficus (Urostigma) seeds. Zoo Biol 23:273–278. https://doi.org/10.1002/zoo.20008
Robertson AW, Trass A, Ladley JJ, Kelly D (2006) Assessing the benefits of frugivory for seed germination: the importance of thedeinhibition eVect. Funct Ecol 20:58–66
Samuels IA, Levey DJ (2005) Effects of gut passage on seed germination: do experiments answer the question they ask? Funct Ecol 19:365–368. https://www.jstor.org/stable/3599313
Shanahan M, So S, Compton SG, Corlett R (2001) Fig-eating by vertebrate frugivores: a global review. Biol Rev 76:529–572. https://doi.org/10.1017/S1464793101005760
Thabethe V, Thompson LJ, Hart LA, Brown M, Downs CT (2015) Ingestion by an invasive parakeet species reduces germination success of invasive alien plants relative to ingestion by indigenous turaco species in South Africa. Biol Invasions 17:3029–3039. https://doi.org/10.1007/s10530-015-0932-1
Traveset A (1998) Effect of seed passage through vertebrate frugivores’ guts on germination: a review. Perspect Plant Ecol Evol Sys 1:151–190. https://doi.org/10.1078/1433-8319-00057
Traveset A, Riera N, Mas RE (2001) Passage through bird guts causes interspecific differences in seed germination characteristics. Funct Ecol 15:669–675. https://doi.org/10.1046/j.0269-8463.2001.00561.x
Vaz Ferreira A, Bruna EM, Vasconcelos HL (2011) Seed predators limit plant recruitment in Neotropical savannas. Oikos 120:1013–1022. https://doi.org/10.1111/j.1600-0706.2010.19052.x
Voigt F, Farwig N, Johnson SD (2011) Interactions between the invasive tree Melia azedarach (Meliaceae) and native frugivores in South Africa. J Trop Ecol 27:355–363
Wilson A-L, Downs CT (2011) Food preferences of Knysna and purple-crested turacos fed varying concentrations of equicaloric and equimolar artificial fruit. J Exp Biol 214:613–618. https://doi.org/10.1242/jeb.047217
Wilson A-L, Downs CT (2012) Knysna turacos (Tauraco corythaix) do not improve seed germination of ingested fruit of some indigenous South African tree species. S Afr J Bot 78:55–62. https://doi.org/10.1016/j.sajb.2011.05.006
Witkowski ETF, Garner RD (2008) Seed production, seed bank dynamics, resprouting and long-term response to clearing of the alien invasive Solanum mauritianum in a temperate to subtropical riparian ecosystem. S Afr J Bot 74:476–484. https://doi.org/10.1016/j.sajb.2008.01.173
Witmer MC, Cheke AS (1991) The dodo and the tambalacoque tree: an obligate mutualism reconsidered. Oikos 61:133–137. https://www.jstor.org/stable/3545415
Yagihashi T, Hayashida M, Miyamoto T (1998) Effects of bird ingestion on seed germination of Sorbus commixta. Oecologia 114:209–212. https://doi.org/10.1007/s004420050438
Yoshikawa T, Isagi Y (2014) Negative effect of removing pulp from unripe fleshy fruits: seed germination pattern of Celtis sinensis in relation to the temporal context of fruit consumption. J Forest Res 19:411–416. https://doi.org/10.1007/s10310-013-0430-1
Young LM, Kelly D, Nelson XJ (2012) Alpine flora may depend on declining frugivorous parrot for seed dispersal. Biol Conserv 147:133–142. https://doi.org/10.1016/j.biocon.2011.12.023
Zhang G, Song Q, Yang D (2006) Phenology of Ficus racemosa in Xishuangbanna, Southwest China. Biotropica 38:334–341. https://doi.org/10.1111/j.1744-7429.2006.00150.x
Acknowledgements
Thanks to T. Mjara, E. Ally and M. Nkomo for assisting with experimental setup and data collection.
Funding
We would like to thank the University of KwaZulu-Natal (ZA) and the National Research Foundation (NRF, ZA, Grant 98404) for financial support, especially IAR with a NRF-TWAS African Renaissance bursary. We thank the Ford Wildlife Foundation (ZA) for vehicle support.
Author information
Authors and Affiliations
Contributions
All authors conceived the research idea. CTD sort funding. IAR collected and analysed the data. IAR drafted the manuscript. All authors provided comments and revisions to the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest nor competing interests.
Ethical approval
Ethics clearance was obtained from the University of KwaZulu-Natal Animal Ethics Committee (020/15/Animal).
Consent for publication
All authors approved the manuscript for publication.
Additional information
Communicated by F. Bairlein.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Raji, I.A., Thabethe, V. & Downs, C.T. The role of avian frugivores in the germination and dispersal of fleshy-fruited Ficus species in KwaZulu-Natal, South Africa. J Ornithol 163, 395–404 (2022). https://doi.org/10.1007/s10336-022-01963-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-022-01963-8