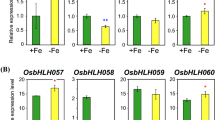
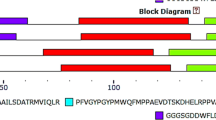
Abstract
Iron (Fe) is a micronutrient that is essential for plant development and growth. Basic helix-loop-helix (bHLH) transcription factors are a superfamily of transcription factors that are important regulatory components in transcriptional networks in plants. bHLH transcription factors have been divided into subclasses based on their amino acid sequences and domain structures. Among the members of clade IVb (PYE, bHLH121, and bHLH11), the functions of bHLH11 remain unclear. In the present study, we characterized bHLH11 as a negative regulator of Fe homeostasis. bHLH11 expression levels were high in the roots and up-regulated after plants were transferred to Fe sufficient conditions. Although T-DNA knockout mutants of bHLH11 were lethal, dominant negative (DN-) and overexpression (OX-) of bHLH11 plants exhibited sensitivity to Fe deficiency. Furthermore, the expression of FIT, a master regulator of Fe deficiency responses, was suppressed in the transgenic plants. These results suggest that the transcriptional repressor bHLH11 functions as a negative regulator of FIT-dependent Fe uptake and modulates Fe levels in Arabidopsis plants. Salicylic acid (SA) modulates the expression of genes involved in Fe-deficient responses. We found that SA levels were elevated in DN- and OX-bHLH11 plants. The T-DNA insertion mutant sid2-1, which was defective for the production of SA, did not exhibit sensitivity to Fe deficiency; however, the crossed plants of OX-bHLH11 and sid2-1 relieved sensitivity to the Fe deficiency observed in OX-bHLH11 plants. These results suggest that the accumulation of SA is closely related to iron homeostasis.







Similar content being viewed by others
References
Alonso JM (2003) Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301(5633):653–657
Arnon DI (1949) Copper enzymes in isolated chloroplasts. polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15
Aznar A, Chen NW, Rigault M, Riache N, Joseph D, Desmaële D, Mouille G, Boutet A, Soubigou-Taconnat L, Renou J, Thomine S, Expert D, Dellagi A (2014) Scavenging iron: a novel mechanism of plant immunity activation by microbial siderophores. Plant Physiol 164:2167–2183
Aznar A, Chen NW, Thomine S, Dellagi A (2015) Immunity to plant pathogens and iron homeostasis. Plant Sci 240:90–97
Bauer P, Ling HQ, Guerinot ML (2007) Fit, the fer-like iron deficiency induced transcription factor in arabidopsis. Plant Physiol Biochem 45:260–261
Brumbarova T, Bauer P, Ivanov R (2015) Molecular mechanisms governing Arabidopsis iron uptake. Trends Plant Sci 20:124–133
Colangelo EP, Guerinot ML (2004) The essential basic helix-loop-helix protein FIT1 is required for the iron deficiency response. Plant Cell 16:3400–3412
Dellagi A, Segond D, Rigaut M, Fegard M, Simon C, Saindrenan P, Expert D (2009) Microbial Siderophores exert a subtle role in Arabidopsis during infection by manipulating the immune response and the iron status. Plant Physiol 150:1687–1696
Eide D, Broderius M, Fett J, Guerinot ML (1996) A novel iron-regulated metal transporter from plants identified by functional expression in yeast. Proc Natl Acad Sci USA 93:5624–5628
Fragnière C, Serrano M, Abou-Mansour E, Métraux JP, L’Haridon F (2011) Salicylic acid and its location in response to biotic and abiotic stress. FEBS Lett 585:1847–1852
Furniss JJ, Spoel SH (2015) Cullin-RING ubiquitin ligases in salicylic acid-mediated plant immune signaling. Front Plant Sci 6:154. https://doi.org/10.3389/fpls.2015.00154
García MJ, Lucena C, Romera FJ, Alcántara E, Pérez-Vicente R (2010) Ethylene and nitric oxide involvement in the up-regulation of key genes related to iron acquisition and homeostasis in Arabidopsis. J Exp Bot 61:3885–3899
Garcion C, Lohmann A, Lamodière E, Catinot J, Buchala A, Doermann P, Métraux JP (2008) Characterization and biological function of the isochorismate synthase2 gene of Arabidopsis. Plant Physiol 147:1279–1287
Gierth M, Mäser P, Schroeder JI (2005) The potassium transporter AtHAK5 functions in K+ deprivation-induced high-affinity K+ uptake and AKT1 K+ channel contribution to K+ uptake kinetics in Arabidopsis roots. Plant Physiol 137:1105–1114
