Abstract
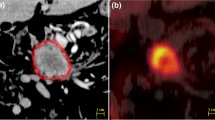
We sought to determine whether dual-energy computed tomography (DECT) measurements correlate with positron emission tomography (PET) standardized uptake values (SUVs) in pancreatic adenocarcinoma, and to determine the optimal DECT imaging variables and modeling strategy to produce the highest correlation with maximum SUV (SUVmax). We reviewed 25 patients with unresectable pancreatic adenocarcinoma seen at Mayo Clinic, Scottsdale, Arizona, who had PET–computed tomography (PET/CT) and enhanced DECT performed the same week between March 25, 2010 and December 9, 2011. For each examination, DECT measurements were taken using one of three methods: (1) average values of three tumor regions of interest (ROIs) (method 1); (2) one ROI in the area of highest subjective DECT enhancement (method 2); and (3) one ROI in the area corresponding to PET SUVmax (method 3). There were 133 DECT variables using method 1, and 89 using the other methods. Univariate and multivariate analysis regression models were used to identify important correlations between DECT variables and PET SUVmax. Both R 2 and adjusted R 2 were calculated for the multivariate model to compensate for the increased number of predictors. The average SUVmax was 5 (range, 1.8–12.0). Multivariate analysis of DECT imaging variables outperformed univariate analysis (r = 0.91; R 2 = 0.82; adjusted R 2 = 0.75 vs r < 0.58; adjusted R 2 < 0.34). Method 3 had the highest correlation with PET SUVmax (R 2 = 0.82), followed by method 1 (R 2 = 0.79) and method 2 (R 2 = 0.57). DECT thus has clinical potential as a surrogate for, or as a complement to, PET in patients with pancreatic adenocarcinoma.





Similar content being viewed by others
Abbreviations
- CfsSubsetEval:
-
Correlation-based feature subset selection evaluation
- CT:
-
Computed tomography
- DECT:
-
Dual-energy computed tomography
- PA:
-
Pancreatic adenocarcinoma
- PET:
-
Positron emission tomography
- ROI:
-
Region of interest
- SUV:
-
Standardized uptake value
- SUVmax :
-
Maximum standardized uptake value
- VIF:
-
Variance inflation factor
References
National Comprehensive Cancer Network (NCCN). NCCN clinical practice guidelines in oncology: pancreatic adenocarcinoma [Internet]. Fort Washington (PA): National Comprehensive Cancer Network; 2011 Dec 7 [updated 2013 Apr 9; cited 2012 Aug 2012]. Available from: http://www.nccn.org/professionals/physician_gls/PDF/pancreatic.pdf
Montgomery DC, Peck EA, Vining GG. Introduction to linear regression analysis. 4th ed. Hoboken (NJ). Wiley-Interscience; c2006: p. 110
Pery C, Meurette G, Ansquer C, Frampas E, Regenet N: Role and limitations of 18F-FDG positron emission tomography (PET) in the management of patients with pancreatic lesions. Gastroenterol Clin Biol 34(8–9):465–474, 2010
Delbeke D, Martin WH: PET and PET/CT for pancreatic malignancies. Surg Oncol Clin N Am 19(2):235–254, 2010
Cameron K, Golan S, Simpson W, Peti S, Roayaie S, Labow D, Kostakoglu L: Recurrent pancreatic carcinoma and cholangiocarcinoma: 18F-fluorodeoxyglucose positron emission tomography/computed tomography (PET/CT). Abdom Imaging 36(4):463–471, 2011
Silva AC, Morse BG, Hara AK, Paden RG, Hongo N, Pavlicek W: Dual-energy (spectral) CT: applications in abdominal imaging. Radiographics 31(4):1031–1046, 2011
Graser A, Johnson TR, Bader M, Staehler M, Haseke N, Nikolaou K, Reiser MF, Stief CG, Becker CR: Dual energy CT characterization of urinary calculi: initial in vitro and clinical experience. Investig Radiol 43(2):112–119, 2008
Graser A, Johnson TR, Hecht EM, Becker CR, Leidecker C, Staehler M, Stief CG, Hildebrandt H, Godoy MC, Finn ME, Stepansky F, Reiser MF, Macari M: Dual-energy CT in patients suspected of having renal masses: can virtual nonenhanced images replace true nonenhanced images? Radiology 252(2):433–440, 2009
