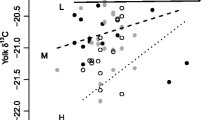
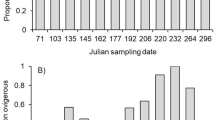
Abstract
Breeding birds use a range of nutrient accumulation and allocation strategies to meet the nutritional demands of clutch formation and incubation. On one end of the spectrum, capital breeders use stored nutrients acquired prior to clutch formation and incubation to sustain metabolism during reproduction, while on the opposite end, income breeders derive nutrients solely from exogenous sources on the breeding grounds. Blue-winged Teal (Anas discors) are an ideal candidate to test for adoption of an income strategy among migratory waterfowl because of their small body size, temperate breeding range, and timing of reproduction relative to pulses in nutrient availability within breeding habitats. We collected migrating and pre-breeding Blue-winged Teal (n = 110) during the warmest spring in over a century in the southern edge of the species’ breeding range, which produced ideal conditions to test for adoption of an income breeding strategy among migratory waterfowl. Regression analyses revealed that females accumulated protein and fat reserves early in follicle development and appeared to mobilize at least some reserves coincident with the onset of clutch formation. Accumulation and subsequent mobilization of nutrient reserves was inconsistent with adherence to an income breeding strategy and suggested breeding Blue-winged Teal used capital (albeit locally acquired) for reproduction. Our results add to existing knowledge on the ubiquity of endogenous nutrient reserve accumulation prior to and during reproduction by waterfowl, perhaps suggesting endogenous nutrient reserves are universally used for clutch formation or incubation to some degree. If indeed Blue-winged Teal and other waterfowl universally use capital for breeding, research and conservation efforts should shift from evaluating whether an income breeding strategy is used and focus on when and where necessary capital is acquired prior to clutch formation.
Zusammenfassung
Ist „income breeding“ ein angemessenes Modell für Wasservögel?
Brutvögel setzen eine Reihe von Strategien für Erwerb und Zuweisung von Nahrung ein, um die Bedürfnisse bei Eiablage und Brut zu befriedigen. Am einen Ende des Spektrums verwenden die „capital breeders“ gespeicherte Nährstoffe, die sie vor Beginn der Eiablage und des Brütens aufgenommen haben, um während des Brütens ihren Stoffwechsel aufrechtzuerhalten; am entgegengesetzten Ende des Spektrums beziehen die „income breeders“ ihre Nährstoffe ausschließlich aus exogenen Quellen aus der Umgebung des Nistplatzes. Blauflügelenten (Anas discors) sind das ideale Modell, den Einsatz der „income“ Strategie bei Wassergeflügel zu testen, aufgrund ihrer geringen Körpergröße, dem Brutareal in gemäßigtem Klima und der zeitlichen Anpassung der Fortpflanzung relativ zu Schwankungen in der Nahrungsverfügbarkeit in den Bruthabitaten. Wir fingen ziehende Blauflügelenten vor dem Brüten (n = 110) im wärmsten Frühling seit über 100 Jahren am südlichsten Rand des Verbreitungsgebiets der Art, was ideale Bedingungen ergab, um auf das Vorhandensein einer „income“ Strategie bei ziehenden Wasservögeln zu testen. Regressionsanalysen zeigten, dass Weibchen Protein- und Fettreserven früh in der Entwicklung des Follikels anlegten, und sie schienen zumindest einen Teil der Reserven gleichzeitig mit Beginn der Eiablage zu mobilisieren. Anlage und anschließende Mobilisierung von Nährstoffreserven widersprachen einer „income“ Strategie und legten nahe, dass brütende Blauflügelenten auf zuvor angelegte Vorräte („capital“) zurückgriffen. Unsere Ergebnisse unterstützen bisherige Erkenntnisse über die allgemeine Verbreitung des Aufbaus endogener Nährstoffreserven bei Wasservögeln vor Brutbeginn, und weisen möglicherweise darauf hin, dass endogene Nährstoffreserven universell zu gewissen Teilen bei Eiablage und Brüten zum Einsatz kommen. Wenn Blauflügelenten und andere Wasservögel tatsächlich Vorräte („capital“) universell fürs Brüten einsetzen, sollte sich der Focus von Forschung und Artenschutz davon abwenden, zu untersuchen, ob eine „income breeding“ Strategie eingesetzt wird, und sich der Frage zuwenden, wann und wo die nötigen Vorräte („capital“) vor der Eiablage angelegt werden.




