Abstract
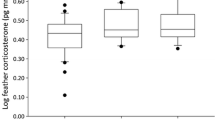
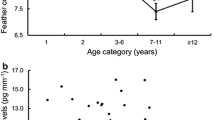
In birds, integrated levels of corticosterone (CORT) measured in feathers (CORTf) allow us to make inferences on past levels of stress demands. It has been suggested that levels of CORTf track carry-over effects across seasons. Nevertheless, our understanding of how this measure can be used to assess future health status is far from complete. In this study, we aimed to investigate whether CORT deposited in feathers over the moulting period was related to subsequent mortality and health status in wild raptors admitted to rehabilitation centres. Thirty-four Eurasian Sparrowhawks (Accipiter nisus) admitted during the non-moulting period were sampled. Body condition (BC) was used as an indicator of health status to classify individuals’ health as good, poor or cachexia depending on their pectoral muscle score. Mortality was recorded over the non-moulting period. Other potential sources of CORTf variation were assessed, such as sex, age and feather type, primary or body covert feathers. While CORTf did not vary with age or sex, significant differences were found between primary and body feathers, highlighting the importance of sampling the same feather type. Our results also revealed that birds in poor BC showed higher CORTf levels than individuals in good condition; however, CORTf levels in cachectic birds did not differ from those in good condition. This finding suggests caution when assuming that only high CORTf levels represent individuals in poor condition, and limits the utility of CORTf for the prediction of BC. The present study also showed that individuals which died following admission had higher CORTf levels than individuals which survived, suggesting the potential utility of CORTf as a metric for the study of subsequent mortality.
Zusammenfassung
Zusammenhang zwischen Feder-Kortikosteron und späterem Gesundheitszustand und Überleben bei Europäischen Sperbern
Der Messung von Kortikosteron in Federn (CORTf) erlaubt Rückschlüsse auf vergangene Stresssituationen. Es werde angenommen, dass über CORTf-Spiegel Carry-over Effekte über die Zeit hinweg verfolgt werden können. Gleichwohl ist unser Verständnis, wie solche Werte zur Abschätzung des zukünftigen Gesundheitszustandes genutzt werden können bei Weitem nicht vollständig. Ziel dieser Studie war zu untersuchen, ob in Federn zur Mauserzeit eingelagertes Kortikosteron (CORT) mit späterer Mortalität und dem Gesundheitszustand von Sperbern, die in Greifvogelauffangstationen eingeliefert wurden, zusammenhängen. Dazu beprobten wir 34 außerhalb der Mauserperiode eingelieferte Europäische Sperber (Accipiter nisus). In Abhängigkeit vom Brustmuskelwert wurde die individuelle Gesundheit als gut, schwach oder abgemagert klassifizieren. Zudem wurde die Sterblichkeit über die Nichtmauserzeit ermittelt. Andere potentielle Quellen für Unterschiede im CORTf wie bspw. Geschlecht, Altern und Federtypen, Großgefieder oder Körperfedern wurden ebenfalls berücksichtigt. Während CORTf nicht mit Alter und Geschlecht variierte, wurden signifikante Unterschiede zwischen Groß- und Körpergefieder gefunden, was die Bedeutung der Beprobung desselben Federtyps hervorhebt. Unsere Ergebnisse zeigen auch, dass Vögel mit schlechter Kondition höhere CORTf Werte zeigten als Individuen in guter Kondition, wobei aber die CORTf Werte abgemagerter Vögel sich nicht von denen in guter Kondition unterschieden. Dieses Ergebnis unterstreicht die nötige Vorsicht, wenn angenommen wird, dass nur hohe CORtf Werte für Individuen mit schwacher Kondition stehen und reduziert gleichzeitig die Nützlichkeit von CORTf zur Vorhersage von Körperkonditionen. Die vorliegende Studie zeigt auch, dass Individuen, die später starben, höhere CORTf Werte hatten als Individuen, die überlebten, was zeigt, dass Federkortikosteron als ein Maß für spätere Mortalität verwendet werden kann.