Gyana RR, Sunita S (2015) Role of iron in plant growth and metabolism. Rev Agric Sci 3:1–24
Hiratsu K, Ohta M, Matsui K, Ohme-Takagi M (2002) The SUPERMAN protein is an active repressor whose carboxy-terminal repression domain is required for the development of normal flowers. FEBS Lett 514:351–354
Hiratsu K, Matsui K, Koyama T, Ohme-Takagi M (2003) Dominant repression of target genes by chimeric repressors that include the EAR motif, a repression domain, in Arabidopsis. Plant J 34:733–739
Hiratsu K, Mitsuda N, Matsui K, Ohme-Takagi M (2004) Identification of the minimal repression domain of SUPERMAN shows that the DLELRL hexapeptide is both necessary and sufficient for repression of transcription in Arabidopsis. Biochem Biophys Res Commun 321:172–178
Hong JP, Takeshi Y, Kondou Y, Schachtman DP, Matsui M, Shin R (2013) Identification and characterization of transcription factors regulating Arabidopsis HAK5. Plant Cell Physiol 54:1478–1490
Ivanov R, Brumbarova T, Bauer P (2012) Fitting into the harsh reality: regulation of iron-deficiency responses in dicotyledonous plants. Mol Plant 5:27–42
Jaspers P, Blomster T, Brosché M, Salojärvi J, Ahlfors R, Vainonen JP, Reddy RA, Immink R, Angenent G, Turck F, Overmyer K, Kangasjärvi J (2009) Unequally redundant RCD1 and SRO1 mediate stress and developmental responses and interact with transcription factors. Plant J 60:268–279
Kang HG, Foley RC, Oñate-Sánchez L, Lin C, Singh KB (2003) Target genes for OBP3, a Dof transcription factor, include novel basic helix-loop-helix domain proteins inducible by salicylic acid. Plant J 35:362–372
Kawai-Yamada M, Ohori Y, Uchimiya H (2004) Dissection of Arabidopsis Bax inhibitor-1 suppressing Bax-, hydrogen peroxide-, and salicylic acid-induced cell death. Plant Cell 16:21–32
Lei GJ, Zhu XF, Wang ZW, Dong F, Dong NY, Zheng SJ (2014) Abscisic acid alleviates iron deficiency by promoting root iron reutilization and transport from root to shoot in Arabidopsis. Plant Cell Environ 37:852–863
Li Q, Fang X, Olave I, Han H, Yu M, Xiang P, Stamatoyannopoulos G (2006) Transcriptional potential of the γ-globin gene is dependent on the CACCC box in a developmental stage-specific manner. Nucleic Acids Res 34:3909–3916
Li H, Wang L, Yang ZM (2015) Co-expression analysis reveals a group of genes potentially involved in regulation of plant response to iron-deficiency. Gene 554:16–24
Li X, Zhang H, Ai Q, Liang G, Yu D (2016) Two bHLH transcription factors, bHLH34 and bHLH104, regulate iron homeostasis in Arabidopsis thaliana. Plant Physiol 170:2478–2493
Long TA, Tsukagoshi H, Busch W, Lahner B, Salt DE, Benfey PN (2010) The bHLH transcription factor POPEYE regulates response to iron deficiency in Arabidopsis roots. Plant Cell 22:2219–2236
Lorrain S, Vailleau F, Balagué C, Roby D (2003) Lesion mimic mutants: keys for deciphering cell death and defense pathways in plants? Trends Plant Sci 8:263–271
Mai HJ, Pateyron S, Bauer P (2016) Iron homeostasis in Arabidopsis thaliana: transcriptomic analyses reveal novel FIT-regulated genes, iron deficiency marker genes and functional gene networks. BMC Plant Biol 16:211
Marschner H, Römheld V, Kissel M (1986) Different strategies in higher plants in mobilization and uptake of iron. J Plant Nutr 9:695–713
Maruta T, Noshi M, Tanouchi A, Tamoi M, Yabuta Y, Yoshimura K, Ishikawa T, Shigeoka S (2012) H2O2-triggered retrograde signaling from chloroplasts to nucleus plays specific role in response to stress. J Biol Chem 287:11717–11729
Maurer F, Müller S, Bauer P (2011) Suppression of Fe deficiency gene expression by jasmonate. Plant Physiol Biochem 49:530–536
Maurer F, Naranjo-Arcos MA, Bauer P (2014) Responses of a triple mutant defective in three iron deficiency-induced BASIC HELIX-LOOP-HELIX genes of the subgroup Ib(2) to iron deficiency and salicylic acid. PLoS One 9:e99234
Meiser J, Lingam S, Bauer P (2011) Posttranslational regulation of the iron deficiency basic helix-loop-helix transcription factor FIT is affected by iron and nitric oxide. Plant Physiol 157:2154–2166
Naranjo-Arcos MA, Bauer P (2016) Iron nutrition, oxidative stress, and pathogen defense. Nutr Defic Chap. https://doi.org/10.5772/63204
