Abstract
Objective
To assess the course of COVID-19 infections and the tolerability of the mRNA vaccines of Moderna and Pfizer/BioNTech and the viral vector vaccines from Astra Zeneca and Johnson & Johnson in adult patients with epilepsy (PWE).
Methods
From July 2020 to July 2021, we consecutively included adult outpatients with confirmed epilepsy. These PWE were interviewed about COVID-19 infections and vaccinations. Results of follow-up visits were added until the cut-off date (December 31, 2021). The data of COVID-19-infected without vaccinations or fully vaccinated PWE without COVID-19 infections were analyzed. Full vaccination was defined as a double vaccination with the Pfizer/BionTech, Moderna, or Astra Zeneca vaccines or a single Johnson & Johnson vaccination.
Results
At cut-off, 612 of 1152 PWE fulfilled the inclusion criteria: 51 PWE had been infected without vaccination and 561 had full vaccination without infection. Among the infected PWE, 76.5% presented with symptoms; 9.8% had a severe course (one death). The leading symptoms were influenza-like disorders (48.7% of infected PWE with symptoms), anosmia (28.2%), and ageusia (20.5%). Seizure increases or relapses after sustained seizure freedom occurred in 7.8%. Adverse events (AEs) were reported by 113 vaccinated PWE (20.1% of all vaccinated PWE). The leading AEs were fatigue, fever, and headache. The AE rate per vaccine was 14.0% for Pfizer/BionTech, 32.7% for Moderna, 25.8% for Astra Zeneca, and 46.2% for Johnson & Johnson. Of the AEs, 93.3% lasted ≤ 1 week. Seizure increase or relapse occurred in 1.4% and was significantly less frequent than in the infected group (p = 0.0016).
Conclusion
The course of COVID-19 infections and the tolerability of the vaccines were similar as in the general population, yet, seizure worsening occurred more often after the infection than after the vaccination.
Zusammenfassung
Zielsetzung
Auswertung des Verlaufs von COVID-19-Infektionen und der Verträglichkeit der mRNA-Impfstoffe der Fa. Moderna und Fa. Pfizer/BioNTech sowie der viralen Impfstoffe der Fa. Astra Zeneca und der Fa. Johnson & Johnson bei erwachsenen Epilepsiepatienten.
Methoden
Von Juli 2020 bis Juli 2021 wurden erwachsene Ambulanzpatienten mit gesicherten Epilepsien konsekutiv erfasst. Sie wurden hinsichtlich Infektionen und Impfungen befragt. Ergaben sich zusätzliche Informationen bei diesen Patienten bis zum Zeitpunkt der Auswertung (31.12.2021), wurden diese Zusatzdaten berücksichtigt. Ausgewertet wurden dann nur Daten von nicht geimpften Patienten mit gesicherter Infektion oder von vollständig geimpften Patienten ohne Infektion. Als vollständig wurden 2 Impfungen mit den Impfstoffen von Fa. Pfizer/BionTech, Fa. Moderna oder Fa. Astra Zeneca oder eine Einzelimpfung mit der Vakzine von Fa. Johnson & Johnson definiert.
Ergebnisse
Insgesamt erfüllten 612 von 1152 Patienten die Einschlusskriterien. Über eine gesicherte Infektion berichteten 51 Patienten. Vollständig geimpft waren 561. Von den Infizierten waren 76,5 % symptomatisch. Einen schweren Verlauf mit Hospitalisierung (ein Todesfall) wiesen 9,8 % auf. Die führenden Symptome waren grippeartig (48,7 % der infizierten symptomatischen Patienten) oder bestanden aus Anosmie (28,2 %) und Ageusie (20,5 %). Eindeutige Anfallszunahmen oder Anfälle nach langer Anfallsfreiheit betrafen 7,8 % der Patienten. Über unerwünschte Ereignisse nach Impfungen berichteten 113 Patienten (20,1 % aller geimpften Personen). Die häufigsten Symptome waren Müdigkeit, Fieber und Kopfschmerzen. Die Häufigkeit unerwünschter Ereignisse pro Impfstoff betrug 14,0 % für Pfizer/BionTech, 32,7 % für Moderna, 25,8 % für Astra Zeneca und 46,2 % für Johnson & Johnson. Bei Auftreten unerwünschter Ereignisse dauerten 93,3 % der Symptome ≤ 1 Woche. Anfallshäufungen oder Anfallsrezidive nach Anfallsfreiheit betrafen 1,4 % und waren somit statistisch signifikant seltener als nach Infektionen (p = 0,0016).
