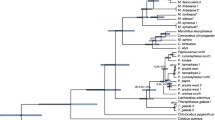
Abstract
Howler monkeys (Alouatta), comprising between nine and 14 species and ranging from southern Mexico to northern Argentina, are the most widely distributed platyrrhines. Previous phylogenetic studies of howlers have used chromosomal and morphological characters and a limited number of molecular markers; however, branching patterns conflict between studies or remain unresolved. We performed a new phylogenetic analysis of Alouatta using both concatenated and coalescent-based species tree approaches based on 14 unlinked non-coding intergenic nuclear regions. Our taxon sampling included five of the seven South American species (Alouatta caraya, Alouatta belzebul, Alouatta guariba, Alouatta seniculus, Alouatta sara) and the two recognized species from Mesoamerica (Alouatta pigra, Alouatta palliata). Similarly to previous studies, our phylogenies supported a Mesoamerican clade and a South American clade. For the South American howlers, both methods recovered the Atlantic Forest endemic A. guariba as sister to all remaining South American species, albeit with moderate support. Moreover, we found no support for the previously proposed sister relationship between A. guariba and A. belzebul. For the first time, a clade composed of A. sara and A. caraya was identified. The relationships among the other South American howlers, however, were not fully supported. Our estimates for divergence times within Alouatta are generally older compared to estimates in earlier studies. However, they conform to recent studies proposing a Miocene age for the Isthmus of Panama and for the uplift of the northern Andes. Our results also point to an early genetic isolation of A. guariba in the Atlantic Forest, in agreement with the hypothesis of biotic exchange across South American rain forests in the Miocene. Collectively, these findings contribute to a better understanding of the diversification processes among howler monkey species; however, they also suggest that further comprehension of the evolutionary history of the Alouatta radiation will rely on broadened taxonomic, geographic, and genomic sampling.






Similar content being viewed by others
References
Arroyo-Rodríguez V, Andresen E, Bravo SP, Stevenson PR (2015) Seed dispersal by howlers: current knowledge, conservation implications, and future directions. In: Howler monkeys. Springer, New York, pp 111–139
Bacon CD, Silvestro D, Jaramillo C, Smith BT, Chakrabarty P, Antonelli A (2015) Biological evidence supports an early and complex emergence of the Isthmus of Panama. Proc Natl Acad Sci 112(19):6110–6115
Bigarella JJ (1975) Considerações a respeito das mudanças paleoambientais na distribuição de algumas espécies vegetais e animais no Brasil. An Acad Bras Cienc 47:411–464
Cortés-Ortiz L, Bermingham E, Rico C, Rodrıguez-Luna E, Sampaio I, Ruiz-Garcıa M (2003) Molecular systematics and biogeography of the Neotropical monkey genus, Alouatta. Mol Phylogenet Evol 26(1):64-81
Cortés-Ortiz L, Rylands AB, Mittermeier RA (2015) The taxonomy of howler monkeys: integrating old and new knowledge from morphological and genetic studies. In: Kowalewski MM, Garber PA, Cortés-Ortiz L, Urbani B, Youlatos D (eds) Howler monkeys: adaptive radiation, systematics, and morphology. Springer, New York, pp 55–84
Costa LP (2003) The historical bridge between the Amazon and Atlantic Forest of Brazil: a study of molecular phylogeography with small mammals. J Biogeogr 30:71–86
Crockett CM, Eisenberg JF (1986) Howlers: variation in group sizes and demography. In: Smuts BB, Cheney DL, Seyfarth RM, Wrangham RW, Struhsaker TT (eds) Primate societies. University of Chicago, Chicago, pp 54–68
Dal Vechio F, Prates I, Grazziotin FG, Zaher H, Rodrigues MT (2018) Phylogeography and historical demography of the arboreal pit viper Bothrops bilineatus (Serpentes, Crotalinae) reveal multiple connections between Amazonian and Atlantic rainforests. J Biogeog 45:2415–2426
Dal Vechio F, Prates I, Grazziotin FG, Zaher H, Grabosky R, Rodrigues MT (2020) Rain forest shifts through time and riverine barriers shaped the diversification of South American terrestrial pit vipers (Bothrops jararacussu species group). J Biogeogr 47(2):516–526
de Moura AC, McConkey KR (2007) The capuchin, the howler, and the caatinga: seed dispersal by monkeys in a threatened Brazilian forest. Am J Primatol 69(2):220–226
