Abstract
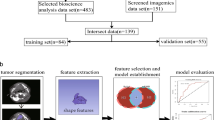
Forkhead box P3 (FOXP3) has been identified as a novel molecular marker in various types of cancer. The present study assessed the expression of FOXP3 in patients with head and neck squamous cell carcinoma (HNSCC) and its potential as a clinical prognostic indicator, and developed a radiomics model based on enhanced computed tomography (CT) imaging. Data from 483 patients with HNSCC were downloaded from the Cancer Genome Atlas for FOXP3 prognostic analysis and enhanced CT images from 139 patients included in the Cancer Imaging Archives, which were subjected to the maximum relevance and minimum redundancy and recursive feature elimination algorithms for radiomics feature extraction and processing. Logistic regression was used to build a model for predicting FOXP3 expression. A prognostic scoring system for radiomics score (RS), FOXP3, and patient clinicopathological factors was established to predict patient survival. The area under the receiver operating characteristic (ROC) curve (AUC) and calibration curve and decision curve analysis (DCA) were used to evaluate model performance. Furthermore, the relationship between FOXP3 and the immune microenvironment, as well as the association between RS and immune checkpoint-related genes, was analyzed. Results of analysis revealed that patients with HNSCC and high FOXP3 mRNA expression exhibited better overall survival. Immune infiltration analysis revealed that FOXP3 had a positive correlation with CD4 + and CD8 + T cells and other immune cells. The 8 best radiomics features were selected to construct the radiomics model. In the FOXP3 expression prediction model, the AUC values were 0.707 and 0.702 for the training and validation sets, respectively. Additionally, the calibration curve and DCA demonstrated the positive diagnostic utility of the model. RS was correlated with immune checkpoint-related genes such as ICOS, CTLA4, and PDCD1. A predictive nomogram was established, the AUCs were 0.87, 0.787, and 0.801 at 12, 24, and 36 months, respectively, and DCA demonstrated the high clinical applicability of the nomogram. The enhanced CT radiomics model can predict expression of FOXP3 and prognosis in patients with HNSCC. As such, FOXP3 may be used as a novel prognostic marker to improve individualized clinical diagnosis and treatment decisions.






Similar content being viewed by others
Data Availability
This data can be found in publicly available datasets: the patients’ data were obtained from TCGA (https://portal.gdc.cancer.Gov/) and the radiomics image from TCIA (https://www.cancerimagingarchive.net/).
References
Caudell JJ, et al.: NCCN Guidelines® Insights: Head and Neck Cancers, Version 1.2022. J Natl Compr Canc Netw 20:224–234, 2022
Gu Z, et al.: Pharmacogenomic landscape of head and neck squamous cell carcinoma informs precision oncology therapy. Sci Transl Med 14:eabo5987, 2022
Lechner M, Liu J, Masterson L, Fenton TR: HPV-associated oropharyngeal cancer: epidemiology, molecular biology and clinical management. Nat Rev Clin Oncol 19:306-327, 2022
Deneka AY, et al.: Association of TP53 and CDKN2A Mutation Profile with Tumor Mutation Burden in Head and Neck Cancer. Clin Cancer Res 28:1925-1937, 2022
Schinke H, et al.: A transcriptomic map of EGFR-induced epithelial-to-mesenchymal transition identifies prognostic and therapeutic targets for head and neck cancer. Mol Cancer 21:178, 2022
Chen J, et al.: Reprogramming immunosuppressive myeloid cells by activated T cells promotes the response to anti-PD-1 therapy in colorectal cancer. Signal Transduct Target Ther 6:4, 2021
Sakaguchi S, Yamaguchi T, Nomura T, Ono M: Regulatory T cells and immune tolerance. Cell 133:775-787, 2008
van der Veeken J, et al.: Genetic tracing reveals transcription factor Foxp3-dependent and Foxp3-independent functionality of peripherally induced Treg cells. Immunity 55, 2022
Yang S, et al.: Induced, but not natural, regulatory T cells retain phenotype and function following exposure to inflamed synovial fibroblasts. Sci Adv 6, 2020
Jia H, et al.: The expression of FOXP3 and its role in human cancers. Biochim Biophys Acta Rev Cancer 1871:170-178, 2019
Cui X, et al.: Dual CRISPR interference and activation for targeted reactivation of X-linked endogenous FOXP3 in human breast cancer cells. Mol Cancer 21:38, 2022
Peng J, Yang S, Ng CSH, Chen GG: The role of FOXP3 in non-small cell lung cancer and its therapeutic potentials. Pharmacol Ther 241:108333, 2023
Ma S-R, et al.: Blockade of adenosine A2A receptor enhances CD8+ T cells response and decreases regulatory T cells in head and neck squamous cell carcinoma. Mol Cancer 16:99, 2017
Oreiller V, et al.: Head and neck tumor segmentation in PET/CT: The HECKTOR challenge. Med Image Anal 77:102336, 2022
Chen Q, et al.: Radiomics in precision medicine for gastric cancer: opportunities and challenges. Eur Radiol 32:5852-5868, 2022
Lv W, et al.: Context-Aware Saliency Guided Radiomics: Application to Prediction of Outcome and HPV-Status from Multi-Center PET/CT Images of Head and Neck Cancer. Cancers (Basel) 14, 2022
