Abstract
Purpose
Previous microneurographic studies found that muscle sympathetic nerve activity (MSNA) increased in normotensive pregnant women and was even greater in women with gestational hypertension and preeclampsia during the third trimester. It is possible that sympathetic activation during the latter months of normal pregnancy helps return arterial pressure to non-pregnant levels. However, when the increase in sympathetic activity is excessive, hypertension ensues. The key question that must be addressed is whether sympathetic activation develops early during pregnancy and remains high throughout gestation, or whether this sympathetic overactivity only occurs at term, providing the substrate for preeclampsia and other pregnancy-associated cardiovascular complications.
Methods
This was a literature review of autonomic neural control during pregnancy.
Results
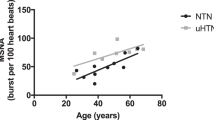
Recent work from our laboratory and other laboratories showed that in healthy women resting MSNA increased in early pregnancy, increased further in late pregnancy, and returned to the pre-pregnancy levels shortly after delivery. We found that women who exhibited excessive sympathetic activation during the first trimester, before any clinical signs and symptoms appeared, developed gestational hypertension at term. We also found that the level of corin, an atrial natriuretic peptide-converting enzyme, was increased in the maternal circulation, especially during late pregnancy, as a homeostatic response to elevated sympathetic activity.
Conclusion
These findings provide important insight into the neural mechanisms underlying hypertensive disorders during pregnancy. With this knowledge, early prevention or treatment targeted to the appropriate pathophysiology may be initiated, which may reduce maternal and fetal death or morbidity, as well as cardiovascular risks in women later in life.





Similar content being viewed by others
References
Chapman AB, Abraham WT, Zamudio S, et al (1998) Temporal relationships between hormonal and hemodynamic changes in early human pregnancy. Kidney Int 54(6):2056–2063
Fu Q, Levine BD (2009) Autonomic circulatory control during pregnancy in humans. Semin Reprod Med 27(4):330–337
Sala C, Campise M, Ambroso G, Motta T, Zanchetti A, Morganti A (1995) Atrial natriuretic peptide and hemodynamic changes during normal human pregnancy. Hypertension 25(4 Pt 1):631–636
Meah VL, Cockcroft JR, Backx K, Shave R, Stohr EJ (2016) Cardiac output and related haemodynamics during pregnancy: a series of meta-analyses. Heart 102(7):518–526
Hunter S, Robson SC (1992) Adaptation of the maternal heart in pregnancy. Br Heart J 68(6):540–543
Greenwood JP, Scott EM, Walker JJ, Stoker JB, Mary DA (2003) The magnitude of sympathetic hyperactivity in pregnancy-induced hypertension and preeclampsia. Am J Hypertens 16(3):194–199
Schobel HP, Fischer T, Heuszer K, Geiger H, Schmieder RE (1996) Preeclampsia: a state of sympathetic overactivity. N Engl J Med 335(20):1480–1485
Fischer T, Schobel HP, Frank H, Andreae M, Schneider KT, Heusser K (2004) Pregnancy-induced sympathetic overactivity: a precursor of preeclampsia. Eur J Clin Invest 34(6):443–448
Greenwood JP, Scott EM, Stoker JB, Walker JJ, Mary DA (2001) Sympathetic neural mechanisms in normal and hypertensive pregnancy in humans. Circulation 104(18):2200–2204
Greenwood JP, Stoker JB, Walker JJ, Mary DA (1998) Sympathetic nerve discharge in normal pregnancy and pregnancy-induced hypertension. J Hypertens 16(5):617–624
Jarvis SS, Shibata S, Bivens TB, et al (2012) Sympathetic activation during early pregnancy in humans. J Physiol 590(15):3535–3543
Okada Y, Best SA, Jarvis SS, et al (2015) Asian women have attenuated sympathetic activation but enhanced renal-adrenal responses during pregnancy compared to Caucasian women. J Physiol 593(5):1159–1168
Hissen SL, El Sayed K, Macefield VG, Brown R, Taylor CE (2017) Muscle sympathetic nerve activity peaks in the first trimester in healthy pregnancy: a longitudinal case study. Clin Auton Res 27(6):401–406
Reyes LM, Usselman CW, Skow RJ, et al (2018) Sympathetic neurovascular regulation during pregnancy: a longitudinal case series study. Exp Physiol 103(3):318–323
Usselman CW, Skow RJ, Matenchuk BA, et al (2015) Sympathetic baroreflex gain in normotensive pregnant women. J Appl Physiol (1985) 119(5):468–474