Schlussfolgerung
Der Verlauf der COVID-19-Infektionen und -Impfungen war ähnlich wie in der Allgemeinbevölkerung. Zu Anfallszunahmen, die insgesamt selten waren, kam es häufiger im Zusammenhang mit Infektionen.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since the outbreak of the SARS-CoV‑2 virus (COVID-19) pandemic at the end of 2019 in Wuhan, China, life changed considerably all around the world.
Concerning seizures and epilepsy, the questions arose of whether COVID-19 infections were a potential trigger of new-onset seizures and epilepsies and whether they influenced the natural course of previously diagnosed and pharmacologically treated epilepsies.
Four vaccines were approved by the European Medicine Agency (EMA) by the end of 2021: the mRNA vaccines Comirnaty® from Pfizer/BionTech and mRNA-1273® from Moderna and the viral vector vaccines Vaxzevria® from Astra Zeneca as well as the vaccine Ad26.COV2-S® from Johnson & Johnson.
From the beginning of the pandemic throughout the roll-out of the vaccination program, adult patients with epilepsy (PWE) and their relatives were concerned about a potentially elevated risk of infection but especially about the adverse events (AEs) of the vaccinations. Early on, the majority of physicians recommended the vaccinations especially after approval by the World Health Organization (WHO) and the US Food and Drug Administration (FDA). However, there were concerns about the use of these vaccines in all PWE irrespective of special populations [2].
Therefore, we decided to collect data on COVID-19 infections and vaccinations among the PWE who had an outpatient appointment with the first author of this article between July 2020 and July 2021 and followed them up thereafter until the end of 2021 in order to address the following issues:
-
1.
The course of confirmed COVID-19 infections in PWE without vaccinations.
-
2.
The impact of vaccinations on tolerability and seizure situation in PWE who had been fully vaccinated with one of the four vaccines that had been approved at that time and had not been aware of an infection.
Methods
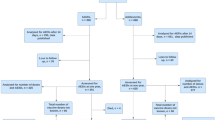
At cut-off (31 December 2021) we included only PWE who either had been infected with COVID-19 confirmed by polymerase chain reaction (PCR) testing and without vaccinations (group 1) or had been fully vaccinated with the last vaccination at least 14 days prior to the interview and without a previous infection (group 2). All PWE were registered as fully vaccinated (a) if they had undergone a double vaccination with the vaccines Comirnaty™ from Pfizer/BionTech, mRNA-127® from Moderna, or Vaxzevria® from Astra Zeneca with one repetition or a combination of these three or (b) if the COVID-19 vaccine Ad26.COV.2® from Johnson & Johnson had been applied once. All other PWE from the original dataset were excluded (see Fig. 1).
Data were collected on the etiology and classification of the epilepsy syndromes, on demographics, and on the current seizure situation. The PWE were classified as seizure-free if no seizures had occurred for ≥ 1 year.
We assessed whether infected PWE showed any signs or symptoms (“symptomatic”). In symptomatic patients, we recorded the symptoms, their duration, and their severity. Symptoms were classified as severe if a hospitalization had been necessary. In vaccinated PWE, the vaccine, the order of vaccinations, AEs, and their intensity and duration were recorded. Single arm pain was not registered as an AE.
Seizure increase was scored both for infected and vaccinated PWE when seizures relapsed within 1 week of the confirmed infection or vaccination in formerly seizure-free patients for ≥ 1 year or the seizure frequency at least doubled during this period in patients with ongoing seizures.
Descriptive statistics were used to summarize the characteristics of the data. For statistical comparison, we applied a chi-square test with a p level of 0.05. A chi-square test of the adjusted residuals with Bonferroni correction was used for post hoc analysis. The study was approved by the local ethics committee of the University of Freiburg, Germany (EK21-1644), and registered in the clinical trial registry of Germany (DRKS00028046).