de Oliveira EHC, De Lima MMC, Sbalqueiro IJ, Dasilva AF (1999) Analysis of polimorphic NORs in Alouatta species (Primates, Atelidae). Caryologia 52(3–4):169–175
de Oliveira EH, Neusser M, Figueiredo WB, Nagamachi C, Pieczarka JC, Sbalqueiro IJ, Wienberg J, Müller S (2002) The phylogeny of howler monkeys (Alouatta, Platyrrhini): reconstruction by multicolor cross-species chromosome painting. Chromosome Res 10(8):669–683
Degnan JH, Rosenberg NA (2009) Gene tree discordance, phylogenetic inference and the multispecies coalescent. Trends Ecol Evol 24(6):332–340
Delson E, Rosenberger AL (1984) Are there any anthropoid primate living fossils? Living fossils. Springer, New York, pp 50–61
Dias PA, Rangel-Negrín A (2015) Diets of howler monkeys. Howler monkeys. Springer, New York, pp 21–56
Drummond AJ, Suchard MA, Xie D, Rambaut A (2012) Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol Biol Evol 29:1969–1973
Drummond AJ, Ho SY, Phillips MJ, Rambaut A (2006) Relaxed phylogenetics and dating with confidence. PLoS Biol 4:e88
Edwards SV (2009) Is a new and general theory of molecular systematics emerging? Evolution 63(1):1–19
Edwards SV, Xi Z, Janke A, Faircloth BC, McCormack JE, Glenn TC, Zhong B, Wu S, Lemmon EM, Lemmon AR, Leaché AD, Liu L, Davis CC (2016) Implementing and testing the multispecies coalescent model: a valuable paradigm for phylogenomics. Mol Phylogenet Evol 94(Pt A):447–462
Evenstar LA, Stuart FM, Hartley AJ, Tattitch B (2015) Slow Cenozoic uplift of the western Andean Cordillera indicated by cosmogenic 3He in alluvial boulders from the Pacific Planation Surface. Geophys Res Lett 42(20):8448–8455
Figueiredo WB, Carvalho-Filho NM, Schneider H, Sampaio I (1998) Mitochondrial DNA sequences and the taxonomic status of Alouatta seniculus populations in northeastern Amazonia. Neotrop Primates 6:73–77
Fleagle JG, Kay RF, Anthony MRL (1997) Fossil New World monkeys. In: Kay RF, Madden RH, Cifelli RL, Flynn JJ (eds) Vertebrate paleontology in the Neotropics: the Miocene fauna of La Venta, Colombia. Smithsonian Institution Press, Washington, pp 473–495
Flot JF (2010) seqphase: a web tool for interconverting phase input/output files and fasta sequence alignments. Mol Ecol Resour 10(1):162–166
Flynn JJ, Guerrero J, Swisher CC (1997) Geochronology of the Honda Group, Columbia. In: Kay R, Madden R, Cifelli R, Flynn J (eds) Vertebrate paleontology in the Neotropics. The Miocene fauna of La Venta. Smithsonian Institution Press, Colombia, pp 44–59
Garzione CN, Hoke GD, Libarkin JC, Withers S, MacFadden B, Eiler J, Ghosh P, Mulch A (2008) Rise of the Andes. Science 320(5881):1304–1307
Graur D, Martin W (2004) Reading the entrails of chickens: molecular timescales of evolution and the illusion of precision. Trends Genet 20(2):80–86
Gregorin P (2006) Taxonomia e variação geográfica das espécies do gênero Alouatta Lacépède (Primates, Atelidae) no Brasil. Rev Bras Zool 23:64–144
Haffer J (1969) Speciation in Amazonian forest birds. Science 165(3889):131–137
Harris EE, Gifalli-Iughetti C, Braga ZH, Koiffman CP (2005) Cytochrome b sequences show subdivision between populations of the brown howler monkey (Alouatta guariba) from Rio de Janeiro and Santa Catarina, Brazil. Neotrop Primates 13:16–21
Hartwig W, Meldrum DJ (2002) Miocene platyrrhines of the northern Neotropics. In: Hartwig WC (ed) The primate fossil record. Cambridge University, Cambridge, pp 175–188
Heled J, Drummond J (2010) Bayesian inference of species trees from multilocus data. Mol Biol Evol 27:570–580
Hershkovitz P (1949) Mammals of Northern Colombia, preliminary report no. 4: monkeys (Primates), with taxonomic revisions of some forms. Proc US Natl Mus 98(3232):323–427
Hershkovitz P (1970) Notes on Tertiary platyrrhine monkeys and description of a new genus from the late Miocene of Colombia. Folia Primatol 12:1–37
Hill WCO (1962) Primates comparative anatomy and taxonomy. V. Cebidae, part B. Edinburgh University Press, Edinburgh
Ho SY, Phillips MJ (2009) Accounting for calibration uncertainty in phylogenetic estimation of evolutionary divergence times. Syst Biol 58(3):367–380
International Union for Conservation of Nature (IUCN) (2019) The IUCN Red List of Threatened Species. Version 2019. https://www.iucnredlist.org. Downloaded in 2019.
