Abstract
Osteoporosis (OP), a skeletal disease making bone mechanically deteriorate and easily fracture, is a global public health issue due to its high prevalence. It has been well recognized that besides bone loss, microarchitecture degradation plays a crucial role in the mechanical deterioration of OP bones, but the specific role of microarchitecture in OP has not been well clarified and quantified from mechanics perspective. Here, we successfully decoupled and identified the specific roles of microarchitecture, bone mass and tissue property in the failure properties of cancellous bones, through μCT-based digital modeling and finite element method simulations on bone samples from healthy and ovariectomy-induced osteoporotic mice. The results show that the microarchitecture of healthy bones exhibits longitudinal superiority in mechanical properties such as the effective stiffness, strength and toughness, which fits them well to bearing loads along their longitudinal direction. OP does not only reduce bone mass but also impair the microarchitecture topology. The former is mainly responsible for the mechanical degradation of bones in magnitude, wherever the latter accounts for the breakdown of their function-favorable anisotropy, the longitudinal superiority. Hence, we identified the microarchitecture-deterioration-induced directional mismatch between material and loading as a hazardous feature of OP and defined a longitudinal superiority index as measurement of the health status of bone microarchitecture. These findings provide useful insights and guidelines for OP diagnosis and treat assessment.


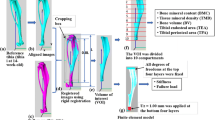
taken from the cancellous bone domain in the distal femur samples; (b) all the VOIs separated into three groups according to their BV/TV: healthy, mild OP and severe OP, with the insets showing their typical 3D-microarchitectures; (c) an example of FEM model; (d) the constitutive curve of bone tissue





Adapted from Ref.(Liu et al. 2019))





Similar content being viewed by others
References
Barak MM, Black MA (2018) A novel use of 3D printing model demonstrates the effects of deteriorated trabecular bone structure on bone stiffness and strength. J Mech Behav Biomed 78:455–464. https://doi.org/10.1016/j.jmbbm.2017.12.010
Becker DJ, Kilgore ML, Morrisey MA (2010) The societal burden of osteoporosis. Curr Rheumatol Rep 12:186–191
Bevill G, Keaveny TM (2009) Trabecular bone strength predictions using finite element analysis of micro-scale images at limited spatial resolution. Bone 44:579–584. https://doi.org/10.1016/j.bone.2008.11.020
Buehler MJ (2008) Nanomechanics of collagen fibrils under varying cross-link densities: Atomistic and continuum studies. J Mech Behav Biomed 1:59–67. https://doi.org/10.1016/j.jmbbm.2007.04.001
Buie HR, Campbell GM, Klinck RJ, MacNeil JA, Boyd SK (2007) Automatic segmentation of cortical and trabecular compartments based on a dual threshold technique for in vivo micro-CT bone analysis. Bone 41:505–515. https://doi.org/10.1016/j.bone.2007.07.007
Chappard D, Baslé M-F, Legrand E, Audran M (2008) Trabecular bone microarchitecture: a review. Morphologie 92:162–170
Ciarelli TE, Fyhrie DP, Schaffler MB, Goldstein SA (2000) Variations in three-dimensional cancellous bone architecture of the proximal femur in female hip fractures and in controls. J Bone Miner Res 15:32–40. https://doi.org/10.1359/jbmr.2000.15.1.32
Creecy A et al (2020) The age-related decrease in material properties of BALB/c mouse long bones involves alterations to the extracellular matrix. Bone 130:115126
Cummings SR, Karpf DB, Harris F, Genant HK, Ensrud K, LaCroix AZ, Black DM (2002) Improvement in spine bone density and reduction in risk of vertebral fractures during treatment with antiresorptive drugs. Am J Med 112:281–289. https://doi.org/10.1016/S0002-9343(01)01124-X