Zhang Q, et al.: Predicting local persistence/recurrence after radiation therapy for head and neck cancer from PET/CT using a multi-objective, multi-classifier radiomics model. Front Oncol 12:955712, 2022
Fang Q, Chen H: The significance of m6A RNA methylation regulators in predicting the prognosis and clinical course of HBV-related hepatocellular carcinoma. Mol Med 26:60, 2020
Peng H, Long F, Ding C: Feature selection based on mutual information: criteria of max-dependency, max-relevance, and min-redundancy. IEEE Trans Pattern Anal Mach Intell 27:1226-1238, 2005
Guyon I, Weston J, Barnhilt S, et al. Gene selection for cancer classification using support vector machines. Machine Learning 46:389-422, 2002
Okuyama K, Yanamoto S: TMEM16A as a potential treatment target for head and neck cancer. J Exp Clin Cancer Res 41:196, 2022
Shonka DC, et al.: American Head and Neck Society Endocrine Surgery Section and International Thyroid Oncology Group consensus statement on mutational testing in thyroid cancer: Defining advanced thyroid cancer and its targeted treatment. Head Neck 44:1277-1300, 2022
Qiu Y, et al.: FOXP3+ regulatory T cells and the immune escape in solid tumours. Frontiers In Immunology 13:982986, 2022
Wang J, Gong R, Zhao C, Lei K, Sun X, Ren H: Human FOXP3 and tumour microenvironment. Immunology 168:248-255, 2023
Revenko A, et al.: Direct targeting of FOXP3 in Tregs with AZD8701, a novel antisense oligonucleotide to relieve immunosuppression in cancer. J Immunother Cancer 10, 2022
Ma L, Diao B, Huang Z, Wang B, Yu J, Meng X: The efficacy and possible mechanisms of immune checkpoint inhibitors in treating non-small cell lung cancer patients with epidermal growth factor receptor mutation. Cancer Commun (Lond) 41:1314-1330, 2021
Jiang M, et al.: FOXP3-based immune risk model for recurrence prediction in small-cell lung cancer at stages I-III. J Immunother Cancer 9, 2021
Adil AAM, et al.: Differential expression of Helios, Neuropilin-1 and FoxP3 in head and neck squamous cell carcinoma (HNSCC) patients. 3 Biotech 9:178, 2019
Chen BJ, Zhao JW, Zhang DH, Zheng AH, Wu GQ: Immunotherapy of Cancer by Targeting Regulatory T cells. Int Immunopharmacol 104:108469, 2022
Duhen R, et al.: PD-1 and ICOS coexpression identifies tumor-reactive CD4+ T cells in human solid tumors. J Clin Invest 132, 2022
Xing X, et al.: Effect of neoadjuvant chemotherapy on the immune microenvironment in gastric cancer as determined by multiplex immunofluorescence and T cell receptor repertoire analysis. J Immunother Cancer 10, 2022
Chen H, et al.: Depleting tumor-associated Tregs via nanoparticle-mediated hyperthermia to enhance anti-CTLA-4 immunotherapy. Nanomedicine (Lond) 15:77-92, 2020
Pei X, et al.: Comparison of Different Machine Models Based on Contrast-Enhanced Computed Tomography Radiomic Features to Differentiate High From Low Grade Clear Cell Renal Cell Carcinomas. Front Oncol 11:659969, 2021
An H, et al.: CT texture analysis in histological classification of epithelial ovarian carcinoma. Eur Radiol 31:5050-5058, 2021
Aerts HJWL, et al.: Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach. Nat Commun 5:4006, 2014
Seidler M, et al.: Dual-Energy CT Texture Analysis With Machine Learning for the Evaluation and Characterization of Cervical Lymphadenopathy. Comput Struct Biotechnol J 17:1009-1015, 2019
Wang F, Zhang W, Chai Y, Wang H, Liu Z, He Y: Constrast-enhanced computed tomography radiomics predicts CD27 expression and clinical prognosis in head and neck squamous cell carcinoma. Front Immunol 13:1015436, 2022
Yuan Y, Ren J, Shi Y, Tao X: MRI-based radiomic signature as predictive marker for patients with head and neck squamous cell carcinoma. Eur J Radiol 117:193-198, 2019
Funding
This study was supported by the Guizhou Provincial Health Commission Science and Technology Fund (gzwkj2021-053), the Natural Science Foundation of Guizhou Province (Qian Ke He Basic Project ZK[2023]525), the Science and Technology Plan Project in the Field of Social Development in Zhuhai (No. 2220004000169), the Scientific Research start-up Project of Zhuhai People's Hospital (No. 2021KYQD-04) and the Zunyi Medical University Graduate Student Research Fund (No. ZYK148).
Author information
Authors and Affiliations
Contributions
XG and GZ designed the study. NC, GH, and GF collected the data and performed the calculations. YW, JY, and KZ wrote the main manuscript text, and all the authors approved the manuscript.
Corresponding authors
Ethics declarations
Ethnics Approval
Both TCGA and TCIA data are public, so our study was exempted from the approval of the Ethics Committee of the Second Affiliated Hospital of Zunyi Medical University.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Ye, J., Zhou, K. et al. Radiomics Features on Enhanced Computed Tomography Predict FOXP3 Expression and Clinical Prognosis in Patients with Head and Neck Squamous Cell Carcinoma. J Digit Imaging. Inform. med. (2024). https://doi.org/10.1007/s10278-023-00910-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10278-023-00910-0