Kay RF, Cozzuol MA (2006) New platyrrhine monkeys from the Solimões Formation (Late Miocene, Acre State, Brazil). J Hum Evol 50:673–686
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Buxton S, Cooper A, Markowitz S, Duran C, Thierer T, Ashton B, Meintjes P, Drummond A (2012) Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28(12):1647–1649
Kiesling NMJ, Soojin VY, Xu K, Sperone FG, Wildman DE (2015) The tempo and mode of New World monkey evolution and biogeography in the context of phylogenomic analysis. Mol Phylogenet Evol 82:386–399
Maddison WP, Knowles LL (2006) Inferring phylogeny despite incomplete lineage sorting. Syst Biol 55(1):21–30
Meireles CM, Czelusniak J, Schneider MP, Muniz JA, Brigido MC, Ferreira HS, Goodman M (1999) Molecular phylogeny of Ateline New World monkeys (Platyrrhini, Atelinae) based on γ-globin gene sequences: evidence that Brachyteles is the sister group of Lagothrix. Mol Phylogenet Evol 12(1):10–30
Montes C, Cardona A, Jaramillo C, Pardo A, Silva JC, Valencia V, Ayala C, Pérez-Angel LC, Rodriguez-Parra LA, Ramirez V, Niño H (2015) Middle Miocene closure of the Central American Seaway. Science 348(6231):226–229
Molnar P, Garzione CN (2007) Bounds on the viscosity coefficient of continental lithosphere from removal of mantle lithosphere beneath the Altiplano and Eastern Cordillera. Tectonics 26(2):1–9
Moore WS (1995) Inferring phylogenies from mtDNA variation: mitoschondrial-gene trees versus nuclear-gene trees. Evolution 49(4):718–726
Pamilo P, Nei M (1988) Relationships between gene trees and species trees. Mol Biol Evol 5:568–583
Posada D (2008) jModelTest: phylogenetic model averaging. Mol Biol Evol 25(7):1253–1256
Pozzi L, Disotell TR, Masters JC (2014) A multilocus phylogeny reveals deep lineages within African galagids (Primates: Galagidae). BMC Evol Biol 14:72
Prates I, Rivera D, Rodrigues MT, Carnaval AC (2016) A mid Pleistocene rainforest corridor enabled synchronous invasion of the Atlantic Forest by Amazonian anole lizards. Mol Ecol 25:5174–5186
Prates I, Melo-Sampaio PR, Drummond LO, Teixeira Jr M, Rodrigues MT, Carnaval AC (2017) Biogeographic links between southern Atlantic Forest and western South America: rediscovery, redescription, and phylogenetic relationships of two rare montane anole lizards from Brazil. Mol Phyl Evol 113:49–58
Prates I, Penna A, Rodrigues MT, Carnaval AC (2018) Local adaptation in mainland anole lizards: integrating population history and genome-environment associations. Ecol Evol 8(23):11932–11944
Prates I, Melo-Sampaio PR, de Queiroz K, Carnaval AC, Rodrigues MT, Drummond LO (2020) Discovery of a new species of Anolis lizards from Brazil and its implications for the historical biogeography of montane Atlantic Forest endemics. Amphibia-Reptilia 41(1):87–103
Rambaut A, Drummond AJ (2009) FigTree version 1.3.1 (http://tree.bio.ed.ac.uk/software/figtree/)
Rambaut A, Drummond AJ, Suchard M (2009) Tracer, version 1.5 for Macintosh. [Computer program and documentation distributed by the authors (http://beast.bio.ed.ac.uk/Tracer)]
Rosenberger AL (1992) Evolution of feeding niches in New World monkeys. Am J Phys Anthropol 88:525–562
Rosenberger AL, Cooke SB, Halenar LB, Tejedor MF, Hartwig WC, Novo NM, Muñoz-Saba Y (2015) Fossil alouattines and the origins of Alouatta: craniodental diversity and interrelationships. Howler monkeys. Springer, New York, pp 21–54
Ronquist F, Teslenko M, van der Mark P, Ayres DL, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542
Sambrock J, Fritsch E, Maniatis T (1989) Molecular cloning: a laboratory manual, vol 1, 2nd edn. Cold Spring Harbor Laboratory Press, New York
Sanderson MJ (1997) A nonparametric approach to estimating divergence times in the absence of rate constancy. Mol Phylogenet Evol 14:1218–1231
Sauquet HSY, Gandolfo MA, Jordan GJ, Wilf P, Cantrill DJ, Bayly MJ, Bromham L, Brown GK, Carpenter RJ, Lee DM, Murphy DJ, Sniderman JM, Udovicic F (2012) Testing the impact of calibration on molecular divergence times using a fossil-rich group: the case of Nothofagus (Fagales). Syst Biol 61(2):289–313