Results
Between July 2020 and July 2021, we consecutively recruited 1152 PWE (48% male; mean age: 42.7 years, range: 17–89 years).
At data cut-off, the total group of PWE included for the final analysis comprised 612 PWE. Figure 1 shows the flowchart of the selection procedure.
Of the PWE, 51 had been infected without vaccination and 561 had undergone full vaccination without awareness of an infection.
The demographic data are shown in Table 1. Notably, there were slight differences between the two groups: In the group of infected PWE, the rate of intellectually disabled and PWE with ongoing seizures was higher (29.4% vs. 22.8% and 54.9% vs. 43.0%, respectively). These differences were not statistically significant (intellectually disabled: p = 0.287; ongoing seizures: p = 0.100).
Infected PWE, not vaccinated
Among the 51 infected PWE, 39 (76.5%) presented with signs or symptoms. Five (9.8%) had a severe course with one death.
The most frequent symptoms comprised influenza-like symptoms (48.7% of infected PWE with symptoms), anosmia (28.2%), and ageusia (20.5%). A total of 14 different symptoms were reported. The mean duration of symptoms was 20 days (median: 1 week; range: 2 days–7 months). Overall, 51.3% reported symptoms with a duration of ≤ 1 week (see Electronic supplementary material for details).
Unequivocal seizure increases or seizure relapses after sustained seizure freedom after the infection occurred in four cases (7.8%). Detailed findings are displayed in Table 3.
On the day of cut-off (31 December 2021) the German population comprised 83.1 million people (www.destatis.de, Bevölkerungsstand: Amtliche Einwohnerzahl Deutschlands 2021—Statistisches Bundesamt [destatis.de], retrieved on 31.12.2021). A COVID-19 infection was recorded in 7.13 million people, 112,000 of whom (1.6%) died in association with the infection (Johns Hopkins University Center for Systems Science and Engineering [JHU CSSE]: https://systems.jhu.edu/, retrieved on 31.12.2021), which reflects a cumulative incidence of 8.6%, with a 1.6% death rate. This death rate of infected PWE was similar to the one observed in our PWE (1/51 infected patients = 2%).
Fully vaccinated patients
The total number of vaccinations was 1109 (Pfizer/BionTech n = 881, Moderna n = 100, Astra Zeneca n = 115, Johnson & Johnson n = 13). Overall, 73.3% (n = 411) PWE had two consecutive vaccinations with the Pfizer/BionTech vaccine, 8.6% (n = 48) with Moderna, 4.6% (n = 26) with Astra Zeneca, and 2.3% (n = 13) with the single vaccination of Johnson & Johnson. If cross-vaccinations were performed, the vaccine of Astra Zeneca was followed by that of Pfizer/BionTech in 10.5% (n = 59) and by the vaccine of Moderna in 0.7% (n = 4).
A total of 113 PWE reported AEs (20.1% of all vaccinated PWE). Among the 548 PWE with two vaccinations (without 13 PWE who received the Johnson & Johnson vaccine), 22 reported AEs after each vaccination independent of the vaccine.
Of these, 12 had AEs after both repetitive vaccinations with the vaccine of Pfizer/BionTech, three after both vaccinations with the vaccine of Moderna and seven both after a first vaccination with the product of Astra Zeneca and the follow-up vaccination with the vaccine of Pfizer/BionTech.
Table 2 shows the AEs in detail. More than one AE was possible in single cases.
The rates of AEs per vaccine were 14.0% for Pfizer/BionTech, 32.7% for Moderna, 25.8% for Astra Zeneca, and 46.2% for Johnson & Johnson. A post hoc chi-square test of the adjusted residuals with Bonferroni correction revealed significantly fewer AEs with the vaccine of Pfizer/BionTech compared to that of Moderna (p < 0.05).