Currey JD (1986) Power law models for the mechanical properties of cancellous bone. Eng Med 15:153–154
Delmas PD, Seeman E (2004) Changes in bone mineral density explain little of the reduction in vertebral or nonvertebral fracture risk with anti-resorptive therapy. Bone 34:599–604. https://doi.org/10.1016/j.bone.2003.12.022
Donnelly E (2011) Methods for assessing bone quality: a review. Clin Orthop Relat R 469:2128–2138. https://doi.org/10.1007/s11999-010-1702-0
Enns-Bray WS, Ferguson SJ, Helgason B (2018) Strain rate dependency of bovine trabecular bone under impact loading at sideways fall velocity. J Biomech 75:46–52. https://doi.org/10.1016/j.jbiomech.2018.04.042
Feldkamp LA, Davis LC, Kress JW (1984) Practical cone-beam algorithm. Josa A 1:612–619
Follet H, Boivin G, Rumelhart C, Meunier PJ (2004) The degree of mineralization is a determinant of bone strength: a study on human calcanei. Bone 34:783–789. https://doi.org/10.1016/j.bone.2003.12.012
Fonseca H, Moreira-Goncalves D, Coriolano HJA, Duarte JA (2014) Bone quality: the determinants of bone strength and fragility. Sports Med 44:37–53. https://doi.org/10.1007/s40279-013-0100-7
Gibson LJ (2003) Cellular solids. Mrs. Bulletin 28:270–274
Gibson LJ (2005) Biomechanics of cellular solids. J Biomech 38:377–399. https://doi.org/10.1016/j.jbiomech.2004.09.027
Gross T, Pahr DH, Zysset PK (2013) Morphology-elasticity relationships using decreasing fabric information of human trabecular bone from three major anatomical locations. Biomech Model Mechan 12:793–800. https://doi.org/10.1007/s10237-012-0443-2
Guo XE, Goldstein SA (2000) Vertebral trabecular bone microscopic tissue elastic modulus and hardness do not change in ovariectomized rats. J Orthop Res 18:333–336
Hambli R (2013a) Micro-CT finite element model and experimental validation of trabecular bone damage and fracture. Bone 56:363–374. https://doi.org/10.1016/j.bone.2013.06.028
Hambli R (2013b) A quasi-brittle continuum damage finite element model of the human proximal femur based on element deletion. Med Biol Eng Comput 51:219–231. https://doi.org/10.1007/s11517-012-0986-5
Hambli R, Bettamer A, Allaoui S (2012) Finite element prediction of proximal femur fracture pattern based on orthotropic behaviour law coupled to quasi-brittle damage. Med Eng Phys 34:202–210. https://doi.org/10.1016/j.medengphy.2011.07.011
Harrison NM, McDonnell PF, O’Mahoney DC, Kennedy OD, O’Brien FJ, McHugh PE (2008) Heterogeneous linear elastic trabecular bone modelling using micro-CT attenuation data and experimentally measured heterogeneous tissue properties. J Biomech 41:2589–2596. https://doi.org/10.1016/j.jbiomech.2008.05.014
Harrison NM, McDonnell P, Mullins L, Wilson N, O’Mahoney D, McHugh PE (2013) Failure modelling of trabecular bone using a non-linear combined damage and fracture voxel finite element approach. Biomech Model Mechan 12:225–241. https://doi.org/10.1007/s10237-012-0394-7
Hengsberger S, Kulik A, Zysset P (2002) Nanoindentation discriminates the elastic properties of individual human bone lamellae under dry and physiological conditions. Bone 30:178–184. https://doi.org/10.1016/S8756-3282(01)00624-X
Homminga J, McCreadie BR, Ciarelli TE, Weinans H, Goldstein SA, Huiskes R (2002) Cancellous bone mechanical properties from normals and patients with hip fractures differ on the structure level, not on the bone hard tissue level. Bone 30:759–764. https://doi.org/10.1016/S8756-3282(02)00693-2
Jordan GR et al (2003) Increased femoral neck cancellous bone and connectivity in coxarthrosis (hip osteoarthritis). Bone 32:86–95. https://doi.org/10.1016/S8756-3282(02)00920-1
Kabel J, van Rietbergen B, Odgaard A, Huiskes R (1999) Constitutive relationships of fabric, density, and elastic properties in cancellous bone architecture. Bone 25:481–486. https://doi.org/10.1016/S8756-3282(99)00190-8
