Abstract
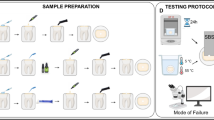
This study was performed to evaluate the effect of diabetes mellitus (DM) on the tensile bond strength (TBS) of dental composite resin bonding to enamel and dentin of extracted human teeth. Thirty caries-free human premolar teeth (10 from type 1 DM (D1), 10 from type 2 DM (D2), and 10 from non-diabetic individuals (control)) were wet ground and polished to obtain flat surfaces of dentin (n = 10). 37% phosphoric acid was used to etch dentin specimens for 15 s. After the application of the adhesive system, the composite resins were applied and cured for 20 s. Then, specimens were subjected to TBS testing by Universal Testing Machine (1 mm/min). One specimen from each group was prepared and observed under a scanning electron microscope (SEM) and a dot map was generated. Data were analyzed by ANOVA and post hoc Tukey tests (α = 0.05). The TBS values were significantly higher in the control group compared to the D1 and D2 groups (P < 0.05). Between DM groups, D2 values were significantly higher than those in the D1 group. Unlike DM groups, the zigzag fracture pattern was only noticed in the control group. DM adversely affected the TBS of dental composite resins to dentin; this negative effect is more exaggerated by type 1 DM than type 2 DM.





Similar content being viewed by others
References
Araki E, Miyazaki J-I. Metabolic disorders in diabetes mellitus: impact of mitochondrial function and oxidative stress on diabetes and its complications. Antioxid Redox Signal. 2007;9(3):289–91.
Papapanou PN. Periodontal diseases: epidemiology. Ann Periodontol. 1996;1(1):1–36.
Fouad AF. Diabetes mellitus as a modulating factor of endodontic infections. J Dent Educ. 2003;67(4):459–67.
Mealey BL, Moritz AJ. Hormonal influences: effects of diabetes mellitus and endogenous female sex steroid hormones on the periodontium. Periodontol. 2003;32(1):59–81.
De Pommereau V, et al. Periodontal status in insulin-dependent diabetic adolescents. J Clin Periodontol. 1992;19(9):628–32.
Gusberti F, et al. Puberty gingivitis in insulin-dependent diabetic children: I cross-sectional observations. J Periodontol. 1983;54(12):714–20.
Sastrowijoto S, et al. Improved metabolic control, clinical periodontal status and subgingival microbiology in insulin-dependent diabetes mellitus: a prospective study. J Clin Periodontol. 1990;17(4):233–42.
Karjalainen KM, Knuuttila ML. The onset of diabetes and poor metabolic control increases gingival bleeding in children and adolescents with insulin-dependent diabetes mellitus. J Clin Periodontol. 1996;23(12):1060–7.
Bačić M, Plančak D, Granić M. CPITN assessment of periodontal disease in diabetic patients. J Periodontol. 1988;59(12):816–22.
Emrich LJ, Shlossman M, Genco RJ. Periodontal disease in non-insulin-dependent diabetes mellitus. J Periodontol. 1991;62(2):123–31.
Shlossman M, et al. Type 2 diabetes mellitus and periodontal disease. J Am Dent Assoc. 1990;121(4):532–6.
Collin H-L, et al. Caries in patients with non-insulin-dependent diabetes mellitus. Oral Surg Oral Med Oral Pathol Oral Radiol Endodontol. 1998;85(6):680–5.
Moore PA, et al. Type 1 diabetes mellitus and oral health: assessment of coronal and root caries. Commun Dent Oral Epidemiol. 2001;29(3):183–94.
Lin B, et al. Dental caries in older adults with diabetes mellitus. Spec Care Dentist. 1999;19(1):8–14.
Twetman S, et al. Caries incidence in young type 1 diabetes mellitus patients in relation to metabolic control and caries-associated risk factors. Caries Res. 2002;36(1):31–5.
Iughetti L, et al. Oral health in children and adolescents with IDDM-a review. J Pediatr Endocrinol Metab. 1999;12(5):603–10.
Miralles Jordá, L., et al., Dental caries in type 1 diabetics: influence of systemic factors of the disease upon the development of dental caries. 2006.
Garber SE, et al. The effect of hyperglycemia on pulpal healing in rats. J Endod. 2009;35(1):60–2.
Bin G, et al. Altered circadian rhythm of pulp sensibility in elderly diabetic and hypertensive patients. Chin Med J. 2007;120(11):1024–6.
Zhang Y-R, et al. Review of research on the mechanical properties of the human tooth. Int J Oral Sci. 2014;6(2):61–9.
Lu QL, et al. Bcl-2 expression in adult and embryonic non-haematopoietic tissues. J Pathol. 1993;169(4):431–7.
Shirzaiy M, et al. Estimation of salivary sodium, potassium, calcium, phosphorus and urea in type II diabetic patients. Diabetes Metab Syndr. 2015;9(4):332–6.
Saghiri MA, et al. Effect of diabetes on tubular density and push-out bond strength of MTA to dentin. J Endod. 2020. https://doi.org/10.1016/j.joen.2020.07.025.
Sudsangiam S, van Noort R. Do dentin bond strength tests serve a useful purpose. J Adhes Dent. 1999;1(1):57–67.