Similar content being viewed by others
References
Afton AD (1980) Factors affecting incubation rhythms of northern shovelers. Condor 82:132–137. doi:10.2307/1367462
Afton AD, Ankney CD (1991) Nutrient-reserve dynamics of breeding lesser scaup: a test of competing hypotheses. Condor 93:89–97
Alisauskas RT, Ankney CD (1992) The cost of egg laying and its relationship to nutrient reserves in waterfowl. In: Batt BDJ (ed) Ecology and management of breeding waterfowl. University of Minnesota Press, Minneapolis, pp 30–61
Alisauskas RT, Ankney CD (1994) Nutrition of breeding female ruddy ducks: the role of nutrient reserves. Condor 96:878–897. doi:10.2307/1369099
Alisauskas RT, DeVink JM (2015) Dealing with deficits: breeding costs, nutrient reserves, and cross-seasonal effects in sea ducks. In: Savard JPL, Derksen DV, Esler D, Eadie JM (eds) Ecology and Conservation of North American Sea Ducks. Studies in Avian Biology. CRC Press, Boca Raton, pp 115–148
Alisauskas RT, Eberhardt RT, Ankney CD (1990) Nutrient reserves of breeding ring-necked ducks (Aythya collaris). Can J Zool 68:2524–2530. doi:10.1139/z90-353
Ankney CD, Alisauskas RT (1991) Nutrient-reserve dynamics and diet of breeding female gadwalls. Condor 93:799–810
Ankney CD, MacInnes CD (1978) Nutrient reserves and reproductive performance of female lesser snow geese. Auk 95:459–471. doi:10.2307/4085149
Ankney CD, Scott DM (1980) Changes in nutrient reserves and diet of breeding brown-headed cowbirds. Auk 97:684–696. doi:10.2307/4085740
Anteau MJ, Afton AD (2004) Nutrient reserves of lesser scaup (Aythya affinis) during spring migration in the Mississippi Flyway: a test of the spring condition hypothesis. Auk 121:917–929. doi:10.1642/0004-8038(2004)121[0917:nrolsa]2.0.co;2
Anteau MJ, Afton AD (2009) Lipid reserves of Lesser Scaup (Aythya affinis) migrating across a large landscape are consistent with the “Spring Condition” hypothesis. Auk 126:873–883. doi:10.1525/auk.2009.08193
Arnold TW (2010) Uninformative parameters and model selection using Akaike’s Information Criterion. J Wildl Manage 74:1175–1178. doi:10.2193/2009-367
Arzel C, Elmberg J, Guillemain M (2006) Ecology of spring-migrating Anatidae: a review. J Ornithol 147:167–184. doi:10.1007/s10336-006-0054-8
Arzel C, Dessborn L, Pöysä H, Elmberg J, Nummi P, Sjöberg K (2014) Early springs and breeding performance in two sympatric duck species with different migration strategies. Ibis 156:288–298. doi:10.1111/ibi.12134
Barzen JA, Serie JR (1990) Nutrient reserve dynamics of breeding canvasbacks. Auk 107:75–85. doi:10.2307/4087804
Bellrose FC (1976) Ducks, geese & swans of North America, 2nd edn. Stackpole Books, Harrisburg
Bêty J, Gauthier G, Giroux J (2003) Body condition, migration, and timing of reproduction in snow geese: a test of the condition-dependent model of optimal clutch size. Am Nat 162:110–121
Bond JC, Esler D, Hobson KA (2007) Isotopic evidence for sources of nutrients allocated to clutch formation by harlequin ducks. Condor 109:698–704. doi:10.1650/8241.1
Bromley RG, Jarvis RL (1993) The energetics of migration and reproduction of dusky Canada geese. The Condor 95:193–210. doi:10.2307/1369400
Cherel Y, Robin J-P, Maho YL (1988) Physiology and biochemistry of long-term fasting in birds. Can J Zool 66:159–166. doi:10.1139/z88-022
Cutting KA, Hobson KA, Rotella JJ, Warren JM, Wainwright-de la Cruz SE, Takekawa JY (2011) Endogenous contributions to egg protein formation in lesser scaup Aythya affinis. J Avian Biol 42:505–513. doi:10.1111/j.1600-048X.2011.05406.x
Cutting KA, Hobson KA, Rotella JJ, Warren JM, Takekawa JY, De La Cruz SEW, Parker M (2014) Relative contribution of lipid sources to eggs of lesser scaup. J Avian Biol 45:197–201. doi:10.1111/j.1600-048X.2013.00238.x