Schneider H, Sampaio I, Harada ML, Barroso CM, Schneider MP, Czelusniak J, Goodman M (1996) Molecular phylogeny of the New World monkeys (Platyrrhini, Primates) based on two unlinked nuclear genes: IRBP intron 1 and epsilon-globin sequences. Am J Phys Anthropol 100(2):153–179
Schneider H, Canavez FC, Sampaio I, Moreira MÂ, Tagliaro CH, Seuánez HN (2001) Can molecular data place each Neotropical monkey in its own branch? Chromosoma 109(8):515–523
Setoguchi T, Watanabe T, Mouri T (1981) The upper dentition of Stirtonia (Ceboidea, Primates) from the Miocene of Colombia, South America and the origin of the postero-internal cusp of upper molars of howler monkeys (Alouatta). Kyoto Univ Overseas Res Rep New World Monkeys 3:51–60
Silvestro D, Tejedor MF, Serrano-Serrano ML, Loiseau O, Rossier V, Rolland J, Zizka A, Antonelli A, Salamin N (2017) Evolutionary history of New World monkeys revealed by molecular and fossil data. bioRxiv 1:178111
Shedden-González A, Rodríguez-Luna E (2010) Responses of a translocated howler monkey Alouatta palliata group to new environmental conditions. Endanger Species Res 12(1):25–30
Stephens M, Donnelly P (2003) A comparison of Bayesian methods for haplotype reconstruction from population genotype data. Am J Hum Genet 73:1162–1169
Stirton RA (1951) Ceboid monkeys from the Miocene of Colombia. Univ Calif Pub Geol Sci 28:315–356
Szalay FS, Delson E (1979) Evolutionary history of the primates. Academic Press, New York
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Villalobos F, Valerio AA, Retana AP (2004) A phylogeny of howler monkeys (Cebidae: Alouatta) based on mitochondrial, chromosomal, and morphological data. Rev Biol Trop 52:665–677
Weisrock DW, Smith SD, Chan LM, Biebouw K, Kappeler PM, Yoder AD (2012) Concatenation and concordance in the reconstruction of mouse lemur phylogeny: an empirical demonstration of the effect of allele sampling in phylogenetics. Mol Biol Evol 29(6):1615–1630
Wiens JJ, Kuczynski CA, Smith SA, Mulcahy DG, Sites JW Jr, Townsend TM, Reeder TW (2008) Branch lengths, support, and congruence: testing the phylogenomic approach with 20 nuclear loci in snakes. Syst Biol 57(3):420-431
Wildman DE, Jameson NM, Opazo JC, Soojin VY (2009) A fully resolved genus level phylogeny of Neotropical primates (Platyrrhini). Mol Phylogenet Evol 53(3):694–702
Zunino GE, González V, Kowalewski MM, Bravo SP (2001a) Alouatta caraya: relations among habitat, density and social organization. Primate Rep 61:37–46
Zunino GE, Gonzalez V, Kowalewski MM, Bravo SP (2001b) Alouatta caraya relations among habitat density and social organization. Primate Rep 61:37–45
Zunino GE, Kowalewski MM, Oklander LI, González V (2007) Habitat fragmentation and population size of the black and gold howler monkey (Alouatta caraya) in a semideciduous forest in northern Argentina. Am J Primatol 69(9):966–975
Acknowledgments
We thank Dr. Derek Wildman (University of Illinois), Cristiani Gifalli-Iughetti (University of São Paulo), and Dr. Liliana Cortés-Ortiz (University of Michigan) for graciously sharing DNA samples of Alouatta with us. We also thank the various members of the Carnaval lab for feedback on the initial manuscript and University of São Paulo/Hemocentro Ribeirão Preto members for assistance with DNA sequencing. This research was funded by the City College of New York, the National Science Foundation (NSF), and a Professional Staff Congress-City University of New York award (66309-0044) to E.E.H.; A. Carnaval acknowledges funding by the NSF, the São Paulo Research Foundation, and the National Aeronautics and Space Administration, through the Dimensions of Biodiversity Program (DEB 1343578).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Doyle, E.D., Prates, I., Sampaio, I. et al. Molecular phylogenetic inference of the howler monkey radiation (Primates: Alouatta). Primates 62, 177–188 (2021). https://doi.org/10.1007/s10329-020-00854-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10329-020-00854-x