Yamazaki PC, Bedran-Russo AK, Pereira PN. Importance of the hybrid layer on the bond strength of restorations subjected to cyclic loading. J Biomed Mater Res B Appl Biomater. 2008;84(1):291–7.
Betancourt DE, Baldion PA, Castellanos JE. Resin–dentin bonding interface: mechanisms of degradation and strategies for stabilization of the hybrid layer. Int J Biomater. 2019;2019:5268342.
Burke F, et al. Methods used in dentine bonding tests: an analysis of 102 investigations on bond strength. Eur J Prosthodont Restor Dent. 2008;16(4):158–65.
Saghiri, et al. The effect of diabetes mellitus on the shear bond strength of composite resin to dentin and enamel. Odontology. 2021. https://doi.org/10.1007/s10266-021-00641-0.
Saghiri MA, et al. A novel polyurethane expandable root canal sealer. J Endod. 2020. https://doi.org/10.1016/j.joen.2020.12.007.
Sano H, et al. The tensile bond strength test: Its historical background and application to bond testing. Jpn Dent Sci Rev. 2020;56(1):24–31.
Frankenberger R, et al. Operator vs. material influence on clinical outcome of bonded ceramic inlays. Dent Mater. 2009;25(8):960–8.
Lohbauer U, et al. Correlation of in vitro fatigue data and in vivo clinical performance of a glassceramic material. Dent Mater. 2008;24(1):39–44.
Silva EM, et al. Influence of organic acids present in the oral biofilm on the tensile bond strength of adhesive systems to human dentin. J Biomed Mater Res B Appl Biomater. 2012;100B(3):735–41.
Pribadi N, Soetojo A. Effects of different saliva pH on hybrid composite resin surface roughness. Dent J. 2011;44:63.
Abou Neel EA, et al. Demineralization-remineralization dynamics in teeth and bone. Int J Nanomed. 2016;11:4743–63.
Shillitoe E, et al. The oral microflora in obesity and type-2 diabetes. J Oral Microbiol. 2012;4:19013.
Yeh C-K, et al. Hyperglycemia and xerostomia are key determinants of tooth decay in type 1 diabetic mice. Lab Invest. 2012;92(6):868–82.
Arrieta-Blanco JJ, et al. Dental problems in patients with diabetes mellitus (II): gingival index and periodontal disease. Med Oral. 2003;8(4):233–47.
Casagrande L, et al. Longevity and associated risk factors in adhesive restorations of young permanent teeth after complete and selective caries removal: a retrospective study. Clin Oral Invest. 2017;21(3):847–55.
Culshaw S. Periodontal disease: its impact on restorative dentistry. Prim Dent J. 2017;6(1):25–31.
Kikushima D, et al. Micro-shear bond strength of adhesive systems to cementum. Am J Dent. 2005;18(5):364–8.
Abbassy MA, et al. Diabetes detrimental effects on enamel and dentine formation. J Dent. 2015;43(5):589–96.
Atar M, et al. Enamel mineral concentration in diabetic rodents. Eur Arch Paediatr Dent. 2007;8(4):195–200.
Delaviz Y, Finer Y, Santerre JP. Biodegradation of resin composites and adhesives by oral bacteria and saliva: a rationale for new material designs that consider the clinical environment and treatment challenges. Dent Mater. 2014;30(1):16–32.
Sajjanhar I, Chandra A, Tikku A. Matrix metalloproteinases: its effect on dentin bonding. J Biol Nat. 2017;7:118–22.
Gupta N, et al. The influence of type 2 diabetes mellitus on salivary matrix metalloproteinase-8 levels and periodontal parameters: a study in an Indian population. Eur J Dent. 2015;9(3):319–23.
Saghiri MA, Saghiri AM. In Memoriam: Dr. Hajar Afsar Lajevardi MD, MSc, MS (1955–2015). Iran J Pediatr. 2017;27(1):1.
Acknowledgements
MAS is a recipient of the New Jersey Health Foundation Award. This publication is dedicated to the memory of Dr. H. Afsar Lajevardi, a legendry pediatrician (1953–2015) [47]. We will never forget Dr. H Afsar Lajevardi’s kindness and support. The views expressed in this paper are those of the authors and do not necessarily reflect the views or policies of the affiliated organizations. The authors hereby announced that they have active cooperation in this scientific study and preparation of the present manuscript. The authors confirm that they have no financial involvement with any commercial company or organization with direct financial interest regarding the materials used in this study. The authors offer special thanks to Dr. Amir Fakhrzadeh for the help in collecting the teeth for use in this current study and to Dr. Saied Amini for helping us conduct the statistical analyses.
Author information
Authors and Affiliations
Contributions
Conceptualization and methodology: MAS, SMM; investigation: MAS, DN, PF; formal analysis: MAS, DN; writing—original draft: DN, PF; writing—review and editing: MAS, DN, PF, SMM.
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saghiri, M.A., Freag, P., Nath, D. et al. The effect of diabetes on the tensile bond strength of a restorative dental composite to dentin. Odontology 110, 648–654 (2022). https://doi.org/10.1007/s10266-022-00697-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10266-022-00697-6