A total of 93.3% of the reported AEs lasted ≤ 1 week, most of them for 1–2 days. Three PWE reported symptoms with a duration of ≥ 1 months (fatigue, n = 2, and sustained pain in the vaccinated arm). Unequivocal seizure increase or seizure relapse after sustained seizure freedom occurred in eight PWE (1.4%), six of these after vaccination with the vaccine of Pfizer/BionTech and two after the vaccine of Moderna.
Out of 411 PWE with two vaccinations with Pfizer/BionTech, 20 (4.9%) had AEs after the first and 58 (14.1%) after the second application. The corresponding figures were three (6.3%) and 15 (31.3%) for Moderna and three (12.7%) and one (4.3%) for Astra Zeneca. Thus, a majority of PWE had an AE after the second vaccination with one of the two mRNA vaccines whereas more patients presented with AEs after the first vaccination with the vaccine of Astra Zeneca.
Seizures increased in 7.8% (n = 4) of the infected and 1.4% (n = 8) of the vaccinated PWE. The comparison between infected and vaccinated PWE revealed a statistically significant higher seizure risk in the infected group (p = 0.0016).
The clinical data of these PWE are shown in Table 3.
Discussion
When we started this study, many PWE were concerned that vaccinations might have been as threatening as infections. Therefore, the primary objective was to investigate the course of infected PWE without vaccinations versus that of vaccinated PWE without apparent infection and we excluded any other PWE in order to rule out any methodological interference.
Infected PWE
Out of 51 PCR-positive PWE, 39 (76.5%) reported signs or symptoms and thus suffered from the COVID-19 disease. In 34 of these 39 infected PWE (87.1%) with signs or symptoms, the course was mild or moderate and short lasting. Along with influenza-like symptoms, anosmia and ageusia were the most common findings. This is in agreement with other reports from the literature [3].
The overall risk of seizure increase in our study was 7.8% and corresponded well with a rate of 8.6% during the acute COVID-19 illness phase according to Sanchez-Larsen et al. [19].
Intellectual disability and institutionalization were hypothesized by other authors to be risk factors for a severe COVID-19 course in PWE [19]. Although they seemed to also be somewhat overrepresented in the group of PWE with seizure increases in our study, neither intellectual disabilities nor difficult-to-treat epilepsies were statistically significant risk confounders for severe courses. Overall, 9.8% had a severe course. The fatality rate was 2% and these figures were well in the range of an Iranian [1] and a Spanish study [19]. Others reported a higher risk of a poor COVID-19 outcome in PWE [20], more deaths related to COVID-19 in epilepsy patients, and more deaths associated with epilepsy in 2020 than in previous years [5]. Case fatality rates were not higher in PWE compared to hospital admissions due to other diseases according to Cabezuedo-Garcia et al. but they were still remarkably high (23%; [4]).
Our selected cohort of out-patients does not enable any conclusions to be drawn about the incidence of COVID-19 infections in PWE compared to the general population. Others reported such an elevated incidence [3] although the data are controversial: In a study from Spain, the COVID-19 prevalence in PWE was 5.53% and thus just slightly higher than in the Spanish population (4.26%; [19]) whereas PWE were not at a higher risk of contracting COVID-19 according to Asadi-Pooya et al. [1].
Several other aspects of the impact of the pandemic on PWE have been addressed by other researchers and were not within the scope of our study: Seizure increases during the COVID-19 pandemic have been reported independently of infections [7, 22]. A seizure increase during the pandemic was partly explained by several problems beyond the infection itself such as dispensing of antiseizure medication (ASM), telehealth, and access to the healthcare system in general, at least in developing countries [9]. Finally, results concerning the pandemic and new-onset seizures and epilepsies were inconsistent [1, 6, 8, 12,13,14, 17].
Fully vaccinated PWE
Vaccinations were generally safe and well tolerated irrespective of the vaccine applied. Only one fifth of PWE reported AEs. Since we interviewed our patients after the vaccinations, did not consider pain in the vaccinated arm as an AE, and did not collect AEs prospectively, the AE rate might have been higher with a different study design. Although a post hoc analysis revealed statistically significantly fewer AEs after the Pfizer/BionTech vaccination than after that of Moderna, the comparison between vaccines should be interpreted with caution because of the unequal distribution of the vaccines. In the case of AEs, there was a tendency toward AEs after the second vaccination with mRNA vaccines and after the first vaccination with vector vaccines, which was also confirmed by others [16, 18, 21]. The main AEs did not differ between the vaccines and comprised fatigue, fever, and headache.