Devink J-M, Clark RG, Slattery SM, Trauger DL (2008) Are late-spring boreal lesser scaup (Aythya affinis) in poor body condition? Auk 125:291–298
Devink JM, Slattery SM, Clark RG, Alisauskas RT, Hobson KA (2011) Combining stable-isotope and body-composition analyses to assess nutrient-allocation strategies in breeding white-winged scoters (Melanitta fusca). Auk 128:166–174. doi:10.1525/auk.2010.10069
Drent RH, Daan S (1980) The prudent parent: energetic adjustments in avian breeding? Ardea 68:225–252
Drent R, Both C, Green M, Madsen J, Piersma T (2003) Pay-offs and penalties of competing migratory schedules. Oikos 103:274–292. doi:10.1034/j.1600-0706.2003.12274.x
Drent RJ, Fox AD, Stahl J (2006) Travelling to breed. J Ornithol 147:122–134. doi:10.1007/s10336-006-0066-4
Drever MC, Clark RG (2007) Spring temperature, clutch initiation date and duck nest success: a test of the mismatch hypothesis. J Anim Ecol 76:139–148. doi:10.1111/j.1365-2656.2006.01183.x
Elmberg J, Nummi P, Poysa H, Gunnarsson G, Sjoberg K (2005) Early breeding teal Anas crecca use the best lakes and have the highest reproductive success. Ann Zool Fenn 42:37–43
Esler D, Grand JB (1994) The role of nutrient reserves for clutch formation by northern pintails in Alaska. Condor 96:422–432
Esler D, Grand JB, Afton AD (2001) Intraspecific variation in nutrient reserve use during clutch formation by lesser scaup. Condor 103:810–820
Ganter B, Cooke F (1996) Pre-incubation feeding activities and energy budgets of snow geese: can food on the breeding grounds influence fecundity? Oecologia 106:153–165. doi:10.2307/4221243
Gaunt AS, Hikida RS, Jehl JR Jr, Fenbert L (1990) Rapid atrophy and hypertrophy of an avian flight muscle. Auk 107:649–659. doi:10.2307/4087994
Gauthier G, Bêty J, Hobson KA (2003) Are greater snow geese capital breeders? New evidence from a stable-isotope model. Ecology 84:3250–3264. doi:10.1890/02-0613
Gorman KB, Esler D, Flint PL, Williams TD (2008) Nutrient-reserve dynamics during egg production by female Greater Scaup (Aythya marila): relationships with timing of reproduction. Auk 125:384–394. doi:10.1525/auk.2008.06151
Guillemain M, Elmberg J, Arzel C, Johnson AR, Simon G (2008) The income-capital breeding dichotomy revisited: late winter body condition is related to breeding success in an income breeder. Ibis 150:172–176
Hobson KA, Atwell L, Wassenaar LI, Yerkes T (2004) Estimating endogenous nutrient allocations to reproduction in Redhead Ducks: a dual isotope approach using delta D and delta C-13 measurements of female and egg tissues. Funct Ecol 18:737–745. doi:10.1111/j.0269-8463.2004.00890.x
Hohman WL, Taylor TS (1986) Indices of fat and protein for ring-necked ducks. J Wildl Manag 50:209–211. doi:10.2307/3801898
Houston DC, Donnan D, Jones PJ (1995) The source of the nutrients required for egg production in zebra finches Poephila guttata. J Zool 235:469–483. doi:10.1111/j.1469-7998.1995.tb01763.x
Jönsson KI (1997) Capital and income breeding as alternative tactics of resource use in reproduction. Oikos 78:57–66. doi:10.2307/3545800
Klaassen M (2003) Relationships between migration and breeding strategies in Arctic breeding birds. In: Berthold P, Gwinner E, Sonnenschein E (eds) Avian Migration. Springer, Berlin Heidelberg, pp 237–249. doi:10.1007/978-3-662-05957-9_16
Klaassen M, Abraham KF, Jefferies RL, Vrtiska M (2006) Factors affecting the site of investment, and the reliance on savings for arctic breeders: the capital-income dichotomy revisited. Ardea 94:371–384
Krapu GL (1981) The role of nutrient reserves in mallard reproduction. Auk 98:29–38