Karasuyama K, Yamamoto T, Motomura G, Sonoda K, Kubo Y, Iwamoto Y (2015) The role of sclerotic changes in the starting mechanisms of collapse: a histomorphometric and FEM study on the femoral head of osteonecrosis. Bone 81:644–648. https://doi.org/10.1016/j.bone.2015.09.009
Keaveny TM, Guo XE, Wachtel EF, Mcmahon TA, Hayes WC (1994) Trabecular bone exhibits fully linear elastic behavior and yields at low strains. J Biomech 27:1127–1136. https://doi.org/10.1016/0021-9290(94)90053-1
Keaveny TM, Morgan EF, Niebur GL, Yeh OC (2001) Biomechanics of trabecular bone. Annu Rev Biomed Eng 3:307–333. https://doi.org/10.1146/annurev.bioeng.3.1.307
Launey ME, Buehler MJ, Ritchie RO (2010) On the Mechanistic origins of toughness in bone. Annu Rev Mater Res 40:25–53. https://doi.org/10.1146/annurev-matsci-070909-104427
Licata A (2009) Bone density versus bone quality: What’s a clinician to do? Clev Clin J Med 76:331–336
Lin JT, Lane JM (2004) Osteoporosis–a review. Clin Orthop Relat R 27:126–134. https://doi.org/10.1097/01.blo.0000132404.30139.f2
Liu P, Liang X, Li Z, Zhu X, Zhang Z, Cai L (2019) Decoupled effects of bone mass, microarchitecture and tissue property on the mechanical deterioration of osteoporotic bones. Compos Part B-Eng 177:107436
Liu XS, Sajda P, Saha PK, Wehrli FW, Bevill G, Keaveny TM, Guo XE (2008) Complete volumetric decomposition of individual trabecular plates and rods and its morphological correlations with anisotropic elastic moduli in human trabecular bone. J Bone Miner Res 23:223–235. https://doi.org/10.1359/Jbmr.071009
McCreadie BR, Morris MD, Chen TC, Rao DS, Finney WF, Widjaja E, Goldstein SA (2006) Bone tissue compositional differences in women with and without osteoporotic fracture. Bone 39:1190–1195. https://doi.org/10.1016/j.bone.2006.06.008
McNamara LM (2010) Perspective on post-menopausal osteoporosis: establishing an interdisciplinary understanding of the sequence of events from the molecular level to whole bone fractures. J R Soc Interface 7:353–372. https://doi.org/10.1098/rsif.2009.0282
Morgan EF, Bayraktar HH, Keaveny TM (2003) Trabecular bone modulus-density relationships depend on anatomic site. J Biomech 36:897–904. https://doi.org/10.1016/S0021-9290(03)00071-X
Niebur GL, Feldstein MJ, Yuen JC, Chen TJ, Keaveny TM (2000) High-resolution finite element models with tissue strength asymmetry accurately predict failure of trabecular bone. J Biomech 33:1575–1583
Nyman JS, Vashishth D (2018) Assessment of bone mass, structure, and quality in rodents. In: Bilezkian JP (ed) Primer on the metabolic bone diseases and disorders of mineral metabolism. Wiley, New York, pp 93–100
O’Connor DT et al (2016) Modeling orthotropic elasticity, localized plasticity and fracture in trabecular bone. Comput Mech 58:423–439. https://doi.org/10.1007/s00466-016-1301-3
Rachner TD, Khosla S, Hofbauer LC (2011) Osteoporosis: now and the future. Lancet 377:1276–1287. https://doi.org/10.1016/S0140-6736(10)62349-5
Riggs BL, Melton LJ (1995) The worldwide problem of osteoporosis–insights afforded by epidemiology. Bone 17:S505–S511. https://doi.org/10.1016/8756-3282(95)00258-4
Rizzoli R, Laroche M, Krieg MA, Frieling I, Thomas T, Delmas P, Felsenberg D (2010) Strontium ranelate and alendronate have differing effects on distal tibia bone microstructure in women with osteoporosis. Rheumatol Int 30:1341–1348. https://doi.org/10.1007/s00296-010-1542-y
Sabet FA, Najafi AR, Hamed E, Jasiuk I (2016) Modelling of bone fracture and strength at different length scales: a review. Interface Focus. https://doi.org/10.1098/rsfs.2015.0055
Sandino C, McErlain DD, Schipilow J, Boyd SK (2017) Mechanical stimuli of trabecular bone in osteoporosis: a numerical simulation by finite element analysis of microarchitecture. J Mech Behav Biomed 66:19–27. https://doi.org/10.1016/j.jmbbm.2016.10.005
Schwiedrzik JJ, Zysset PK (2013) An anisotropic elastic-viscoplastic damage model for bone tissue. Biomech Model Mechan 12:201–213. https://doi.org/10.1007/s10237-012-0392-9