Usselman CW, Wakefield PK, Skow RJ, et al (2015) Regulation of sympathetic nerve activity during the cold pressor test in normotensive pregnant and nonpregnant women. Hypertension 66(4):858–864
Charkoudian N, Usselman CW, Skow RJ, et al (2017) Muscle sympathetic nerve activity and volume-regulating factors in healthy pregnant and nonpregnant women. Am J Physiol Heart Circ Physiol 313(4):H782–h787
Schmidt SML, Usselman CW, Martinek E, et al (2018) Activity of muscle sympathetic neurons during normotensive pregnancy. Am J Physiol Regul Integr Comp Physiol 314(2):R153–R160
Santorelli G, Lawlor DA, West J, Tuffnell D, Farrar D (2019) Population reference and healthy standard blood pressure range charts in pregnancy: findings from the Born in Bradford cohort study. Sci Rep 9(1):18847
Ma Y, Zhang X, Che Q, et al (2020) Reference ranges and trajectories for blood pressure in pregnancy: findings from a follow-up study based on China Maternal and Newborn’s Health Monitoring System. Hypertens Pregnancy 39(2):117–125
Fu Q, Shook RP, Okazaki K, et al (2006) Vasomotor sympathetic neural control is maintained during sustained upright posture in humans. J Physiol 577(Pt 2):679–687
Steinback CD, Fraser GM, Usselman CW, et al (2019) Blunted sympathetic neurovascular transduction during normotensive pregnancy. J Physiol 597(14):3687–3696
Williams DJ, Vallance PJ, Neild GH, Spencer JA, Imms FJ (1997) Nitric oxide-mediated vasodilation in human pregnancy. Am J Physiol 272(2 Pt 2):H748–752
Khalil RA, Crews JK, Novak J, Kassab S, Granger JP (1998) Enhanced vascular reactivity during inhibition of nitric oxide synthesis in pregnant rats. Hypertension 31(5):1065–1069
Stennett AK, Qiao X, Falone AE, Koledova VV, Khalil RA (2009) Increased vascular angiotensin type 2 receptor expression and NOS-mediated mechanisms of vascular relaxation in pregnant rats. Am J Physiol Heart Circ Physiol 296(3):H745–755
Aune B, Vartun A, Oian P, Sager G (2000) Evidence of dysfunctional beta2-adrenoceptor signal system in pre-eclampsia. BJOG 107(1):116–121
Landau R, Dishy V, Wood AJ, Stein CM, Smiley RM (2002) Disproportionate decrease in alpha- compared with beta-adrenergic sensitivity in the dorsal hand vein in pregnancy favors vasodilation. Circulation 106(9):1116–1120
Creanga AA, Syverson C, Seed K, Callaghan WM (2017) Pregnancy-related mortality in the United States, 2011–2013. Obstet Gynecol 130(2):366–373
Cabacungan ET, Ngui EM, McGinley EL (2012) Racial/ethnic disparities in maternal morbidities: a statewide study of labor and delivery hospitalizations in Wisconsin. Matern Child Health J 16(7):1455–1467
Caughey AB, Stotland NE, Washington AE, Escobar GJ (2005) Maternal ethnicity, paternal ethnicity, and parental ethnic discordance: predictors of preeclampsia. Obstet Gynecol 106(1):156–161
Abate NI, Mansour YH, Tuncel M, et al (2001) Overweight and sympathetic overactivity in black Americans. Hypertension 38(3):379–383
Eikelis N, Lambert EA, Phillips S, et al (2017) Muscle sympathetic nerve activity is associated with elements of the plasma lipidomic profile in young Asian adults. J Clin Endocrinol Metab 102(6):2059–2068
Ray CA, Monahan KD (2002) Sympathetic vascular transduction is augmented in young normotensive blacks. J Appl Physiol (1985) 92(2):651–656
Calhoun DA, Mutinga ML, Collins AS, Wyss JM, Oparil S (1993) Normotensive blacks have heightened sympathetic response to cold pressor test. Hypertension 22(6):801–805
Jarvis SS, Shibata S, Okada Y, Levine BD, Fu Q (2014) Neural-humoral responses during head-up tilt in healthy young white and black women. Front Physiol 5:86
Peters SA, van der Schouw YT, Wood AM, et al (2016) Parity, breastfeeding and risk of coronary heart disease: a pan-European case-cohort study. Eur J Prev Cardiol 23(16):1755–1765
Ogunmoroti O, Osibogun O, Kolade OB, et al (2019) Multiparity is associated with poorer cardiovascular health among women from the multi-ethnic study of atherosclerosis. Am J Obstet Gynecol 221(6):631.e631–631.e616
Shen L, Wu J, Xu G, et al (2015) Parity and risk of coronary heart disease in middle-aged and older Chinese women. Sci Rep 5:16834
Rurangirwa AA, Gaillard R, Steegers EA, Hofman A, Jaddoe VW (2012) Hemodynamic adaptations in different trimesters among nulliparous and multiparous pregnant women; the Generation R study. Am J Hypertens 25(8):892–899