Interestingly, seizure aggravations occurred statistically significantly more often in infected than in vaccinated PWE, and therefore vaccine-related seizure aggravation was certainly not a risk factor that suggested specific caution.
Our experiences confirm the reports of other groups in smaller cohorts of PWE:
Von Wrede et al. [21] reported on a group of 54 PWE who got their first COVID-19 vaccination with vaccines from Pfizer/BionTech, Moderna, and Astra Zeneca. Two-thirds tolerated the vaccines generally either very well or well. Overall, 33% reported general vaccination-related AEs such as headache (17%), fatigue (15%), and fever/shivering (9%), which is very similar to our findings. One patient reported an increased seizure frequency 1 day after the first COVID-19 vaccination and one reported the occurrence of a new seizure type.
Massoud et al. [16] reported at least one AE in 80.5% of 82 vaccinated PWE who had been vaccinated either with the Pfizer BioNTech mRNA vaccine or that of AstraZeneca but they also included local pain at the injection site as an AE. Apart from this methodological difference to our study, PWE who received the Pfizer BioNTech mRNA vaccine (first, second dosage) and the vaccine of AstraZeneca (first dose) reported AEs similar to our findings: fatigue (47%, 32.4%, 46.9%, respectively), headache (33.3%, 35.3%, 34.4%, respectively), and myalgia (40%, 35%, 50%, respectively). In total, 95% were symptom-free by the end of the first week after the vaccination. Symptoms were mostly mild or moderate and seizure increases were uncommon.
Özdemir et al. [18] recently reported on 178 PWE mostly with generalized epilepsies, the majority of whom had been fully vaccinated with the Pfizer/BioNTech mRNA vaccine. No seizure increase was observed. The risk of AEs was not associated with epilepsy type, age at onset of epilepsy, the number of ASMs, or with any other clinical feature of epilepsy. All patients with seizure increases (n = 4) presented with fever, which was not the case in our group.
A Chinese study of 491 epilepsy patients, 217 people with other neuropsychiatric diseases, and 273 healthy controls addressed the tolerability of mostly inactivated vaccines. No significant differences were apparent among the three groups. Two epilepsy patients had their first convulsive seizure, two healthy controls had a first epileptic seizure that, however, was not unequivocally explained by the vaccination itself. Overall, 19 PWE (3.9%) reported a seizure increase [15].
Limitations
Our study has several limitations. First, for the main objective of the study, we addressed exclusively infected PWE without vaccinations and vaccinated PWE without infections. Thus, we cannot add any information regarding patients who underwent both infection and vaccination, which has certainly occurred in many instances especially after the appearance of the so-called omicron variant.
Secondly, the results were collected prior to booster vaccinations that had been proposed thereafter and prior to the breakout of the so-called omicron variant of the COVID-19 virus.
Thirdly, at cut-off no vaccinated PWE had experienced a COVID-19 infection. Epilepsy had been listed among other confounders as a possible risk factor for a fatal outcome of the infection in spite of a preceding vaccination [10]. Due to the study design these PWE were excluded.
Furthermore, the younger the patients were, the higher the prevalence of COVID-19-related deaths of PWE [11]. Since we included only adults, our results might not be representative for the whole community of PWE including children and adolescents.
Finally, no structured interview with more standardized questions was used. The interviews were part of the regular out-patient appointments.
Conclusion
Severe or even fatal courses of the infection affected less than 10% of the infected PWE. The vaccines approved in Germany in 2021 were generally well tolerated. Seizure aggravations were exceptions and occurred significantly less often than in COVID-19-infected PWE. More data are needed that should include vaccinated PWE who nevertheless got infected and particularly after the outbreak of the omicron variant.