Seeman E, Delmas PD (2006) Mechanisms of disease–bone quality–the material and structural basis of bone strength and fragility. New Engl J Med 354:2250–2261
Silva MJ, Brodt MD, Lynch MA, McKenzie JA, Tanouye KM, Nyman JS, Wang XD (2009) Type 1 diabetes in young rats leads to progressive trabecular bone loss, cessation of cortical bone growth, and diminished whole bone strength and fatigue life. J Bone Miner Res 24:1618–1627. https://doi.org/10.1359/Jbmr.090316
Stipsitz M, Zysset PK, Pahr DH (2020) Efficient materially nonlinear mu FE solver for simulations of trabecular bone failure. Biomech Model Mechan 19:861–874. https://doi.org/10.1007/s10237-019-01254-x
Tertuliano OA, Greer JR (2016) The nanocomposite nature of bone drives its strength and damage resistance. Nat Mater 15:1195–1202
Tertuliano OA, Edwards BW, Meza LR, Deshpande VS, Greer JR (2021) Nanofibril-mediated fracture resistance of bone Bioinspir Biomim 16:035001. https://doi.org/10.1088/1748-3190/abdd9d
Thurner PJ et al (2007) High-speed photography of compressed human trabecular bone correlates whitening to microscopic damage. Eng Fract Mech 74:1928–1941
Turner CH, Cowin SC, Rho JY, Ashman RB, Rice JC (1990) The fabric dependence of the orthotropic elastic-constants of cancellous bone. J Biomech 23:549–561. https://doi.org/10.1016/0021-9290(90)90048-8
Van Rietbergen B, Odgaard A, Kabel J, Huiskes R (1998) Relationships between bone morphology and bone elastic properties can be accurately quantified using high-resolution computer reconstructions. J Orthop Res 16:23–28. https://doi.org/10.1002/jor.1100160105
Verhulp E, Van Rietbergen B, Muller R, Huiskes R (2008) Micro-finite element simulation of trabecular-bone post-yield behaviour–effects of material model, element size and type. Comput Method Biomec 11:389–395. https://doi.org/10.1080/10255840701848756
Wang H et al (2013) Accuracy of individual trabecula segmentation based plate and rod finite element models in idealized trabecular bone microstructure. J Biomech Eng-T Asme 135:044502. https://doi.org/10.1115/1.4023983
Wang J et al (2015) Trabecular plates and rods determine elastic modulus and yield strength of human trabecular bone. Bone 72:71–80. https://doi.org/10.1016/j.bone.2014.11.006
Wang ZW et al (2017) Unique local bone tissue characteristics in iliac crest bone biopsy from adolescent idiopathic scoliosis with severe spinal deformity. Sci Rep-Uk 7:40265. https://doi.org/10.1038/srep40265
Werner B, Ovesy M, Zysset P (2019) An explicit microFE approach to investigate the post-yield behaviour of trabecular bone under large deformations. Int J Numer Meth Bio 35:e3188
Zysset P, Goulet R, Hollister S (1998a) A global relationship between trabecular bone morphology and homogenized elastic properties. J Biomech Eng 120:640–646
Zysset PK, Guo XE, Hoffler CE, Moore KE, Goldstein SA (1998b) Mechanical properties of human trabecular bone lamellae quantified by nanoindentation. Technol Health Care 6:429–432
Acknowledgements
Our thanks are due to Prof. Huajian Gao, Bin Liu, Dr. Qifang Yin and Mr. Kun Geng for their valuable suggestions. The work was supported by National Natural Science Foundation of China (Grant Nos. 11772240, 11720101002, 11542001, 11502175), Jiangsu Natural Science Foundation (Grant No. BK20150381), the Fundamental Research Funds for the Central University, Translational Medicine and Interdisciplinary Research Joint Fund of Zhongnan Hospital of Wuhan University (Grant No. ZNJC201927) and the Innovative Group Development Program at School of Civil Engineering, Wuhan University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Z., Liu, P., Yuan, Y. et al. Loss of longitudinal superiority marks the microarchitecture deterioration of osteoporotic cancellous bones. Biomech Model Mechanobiol 20, 2013–2030 (2021). https://doi.org/10.1007/s10237-021-01491-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-021-01491-z