Peters SAE, Yang L, Guo Y, et al (2017) Pregnancy, pregnancy loss, and the risk of cardiovascular disease in Chinese women: findings from the China Kadoorie Biobank. BMC Med 15(1):148
Badrov MB, Yoo JK, Steinback CD, Davenport MH, Fu Q (2020) Influence of multiparity on sympathetic nerve activity during normal pregnancy. Am J Physiol Heart Circ Physiol 318(4):H816–h819
Clapp JF 3rd, Capeless E (1997) Cardiovascular function before, during, and after the first and subsequent pregnancies. Am J Cardiol 80(11):1469–1473
Tooher J, Thornton C, Makris A, Ogle R, Korda A, Hennessy A (2017) All hypertensive disorders of pregnancy increase the risk of future cardiovascular disease. Hypertension 70(4):798–803
American College of Obstetricians and Gynecologists; Task Force on Hypertension in Pregnancy (2013) Report of the American College of Obstetricians and Gynecologists’ task force on hypertension in pregnancy. Obstet Gynecol 122(5):1122–1131
Hnat MD, Sibai BM, Caritis S, et al (2002) Perinatal outcome in women with recurrent preeclampsia compared with women who develop preeclampsia as nulliparas. Am J Obstet Gynecol 186(3):422–426
Fischer T, Schneider MP, Schobel HP, Heusser K, Langenfeld M, Schmieder RE (2000) Vascular reactivity in patients with preeclampsia and HELLP (hemolysis, elevated liver enzymes, and low platelet count) syndrome. Am J Obstet Gynecol 183(6):1489–1494
Hieda M, Yoo JK, Sun DD, et al (2018) Time course of changes in maternal left ventricular function during subsequent pregnancy in women with a history of gestational hypertensive disorders. Am J Physiol Regul Integr Comp Physiol 315(4):R587–R594
Badrov MB, Park SY, Yoo JK, et al (2019) Role of corin in blood pressure regulation in normotensive and hypertensive pregnancy. Hypertension 73(2):432–439
Volpe M (2014) Natriuretic peptides and cardio-renal disease. Int J Cardiol 176(3):630–639
Floras JS (1990) Sympathoinhibitory effects of atrial natriuretic factor in normal humans. Circulation 81(6):1860–1873
Macheret F, Heublein D, Costello-Boerrigter LC, et al (2012) Human hypertension is characterized by a lack of activation of the antihypertensive cardiac hormones ANP and BNP. J Am Coll Cardiol 60(16):1558–1565
Zhou Y, Wu Q (2013) Role of corin and atrial natriuretic peptide in preeclampsia. Placenta 34(2):89–94
Yan W, Sheng N, Seto M, Morser J, Wu Q (1999) Corin, a mosaic transmembrane serine protease encoded by a novel cDNA from human heart. J Biol Chem 274(21):14926–14935
Cui Y, Wang W, Dong N, et al (2012) Role of corin in trophoblast invasion and uterine spiral artery remodelling in pregnancy. Nature 484(7393):246–250
Gu Y, Thompson D, Xu J, et al (2018) Aberrant pro-atrial natriuretic peptide/corin/natriuretic peptide receptor signaling is present in maternal vascular endothelium in preeclampsia. Pregnancy Hypertens 11:1–6
Khalil A, Maiz N, Garcia-Mandujano R, Elkhouli M, Nicolaides KH (2015) Longitudinal changes in maternal corin and mid-regional proatrial natriuretic peptide in women at risk of pre-eclampsia. Ultrasound Obstet Gynecol 45(2):190–198
Liu Y, Hu J, Yu Q, Zhang P, Han X, Peng H (2015) Increased serum soluble corin in mid pregnancy is associated with hypertensive disorders of pregnancy. J Womens Health (Larchmt) 24(7):572–577
Miyazaki J, Nishizawa H, Kambayashi A, et al (2016) Increased levels of soluble corin in pre-eclampsia and fetal growth restriction. Placenta 48:20–25
Zaki MA, El-Banawy Sel D, El-Gammal HH (2012) Plasma soluble corin and N-terminal pro-atrial natriuretic peptide levels in pregnancy induced hypertension. Pregnancy Hypertens 2(1):48–52
Acknowledgements
This brief review article was supported, in part, by the National Institutes of Health grants K23 (HL075283), R21 (HL088184), and R01 (HL142605); the American Heart Association Grant-in-Aid grant award (13GRNT16990064); and the Harry S. Moss Heart Trust.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical declaration
This article is based on previously conducted studies, and does not involve any new studies of human or animal subjects performed by either of the authors.
Conflict of interests
The authors declare that they have no conflict of interests regarding this article.
Rights and permissions
About this article
Cite this article
Hissen, S.L., Fu, Q. Neural control of blood pressure during pregnancy in humans. Clin Auton Res 30, 423–431 (2020). https://doi.org/10.1007/s10286-020-00703-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10286-020-00703-3