References
Asadi-Pooya AA, Emami A, Akbari A, Javanmardi F (2021) COVID-19 presentations and outcome in patients with epilepsy. Acta Neurol Scand 143(6):624–628. https://doi.org/10.1111/ane.13404
Asadi‐Pooya AA, Sahraian A, Shervin Badv R, Sahraian MA (2021) Physician’s opinions on the necessity of COVID-19 vaccination in patients with epilepsy. Epileptic Disord 23(3):485–489. https://doi.org/10.1684/epd.2021.1282
Bosak M, Mazurkiewicz I, Wezyk K, Slowik A, Turaj W (2021) COVID-19 among patients with epilepsy: risk factors and course of the disease. Epilepsy Behav 120:107996. https://doi.org/10.1016/j.yebeh.2021.107996
Cabezudo-Garcia P, Ciano-Petersen NL, Mena-Vázquez N, Pons-Pons G, Castro-Sánchez MV, Serrano-Castro P (2020) Incidence and fatality rate of COVID-19 in patients with active epilepsy. Neurology 95(10):e1417–e1425. https://doi.org/10.1212/WNL.0000000000010033
Daniels H, Lacey AS, Mikadze D, Akbari A, Fonferko-Shadrach B, Hollinghurts J, Lyons RA, Rees MI, Sawhney IN, Powell RH, Kerr MP, Pickrell WO (2022) Epilepsy mortality in Wales during COVID-19. Seizure 94:39–42. https://doi.org/10.1016/j.seizure.2021.11017
Emami A, Fadakar N, Akbari A, Lotfi M, Farazdaghi M, Javanmardi F, Rezai T, Asadi-Pooya AA (2020) Seizure in patients with COVID-19. Neurol Sci 41(11):3057–3061. https://doi.org/10.1007/s10072-020-04731-9
Fonseca E, Quintana M, Lallana S, Restrepo JL, Abraira L, Santamarina E, Seijo-Raposo I, Toledo M (2020) Epilepsy in time of COVID-19: A survey-based study. Acta Neurol Scand 142(6):545–554. https://doi.org/10.1111/ane.13335
García-Azorin D, Abenza Abildúa MJ, Erro Aguirre ME, Fernández Fernandéz S, García Moncó JC, Guijarro-Castro C, González Platas M, Romero Delgado F, Andrés Laínez JM, Ezpeleta D, Spanish neuroCOVID registry group (2021) Neurological presentations of COVID-19: findings from the Spanish Society of Neurology neuroCOVID-19 registry. J Neurol Sci 423:117283. https://doi.org/10.1016/j.jns.2020.117283
Guilhoto LM, Mosini AC, Susemihl MA, Pinto LF (2021) COVID-19 and epilepsy: how are people with epilepsy in Brazil? Epilepsy Behav 122:108115. https://doi.org/10.1016/j.yebeh.2021.108115
Hippisley CJ, Coupland AC, Mehta N, Keogh RH, Diaz-Ordaz K, Khunti K, Lyons RA, Kee F, Sheikh A, Rahman S, Valabhji J, Harrison EM, Sellen P, Haq N, Semple MG, Johnson PWM, Hayward A, Nguyen-Van-Tam JS (2021) Risk prediction of covid-19 related death and hospital admission in adults after covid-19 vaccination: national prospective cohort study. BMJ 374:n2244. https://doi.org/10.1136/bmj.n2244
Horváth RA, Sütö Z, Cséke B, Schranz D, Darnai G, Kovács N, Janszky I, Janszky J (2022) Epilepsy is overrepresented among young people who died from COVID-19: analysis of nationwide mortality data from Hungary. Seizure 94:136–141. https://doi.org/10.1016/j.seizure.2021.11.013
Keshavarzi A, Janbabaei G, Kheyrati L, Hoseini Ghavamabad L, Asadi-Pooya AA (2021) Seizure is a rare presenting manifestation of COVID.-19. Seizure 86:16–18. https://doi.org/10.1016/j.seizure.2021.01.009
Khedr EM, Shoyb A, Mohammaden M, Saber M (2021) Acute symptomatic seizures and COVID-19: hospital-based study. Epilepsy Res 174:106650. https://doi.org/10.1016/j.eplepsyres.2021.106650
Kurd M, Hashavya S, Benenson S, Gilboa T (2021) Seizures as the main presenting manifestation of acute SARS-CoV‑2 infection in children. Seizure 92:89–93. https://doi.org/10.1016/j.seizure.2021.08.017