Similar content being viewed by others
References
Aharon-Rotman Y, Buchanan KL, Klaassen M, William A (2015) An experimental examination of interindividual variation in feather corticosterone content in the House Sparrow, Passer domesticus in southeast Australia. Gen Comp Endocrinol. doi:10.1016/j.ygcen.2015.12.010
Angelier F, Weimerskirch H, Dano S, Chastel O (2007) Age, experience and reproductive performance in a long-lived bird: a hormonal perspective. Behav Ecol Sociobiol 61:611–621. doi:10.1007/s00265-006-0290-1
Bonier F, Martin PR, Moore IT, Wingfield JC (2009) Do baseline glucocorticoids predict fitness? Trends Ecol Evol 24:634–642. doi:10.1016/j.tree.2009.04.013
Bortolotti GR (2010) Flaws and pitfalls in the chemical analysis of feathers: bad news–good news for avian chemoecology and toxicology. Ecol Appl 20:1766–1774
Bortolotti GR, Marchant TA, Blas J, German T (2008) Corticosterone in feathers is a long-term, integrated measure of avian stress physiology. Funct Ecol 22:494–500. doi:10.1111/j.1365-2435.2008.01387.x
Bortolotti GR, Mougeot F, Martinez-Padilla J, Webster LMI, Pierney SB (2009) Physiological stress mediates the honesty of social signals. PLoS One 4:e4983. doi:10.1371/journal.pone.0004983
Bourgeon S, Leat EHK, Magnusdóttir E, Furness RW, Strøm H, Petersen A, Gabrielsen GW, Hanssen SA, Bustnes JO (2014) Feather corticosterone levels on wintering grounds have no carry-over effects on breeding among three populations of Great Skuas (Stercorarius skua). PLoS One 9:e100439. doi:10.1371/journal.pone.0100439
Brown CR, Brown MB, Raouf SA, Smith LC, Wingfield JC (2005) Effects of endogenous steroid hormone levels on annual survival in Cliff Swallows. Ecology 86:1034–1046
Busch DS, Hayward LS (2009) Stress in a conservation context: a discussion of glucocorticoid actions and how levels change with conservation-relevant variables. Biol Conserv 142:2844–2853
Carbajal A, Tallo-Parra O, Sabes-Alsina M, Mular I, Lopez-Bejar M (2014) Feather corticosterone evaluated by ELISA in broilers: a potential tool to evaluate broiler welfare. Poult Sci 93:1–3
Carrete M, Bortolotti GR, Sánchez-Zapata JA, Delgado A, Cortés-Avizanda A, Grande JM, Donázar JA (2013) Stressful conditions experienced by endangered Egyptian Vultures on African wintering areas. Anim Conserv 16:353–358. doi:10.1111/acv.12001
Cooper NW, Sherry TW, Marra PP (2015) Experimental reduction of winter food decreases body condition and delays migration in a long-distance migratory bird. Ecology 96:1933–1942
Crawley MJ (1993) GLIM for ecoloagists. Blackwell, Cambridge
Crossin GT, Phillips RA, Lattin CR, Romero LM, Williams TD (2013) Corticosterone mediated costs of reproduction link current to future breeding. Gen Comp Endocrinol 193:112–120. doi:10.1016/j.ygcen.2013.07.011
Debén S, Fernández JA, Aboal JR, Carballeira A (2012) Evaluation of different contour feather types for biomonitoring lead exposure in Northern Goshawk (Accipiter gentilits) and Tawny Owl (Strix aluco). Ecotoxiol Environ Saf 85:115–119. doi:10.1016/j.ecoenv.2012.08.005
Dickens MJ, Romero LM (2013) A consensus endocrine profile for chronically stressed wild animals does not exist. Gen Comp Endocrinol 191:177–189. doi:10.1016/j.ygcen.2013.06.014
Fairhurst GD, Navarro J, Gonzalez-Solis J, Marchant TA, Bortolotti GR (2012) Feather corticosterone of a nestling seabird reveals consequences of sex-specific parental investment. Proc R Soc B Biol Sci 279:177–184. doi:10.1098/rspb.2011.0884
Fairhurst GD, Marchant TA, Soos C, Machin KL, Clark RG (2013) Experimental relationships between levels of corticosterone in plasma and feathers in a free-living bird. J Exp Biol 216:4071–4081. doi:10.1242/jeb.091280
Fairhurst GD, Dawson RD, van Oort H, Bortolotti GR (2014) Synchronizing feather-based measures of corticosterone and carotenoid-dependent signals: what relationships do we expect? Oecologia 174:689–698. doi:10.1007/s00442-013-2830-5
Gill F (2007) Feathers. In: Ornithology, 3rd edn. W.H. Freeman and Company, New York, pp 79–113