Noshi M, Mori D, Tanabe N, Maruta T, Shigeoka S (2016) Arabidopsis clade IV TGA transcription factors, TGA10 and TGA9, are involved in ROS-mediated responses to bacterial PAMP flg22. Plant Sci 252:12–21
Noshi M, Tanabe N, Okamoto Y, Mori D, Ohme-Takagi M, Tamoi M, Shigeoka S (2018) Clade Ib basic helix-loop-helix transcription factor, bHLH101, acts as a regulatory component in photo-oxidative stress responses. Plant Sci 274:101–108
Pires N, Dolan L (2010) Origin and diversification of basic-helix-loop-helix proteins in plants. Mol Biol Evol 27:862–874
Ranieri A, Castagna A, Baldan B, Soldatini GF (2001) Iron deficiency differently affects peroxidase isoforms in sunflower. J Exp Bot 52:25–35
Robinson NJ, Procter CM, Connolly EL, Guerinot ML (1999) A ferric-chelate reductase for iron uptake from soils. Nature 397:694–697
Römheld V (1987) Different strategies for iron acquisition in higher plants. Physiol Plantarum 70:231–234
Römheld V, Marschner H (1986) Evidence for a specific uptake system for iron Phytosiderophores in roots of grasses. Plant Physiol 80:175–180
Rubio F, Santa-María GE, Rodríguez-Navarro A (2000) Cloning of Arabidopsis and barley cDNAs encoding HAK potassium transporters in root and shoot cells. Physiol Plant 109:34–43
Santi S, Schmidt W (2009) Dissecting iron deficiency-induced proton extrusion in Arabidopsis roots. New Phytol 183:1072–1084
Schmidt W, Tittel J, Schikora A (2000) Role of hormones in the induction of iron deficiency responses in Arabidopsis roots. Plant Physiol 122:1109–1118
Séguéla M, Briat JF, Vert G, Curie C (2008) Cytokinins negatively regulate the root iron uptake machinery in Arabidopsis through a growth-dependent pathway. Plant J 55:289–300
Shen C, Yang Y, Liu K, Zhang L, Guo H, Sun T, Wang H (2016) Involvement of endogenous salicylic acid in iron-deficiency responses in Arabidopsis. J Exp Bot 67:4179–4193
Sivitz AB, Hermand V, Curie C, Vert G (2012) Arabidopsis bHLH100 and bHLH101 control iron homeostasis via a FIT-independent pathway. PLoS One 7:e44843
Toledo-Ortiz G, Huq E, Quail PH (2003) The Arabidopsis basic/helix-loop-helix transcription factor family. Plant Cell 15:1749–1770
Vert G, Grotz N, Dédaldéchamp F, Gaymard F, Guerinot ML, Briat JF, Curie C (2002) IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. Plant Cell 14:1223–1233
Wang HY, Klatte M, Jakoby M, Bäumlein H, Weisshaar B, Bauer P (2007) Iron deficiency-mediated stress regulation of four subgroup Ib BHLH genes in Arabidopsis thaliana. Planta 226:897–908
Wang N, Cui Y, Liu Y, Fan H, Du J, Huang Z, Yuan Y, Wu H, Ling HQ (2013) Requirement and functional redundancy of Ib subgroup bHLH proteins for iron deficiency responses and uptake in Arabidopsis thaliana. Mol Plant 6:503–513
Yuan YX, Zhang J, Wang DW, Ling HQ (2005) AtbHLH29 of Arabidopsis thaliana is a functional ortholog of tomato FER involved in controlling iron acquisition in strategy I plants. Cell Res 15:613–621
Yuan Y, Wu H, Wang N, Li J, Zhao W, Du J, Wang D, Ling HQ (2008) FIT interacts with AtbHLH38 and AtbHLH39 in regulating iron uptake gene expression for iron homeostasis in Arabidopsis. Cell Res 18:385–397
Zaharieva TB, Abadia J (2003) Iron deficiency enhances the levels of ascorbate, glutathione, and related enzymes in sugar beet roots. Protoplasma 221:269–275
Zaharieva T, Yamashita K, Matsumoto H (1999) Iron deficiency induced changes in ascorbate content and enzyme activities related to ascorbate metabolism in cucumber roots. Plant Cell Physiol 40:273–280
Zhang J, Liu B, Li M, Feng D, Jin H, Wang P, Liu J, Xiong F, Wang J, Wang HB (2015) The bHLH transcription factor bHLH104 interacts with IAA-LEUCINE RESISTANT3 and modulates iron homeostasis in Arabidopsis. Plant Cell 27:787–805
Zheng L, Ying Y, Wang L, Wang F, Whelan J, Shou H (2010) Identification of a novel iron regulated basic helix-loop-helix protein involved in Fe homeostasis in Oryza sativa. BMC Plant Biol 10:166. https://doi.org/10.1186/1471-2229-10-166
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tanabe, N., Noshi, M., Mori, D. et al. The basic helix-loop-helix transcription factor, bHLH11 functions in the iron-uptake system in Arabidopsis thaliana. J Plant Res 132, 93–105 (2019). https://doi.org/10.1007/s10265-018-1068-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-018-1068-z