Macari M, Spieler B, Kim D, Graser A, Megibow AJ, Babb J, Chandarana H: Dual-source dual-energy MDCT of pancreatic adenocarcinoma: initial observations with data generated at 80 kVp and at simulated weighted-average 120 kVp. AJR Am J Roentgenol 194(1):W27–W32, 2010
Mileto A, Mazziotti S, Gaeta M, Bottari A, Zimbaro F, Giardina C, Ascenti G: Pancreatic dual-source dual-energy CT: is it time to discard unenhanced imaging? Clin Radiol 67(4):334–339, 2012
Schmid-Bindert G, Henzler T, Chu TQ, Meyer M, Nance Jr, JW, Schoepf UJ, Dinter DJ, Apfaltrer P, Krissak R, Manegold C, Schoenberg SO, Fink C: Functional imaging of lung cancer using dual energy CT: how does iodine related attenuation correlate with standardized uptake value of 18FDG-PET-CT? Eur Radiol 22(1):93–103, 2012
Hall M, Frank E, Holmes G, Pfahringer B, Reutemann P, Witten IH: The WEKA data mining software: an update. SIGKDD Explor 11(1):10–18, 2009
Hastie T, Tibshirani R, Friedman J, editors. The elements of statistical learning: data mining, inference, and prediction. New York: Springer Science and Illustration Media, Inc.; c2001. Chapter 3, Linnear methods for regression; p. 41–78
Guyon I, Elisseeff A: An introduction to variable and feature selection. JMLR 3:1157–1182, 2003
Seo S, Doi R, Machimoto T, Kami K, Masui T, Hatano E, Ogawa K, Higashi T, Uemoto S: Contribution of 18F-fluorodeoxyglucose positron emission tomography to the diagnosis of early pancreatic carcinoma. J Hepatobiliary Pancreat Surg 15(6):634–639, 2008
Koyama K, Okamura T, Kawabe J, Nakata B, Chung KH, Ochi H, Yamada R: Diagnostic usefulness of FDG PET for pancreatic mass lesions. Ann Nucl Med 15(3):217–224, 2001
Delbeke D, Rose DM, Chapman WC, Pinson CW, Wright JK, Beauchamp RD, Shyr Y, Leach SD: Optimal interpretation of FDG PET in the diagnosis, staging and management of pancreatic carcinoma. J Nucl Med 40(11):1784–1791, 1999
Lee SM, Kim TS, Lee JW, Kim SK, Park SJ, Han SS: Improved prognostic value of standardized uptake value corrected for blood glucose level in pancreatic cancer using F-18 FDG PET. Clin Nucl Med 36(5):331–336, 2011
Haaga JR, Alfidi RJ, Zelch MG, Meany TF, Boller M, Gonzalez L, Jelden GL: Computed tomography of the pancreas. Radiology 120:589–595, 1976
Sheedy 2nd, PF, Stephens DH, Hattery RR, MacCarty RL: Computed tomography in the evaluation of patients with suspected carcinoma of the pancreas. Radiology 124(3):731–737, 1977
Ros PR, Mortele KJ: Imaging features of pancreatic neoplasms. JBR-BTR 84(6):239–249, 2001
Drabycz S, Stockwell RG, Mitchell JR: Image texture characterization using the discrete orthonormal S-transform. J Digit Imaging 22(6):696–708, 2009
Zhang Y, Zhu H, Mitchell JR, Costello F, Metz LM: T2 MRI texture analysis is a sensitive measure of tissue injury and recovery resulting from acute inflammatory lesions in multiple sclerosis. Neuroimage 47(1):107–111, 2009
Goh V, Ganeshan B, Nathan P, Juttla JK, Vinayan A, Miles KA: Assessment of response to tyrosine kinase inhibitors in metastatic renal cell cancer: CT texture as a predictive biomarker. Radiology 261(1):165–171, 2011
Wang H, Guo XH, Jia ZW, Li HK, Liang ZG, Li KC, He Q: Multilevel binomial logistic prediction model for malignant pulmonary nodules based on texture features of CT image. Eur J Radiol 74(1):124–129, 2010
Yu H, Caldwell C, Mah K, Poon I, Balogh J, MacKenzie R, Khaouam N, Tirona R: Automated radiation targeting in head-and-neck cancer using region-based texture analysis of PET and CT images. Int J Radiat Oncol Biol Phys 75(2):618–625, 2009
Conflict of Interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oldan, J., He, M., Wu, T. et al. Pilot Study: Evaluation of Dual-Energy Computed Tomography Measurement Strategies for Positron Emission Tomography Correlation in Pancreatic Adenocarcinoma. J Digit Imaging 27, 824–832 (2014). https://doi.org/10.1007/s10278-014-9707-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10278-014-9707-y