Hardy J, Crick H, Wernham C, Riley H, Etheridge B, Thompson D (2006) Feathers. In: Raptors: a field guide for surveys and monitoring. The Stationery Office, Edinburgh, pp 243–280
Harms NJ, Fairhurst GD, Bortolotti GR, Smits JEG (2010) Variation in immune function, body condition, and feather corticosterone in nestling Tree Swallows (Tachycineta bicolor) on reclaimed wetlands in the Athabasca oil sands, Alberta, Canada. Environ Pollut 158:841–848. doi:10.1016/j.envpol.2009.09.025
Harms NJ, Legagneux P, Gilchrist HG, Bety J, Love OP, Forbes MR, Bortolotti GR, Soos C (2015) Feather corticosterone reveals effect of moulting conditions in the autumn on subsequent reproductive output and survival in an Arctic migratory bird. Proc R Soc B 282:20142085. doi:10.1098/rspb.2014.2085
Harrison GJ, Ritchie BW (1994) Making decisions in the physical examination. Avian medicine: principles and application. Wingers, Lake Worth, pp 144–175
Harrison XA, Blount JD, Inger R, Norris DR, Bearhop S (2011) Carry-over effects as drivers of fitness differences in animals. J Anim Ecol 80:4–18. doi:10.1111/j.1365-2656.2010.01740.x
Hartman FA (1961) Locomotor mechanisms of birds. Smithsonian Misc Collect 143:1–91
Heidinger BJ, Nisbet IC, Ketterson ED (2006) Older parents are less responsive to a stressor in a long-lived seabird: a mechanism for increased reproductive performance with age? Proc R Soc B Biol Sci 273:2227–2231. doi:10.1098/rspb.2006.3557
Hõrak P, Männiste M, Meitern R, Sild E, Saks L, Sepp T (2013) Dexamethasone inhibits corticosterone deposition in feathers of Greenfinches. Gen Comp Endocrinol 191:210–214. doi:10.1016/j.ygcen.2013.07.002
Jenni L, Jenni-Eiermann S, Spina F, Schwabl H (2000) Regulation of protein breakdown and adrenocortical response to stress in birds during migratory flight. Am J Physiol Regul Integr Comp Physiol 278:R1182–R1189
Jenni-Eiermann S, Helfenstein F, Vallat A, Glauser G, Jenni L (2015) Corticosterone: effects on feather quality and deposition into feathers. Methods Ecol Evol 6:237–246. doi:10.1111/2041-210X.12314
Kennedy EA, Lattin CR, Romero LM, Dearborn DC (2013) Feather coloration in museum specimens is related to feather corticosterone. Behav Ecol Sociobiol 67:341–348. doi:10.1007/s00265-012-1454-9
Koren L, Nakagawa S, Burke T, Soma KK, Wynne-Edwards KE, Geffen E (2012) Non-breeding feather concentrations of testosterone, corticosterone and cortisol are associated with subsequent survival in wild House Sparrows. Proc Biol Sci 279:1560–1566. doi:10.1098/rspb.2011.2062
Kouwenberg AL, Mark Hipfner J, McKay DW, Storey AE (2013) Corticosterone and stable isotopes in feathers predict egg size in Atlantic Puffins Fratercula arctica. Ibis 155:413–418. doi:10.1111/ibi.12030
Kouwenberg AL, Hipfner JM, McKay DW, Storey AE (2016) Corticosterone levels in feathers and blood of Rhinoceros Auklets Cerorhinca monocerata are affected by variation in environmental conditions. Mar Biol 163:42. doi:10.1007/s00227-016-2817-y
Kovács A, Mammen UCC, Wernham CV (2008) European monitoring for raptors and Owls: state of the art and future needs. AMBIO J Hum Environ 37:408–412
Landys MM, Ramenofsky M, Wingfield JC (2006) Actions of glucocorticoids at a seasonal baseline as compared to stress-related levels in the regulation of periodic life processes. Gen Comp Endocrinol 148:132–149. doi:10.1016/j.ygcen.2006.02.013
Lattin CR, Reed JM, DesRochers DW, Romero LM (2011) Elevated corticosterone in feathers correlates with corticosterone-induced decreased feather quality: a validation study. J Avian Biol 42:247–252. doi:10.1111/j.1600-048X.2010.05310.x
Legagneux P, Harms NJ, Gauthier G, Chastel O, Gilchrist HG, Bortolotti GR, Bêty J, Soos C (2013) Does feather corticosterone reflect individual quality or external stress in arctic-nesting migratory birds? PLoS One 8:e82644. doi:10.1371/journal.pone.0082644
Lendvai ÁZ, Giraudeau M, Németh J, Bakó V, McGraw KJ (2013) Carotenoid-based plumage coloration reflects feather corticosterone levels in male House Finches (Haemorhous mexicanus). Behav Ecol Sociobiol 67:1817–1824. doi:10.1007/s00265-013-1591-9