Lack D (1948) Natural selection and family size in the starling. Evolution 2:95–110. doi:10.2307/2405371
MacCluskie MC, Sedinger JS (2000) Nutrient reserves and clutch-size regulation of Northern Shovelers in Alaska. Auk 117:971–979. doi:10.1642/0004-8038(2000)117[0971:nracsr]2.0.co;2
Meijer T, Drent R (1999) Re-examination of the capital and income dichotomy in breeding birds. Ibis 141:399–414. doi:10.1111/j.1474-919X.1999.tb04409.x
National Climate Data Center [NCDC] (2015) National temperature and precipitation maps. http://www.ncdc.noaa.gov/cag/. Accessed 10 January 2015
Owen RB, Reinecke KJ (1979) Bioenergetics of breeding dabbling ducks. Paper presented at the Waterfowl and Wetlands—an integrated review. In: Proceedings of 39th Midwest Fish and Wildlife Conference
Perrins CM (1965) Population fluctuations and clutch-size in the great tit, Parus major L. J Anim Ecol 34:601–647. doi:10.2307/2453
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing. Vienna
Ryder JP (1970) A possible factor in the evolution of clutch size in Ross’ goose. The Wilson Bulletin 82:5–13
Sedinger JS, Alisauskas RT (2014) Cross-seasonal effects and the dynamics of waterfowl populations. Wildfowl 4:277–304
Stephens PA, Boyd IL, McNamara JM, Houston AI (2009) Capital breeding and income breeding: their meaning, measurement, and worth. Ecology 90:2057–2067. doi:10.1890/08-1369.1
Swanson GA, Meyer MI, Serie JR (1974) Feeding ecology of breeding blue-winged teals. J Wildl Manage 38:396–407
Thomas VG (1988) Body condition, ovarian hierarchies, and their relation to egg formation in anseriform and galliform species. Paper presented at the Acta XIX Congressus Internationalis Ornithologici, National Museum of Natural Science, Ottawa
Williams TD, Martyniuk CJ (2000) Tissue mass dynamics during egg-production in female Zebra Finches Taeniopygia guttata: dietary and hormonal manipulations. J Avian Biol 31:87–95. doi:10.1034/j.1600-048X.2000.310112.x
Witter MS, Cuthill IC (1993) The ecological costs of avian fat storage Philosophical Transactions of the Royal Society of London Series B: Biological. Sciences 340:73–92. doi:10.1098/rstb.1993.0050
Acknowledgments
Our research was supported through grants from the South Dakota Department of Game, Fish and Parks, the Department of Natural Resource Management at South Dakota State University, the Kenneth F. Higgins Waterfowl Legacy Research Endowment, and the US Geological Survey through the Northern Prairie Wildlife Research Center and South Dakota Cooperative Fish and Wildlife Research Unit. EnvrionMetal, Inc., made in-kind contributions to the research. R. Murano, P. Mammenga, and M. Grovijahn assisted with the collections and logistics of the field research. J. Rorah and A. Homola assisted with laboratory work. J. DeVink, R. Alisauskas, and one anonymous reviewer provided insightful comments that improved the scope and content of the manuscript. All experimental procedures described herein were reviewed and approved by the Institutional Animal Care and Use Committee at South Dakota State University (permit 12-013A) and authorized under a US Fish and Wildlife Service scientific collecting permit (permit MB068976-0). Any use of trade, product, or firm names is for descriptive purposes only and does not imply endorsement by the US Government.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. G. Guglielmo.
Rights and permissions
About this article
Cite this article
Janke, A.K., Anteau, M.J., Markl, N. et al. Is income breeding an appropriate construct for waterfowl?. J Ornithol 156, 755–762 (2015). https://doi.org/10.1007/s10336-015-1200-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-015-1200-y