Lu L, Zhang Q, Xiao J, Zhang Y, Peng W, Han X, Chen S, Yang D, Sander JW, Zhou D, Xiong W (2022) COVID-19 vaccine take-up rate and safety in adults with epilepsy: data from a multicenter study in China. Epilepsia 63(1):244–251. https://doi.org/10.1111/epi.17138
Massoud F, Ahmad SF, Hassan AM, Alexander KJ, Al Hashel J, Arabi M (2021) Safety and tolerability of the novel 2019 coronavirus disease (COVID-19) vaccines among people with epilepsy (PwE): a cross-sectional study. Seizure 92:2–9. https://doi.org/10.1016/j.seizure.2021.08.001
Neshige S, Aoki A, Takebayashi Y, Shishido T, Yamazaki Y, Iida K, Maruyama H (2021) A longitudinal seizure outcome following the COVID-19 pandemic in 220 and 2021: Transient exacerbation or sustainable mitigation. J Neurol Sci 434:120100. https://doi.org/10.1016/j.jns.2021.120100
Özdemir HN, Dere B, Gökçay F, Gökçay A (2022) Are COVID-19 vaccines safe for people with epilepsy? A cross-sectional study. Neurol Sci. https://doi.org/10.1007/s10072-022-05956-6
Sanchez-Larsen A, Conde-Blanco E, Viloria-Alabesque A, Sanchez-Vizcaino Burndia C, Espinosa Oltra T, Alvaraze-Noval A, Aledo-Serrano A, Martin-Garcia R, Ramos-Araque ME, Campos D, Valle-Penacoba G, Sierra-Gómez A, De Ceballos-Cerrajeria P, Agundez-Sarasola M, Gómez-Ibanez A, Villino-Boquete R, Cabezudo-Garcia P, Rodiguez-Lavado I, Principe A, Sopelana-Garay D (2021) COVID-19 prevalence and mortality in people with epilepsy: a nation-wide multicenter study. Epilepsy Behav 125:108379. https://doi.org/10.1016/j.yebeh.2021.108379
Siahaan YMT, Ketaren RJ, Hartoyo V, Hariyanto TI (2021) Epilepsy and the risk of severe coronavirus disease 2019 outcomes: a systematic review, meta-analysis and meta-regression. Epilepsy Behav 125:108437. https://doi.org/10.1016/j.yebeh.2021.108437
Von Wrede R, Pukropski J, Moskau-Hartmann S, Surges R, Baumgartner T (2021) COVID-19 vaccination in patients with epilepsy: first experiences in a German tertiary epilepsy center. Epilepsy Behav 122:108160. https://doi.org/10.1016/j.yebeh.2021.108160
Zeng C, Meng H, Zhu Y, Yao L, Lian Y, Zhu Y, Zhang M, Dai Y, Wang K, Han X, Li L, Zhang L, Xu H, Yao D, Luo X, Jiang W, Wang X, Zhao C, Chen Y, Deng X, Liu C, Feng L, Song Y, Wu Y, Liao W, Wang F, Zhu S, Xiao B, Wang Q, Long L (2021) Correlation of seizure increase and COVID-19 outbreak in adult patients with epilepsy: findings and suggestions from a Nationwide multi-centre survey in China. Seizure 88:102–108. https://doi.org/10.1016/j.seizure.2021.03.029
Funding
No funding was received by any external source.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
B.J. Steinhoff and T. Intravooth declare that they have no competing interests.
For this article no studies with human participants or animals were performed by any of the authors. All studies mentioned were in accordance with the ethical standards indicated in each case.
Additional information

Supplementary material online – scan QR code
Supplementary Information
Rights and permissions
About this article
Cite this article
Steinhoff, B.J., Intravooth, T. Experiences of adult epilepsy patients with COVID-19 infections and vaccinations. Clin Epileptol 36, 45–51 (2023). https://doi.org/10.1007/s10309-023-00567-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10309-023-00567-3