López-Jiménez L, Blas J, Tanferna A, Cabezas S, Marchant T, Hiraldo F, Sergio F (2016) Ambient temperature, body condition and sibling rivalry explain feather corticosterone levels in developing Black Kites. Funct Ecol 30:605–613. doi:10.1111/1365-2435.12539
Love OP, Bird DM, Shutt LJ (2003) Corticosterone levels during post-natal development in captive American Kestrels (Falco sparverius). Gen Comp Endocrinol 130:135–141. doi:10.1016/S0016-6480(02)00587-7
Madliger CL, Love OP (2014) The need for a predictive, context-dependent approach to the application of stress hormones in conservation. Conserv Biol 28:283–287. doi:10.1111/cobi.12185
Molina-López RA, Casal J, Darwich L (2011) Causes of morbidity in wild raptor populations admitted at a wildlife rehabilitation centre in Spain from 1995–2007: a long term retrospective study. PLoS One 6:e24603. doi:10.1371/journal.pone.0024603
Molina-López RA, Casal J, Darwich L (2013) Final disposition and quality auditing of the rehabilitation process in wild raptors admitted to a wildlife rehabilitation centre in Catalonia, Spain, during a twelve year period (1995–2007). PLoS One 8:e60242. doi:10.1371/journal.pone.0060242
Molina-López RA, Casal J, Darwich L (2015) Prognostic indicators associated with early mortality of wild raptors admitted to a wildlife rehabilitation centre in Spain. Vet Q 35:9–15. doi:10.1080/01652176.2014.985856
Monadjem A, Wolter K, Neser W, Kane A (2014) Effect of rehabilitation on survival rates of endangered Cape Vultures. Anim Conserv 17:52–60. doi:10.1111/acv.12054
Mormède P, Andanson S, Aupérin B, Beerda B, Guémené D, Malmkvist J, Manteca X, Manteuffel G, Prunet P, van Reenen CG, Richard S, Veisser I (2007) Exploration of the hypothalamic–pituitary–adrenal function as a tool to evaluate animal welfare. Physiol Behav 92:317–339. doi:10.1016/j.physbeh.2006.12.003
Moss D (1979) Growth of nesting Sparrowhawk (Accipiter nisus). J Zool 187:297–314
Möstl E, Rettenbacher S, Palme R (2005) Measurement of corticosterone metabolites in birds’ droppings: an analytical approach. Ann N Y Acad Sci 1046:17–34. doi:10.1196/annals.1343.004
Müller C, Jenni-Eiermann S, Blondel J, Perret P, Caro SP, Lambrechts MM, Jenni L (2007) Circulating corticosterone levels in breeding Blue Tits Parus caeruleus differ between island and mainland populations and between habitats. Gen Comp Endocrinol 154:128–136. doi:10.1016/j.ygcen.2007.05.031
Newton I (1979) Population ecology of raptors. Black, London
Patterson AGL, Kitaysky AS, Lyons DE, Roby DD (2015) Nutritional stress affects corticosterone deposition in feathers of Caspian Tern chicks. J Avian Biol 46:18–24. doi:10.1111/jav.00397
Pérez C, Granadeiro JP, Dias MP, Catry P (2016) Sex and migratory strategy influence corticosterone levels in winter-grown feathers, with positive breeding effects in a migratory pelagic seabird. Oecologia. doi:10.1007/s00442-016-3625-2
Rohwer S, Ricklefs RE, Rohwer VG, Copple MM (2009) Allometry of the duration of the flight feather molt in birds. PLoS Biol 7:e1000132. doi:10.1371/journal.pbio.1000132
Romero LM (2002) Seasonal changes in plasma glucocorticoid concentrations in free-living vertebrates. Gen Comp Endocrinol 128:1–24. doi:10.1016/S0016-6480(02)00064-3
Romero LM (2004) Physiological stress in ecology: lessons from biomedical research. Trends Ecol Evol 19:249–255. doi:10.1016/j.tree.2004.03.008
Romero LM, Fairhurst GD (2016) Measuring corticosterone in feathers: strengths, limitations, and suggestions for the future. Comp Biochem Physiol Part A Mol Integr Physiol. doi:10.1016/j.cbpa.2016.05.002
Romero LM, Reed JM (2005) Collecting baseline corticosterone samples in the field: is under 3 min good enough? Comp Biochem Physiol—A Mol Integr Physiol 140:73–79. doi:10.1016/j.cbpb.2004.11.004
Romero LM, Cyr NE, Romero RC (2006) Corticosterone responses change seasonally in free-living House Sparrows (Passer domesticus). Gen Comp Endocrinol 149:58–65. doi:10.1016/j.ygcen.2006.05.004
Romero LM, Dickens MJ, Cyr NE (2009) The reactive scope model—a new model integrating homeostasis, allostasis, and stress. Horm Behav 55:375–389. doi:10.1016/j.yhbeh.2008.12.009
Saino N, Romano M, Caprioli M, Ambrosini R, Rubolini D, Scandolara C, Romano A (2012) A ptilochronological study of carry-over effects of conditions during wintering on breeding performance in the Barn Swallow Hirundo rustica. J Avian Biol 43:513–524. doi:10.1111/j.1600-048X.2012.05622.x
Sapolsky RM, Romero LM, Munck AU (2000) How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative Actions. Endocr Rev 21:55–89
Schoech SJ, Rensel MA, Bridge ES, Boughton RK, Wilcoxen TE (2009) Environment, glucocorticoids, and the timing of reproduction. Gen Comp Endocrinol 163:201–207. doi:10.1016/j.ygcen.2008.09.009
Schwacke LH, Smith CR, Townsend FI, Wells RS, Hart LB, Balmer BC, Collier TK, Guise SD, Fry MM, Guillette LJ, Lamb SV, Lane SM, McFee WE, Place NJ, Tumlin MC, Ylitalo GM, Zolman ES, Rowles TK (2014) Health of Common Bottlenose Dolphins (Tursiops truncatus) in Barataria Bay, Louisiana, following the Deepwater Horizon oil spill. Environ Sci Technol 48:93–103. doi:10.1021/es403610f
Sheriff MJ, Dantzer B, Delehanty B, Palme R, Boonstra R (2011) Measuring stress in wildlife: techniques for quantifying glucocorticoids. Oecologia 166:869–887. doi:10.1007/s00442-011-1943-y
Sink TD, Lochmann RT, Fecteau KA (2008) Validation, use, and disadvantages of enzyme-linked immunosorbent assay kits for detection of cortisol in Channel Catfish, Largemouth Bass, Red Pacu, and Golden Shiners. Fish Physiol Biochem 34:95–101. doi:10.1007/s10695-007-9150-9
Strong RJ, Pereira MG, Shore RF, Henrys PA, Pottinger TG (2015) Feather corticosterone content in predatory birds in relation to body condition and hepatic metal concentration. Gen Comp Endocrinol 214:47–55. doi:10.1016/j.ygcen.2015.03.002
Terwissen CV, Mastromonaco GF, Murray DL (2013) Influence of adrenocorticotrophin hormone challenge and external factors (age, sex, and body region) on hair cortisol concentration in Canada Lynx (Lynx canadensis). Gen Comp Endocrinol 194:162–167. doi:10.1016/j.ygcen.2013.09.010
Touma C, Palme R (2005) Measuring fecal glucocorticoid metabolites in mammals and birds: the importance of validation. Ann N Y Acad Sci New York Acad Sci 1046:54–74. doi:10.1196/annals.1343.006
Will AP, Suzuki Y, Elliott KH, Hatch SA, Watanuki Y, Kitaysky AS (2014) Feather corticosterone reveals developmental stress in seabirds. J Exp Biol 217:2371–2376. doi:10.1242/jeb.098533
Wingfield JC (2013) Ecological processes and the ecology of stress: the impacts of abiotic environmental factors. Funct Ecol 27:37–44. doi:10.1111/1365-2435.12039
Wingfield JC, Kitaysky AS (2002) Endocrine responses to unpredictable environmental events: stress or anti-stress hormones? Integr Comp Biol 42:600–609. doi:10.1093/icb/42.3.600
Wingfield JC, Sapolsky RM (2003) Reproduction and resistance to stress: when and how. J Neuroendocrinol 15:711–724. doi:10.1046/j.1365-2826.2003.01033.x
Zuberogoitia I, Alonso R, Elorriaga J, Palomares LE, Martínez JA (2009) Moult and age determination of Eurasian Sparrowhawk Accipiter nisus in Spain. Ardeola 56:241–251
Acknowledgements
The authors gratefully acknowledge the staff of Torreferrussa Wildlife Rehabilitation Centre for providing all the samples and data that allowed us to carry out the present study; the Fundación Tatiana Pérez de Guzmán el Bueno for financial support through a pre-doctoral fellowship to M. L.; Dr Joaquim Casellas for his helpful advice on the statistical analysis; and Darío Fernández-Bellon for the English language revision of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by L. Fusani.
Rights and permissions
About this article
Cite this article
Monclús, L., Carbajal, A., Tallo-Parra, O. et al. Relationship between feather corticosterone and subsequent health status and survival in wild Eurasian Sparrowhawk. J Ornithol 158, 773–783 (2017). https://doi.org/10.1007/s10336-016-1424-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-016-1424-5