Abstract
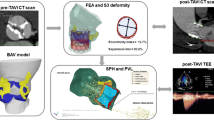
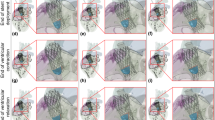
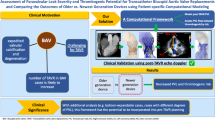
Transcatheter aortic valve replacement (TAVR) has emerged as an effective alternative to conventional surgical valve replacement in high-risk patients afflicted by severe aortic stenosis. Despite newer-generation devices enhancements, post-procedural complications such as paravalvular leakage (PVL) and related thromboembolic events have been hindering TAVR expansion into lower-risk patients. Computational methods can be used to build and simulate patient-specific deployment of transcatheter aortic valves (TAVs) and help predict the occurrence and degree of PVL. In this study finite element analysis and computational fluid dynamics were used to investigate the influence of procedural parameters on post-deployment hemodynamics on three retrospective clinical cases affected by PVL. Specifically, TAV implantation depth and balloon inflation volume effects on stent anchorage, degree of paravalvular regurgitation and thrombogenic potential were analyzed for cases in which Edwards SAPIEN and Medtronic CoreValve were employed. CFD results were in good agreement with corresponding echocardiography data measured in patients in terms of the PVL jets locations and overall PVL degree. Furthermore, parametric analyses demonstrated that positioning and balloon over-expansion may have a direct impact on the post-deployment TAVR performance, achieving as high as 47% in PVL volume reduction. While the model predicted very well clinical data, further validation on a larger cohort of patients is needed to verify the level of the model’s predictions in various patient-specific conditions. This study demonstrated that rigorous and realistic patient-specific numerical models could potentially serve as a valuable tool to assist physicians in pre-operative TAVR planning and TAV selection to ultimately reduce the risk of clinical complications.











Similar content being viewed by others
Change history
21 January 2020
This is to inform that the original article was published without the “Conflict of Interest” statement.
21 January 2020
This is to inform that the original article was published without the ���Conflict of Interest��� statement.
References
Abdelghani M, Soliman OII, Schultz C, Vahanian A, Serruys PW (2016) Adjudicating paravalvular leaks of transcatheter aortic valves: a critical appraisal. Eur Heart J 37:2627–2644. https://doi.org/10.1093/eurheartj/ehw115
Aftab SM, Mohd Rafie AS, Razak NA, Ahmad KA (2016) Turbulence Model Selection for Low Reynolds Number Flows. PLoS ONE 11:e0153755. https://doi.org/10.1371/journal.pone.0153755
Bagur R et al (2012) Need for permanent pacemaker as a complication of transcatheter aortic valve implantation and surgical aortic valve replacement in elderly patients with severe aortic stenosis and similar baseline electrocardiographic findings. JACC Cardiovasc Interv 5:540–551. https://doi.org/10.1016/j.jcin.2012.03.004
Bailey J, Curzen N, Bressloff NW (2015) Assessing the impact of including leaflets in the simulation of TAVI deployment into a patient-specific aortic root. Comput Methods Biomech Biomed Eng 1:1–12. https://doi.org/10.1080/10255842.2015.1058928
Bailey J, Curzen N, Bressloff NW (2016) The impact of imperfect frame deployment and rotational orientation on stress within the prosthetic leaflets during transcatheter aortic valve implantation. J Biomech. https://doi.org/10.1016/j.jbiomech.2016.12.031
Barbanti M et al (2013) Anatomical and procedural features associated with aortic root rupture during balloon-expandable transcatheter aortic valve replacement. Circulation 128:244–253. https://doi.org/10.1161/circulationaha.113.002947
Bianchi M, Marom G, Ghosh RP, Fernandez HA, Taylor JR Jr, Slepian MJ, Bluestein D (2016) Effect of balloon-expandable transcatheter aortic valve replacement positioning: a patient-specific numerical model. Artif Organs 40:E292–e304. https://doi.org/10.1111/aor.12806
Bosi GM, Capelli C, Hong Cheang M, Delahunty N, Mullen M, Taylor AM, Schievano S (2018) Population-specific material properties of the implantation site for transcatheter aortic valve replacement finite element simulations. J Biomech. https://doi.org/10.1016/j.jbiomech.2018.02.017
Bosmans B, Famaey N, Verhoelst E, Bosmans J, Vander Sloten J (2016) A validated methodology for patient specific computational modeling of self-expandable transcatheter aortic valve implantation. J Biomech. https://doi.org/10.1016/j.jbiomech.2016.06.024
Capelli C et al (2012) Patient-specific simulations of transcatheter aortic valve stent implantation. Med Biol Eng Comput 50:183–192. https://doi.org/10.1007/s11517-012-0864-1
Chang J, Rong-Hui L, Sheng-Ping Z, Li-Zhen W, Yu-Bo F (2018) Effect of stent designs on the paravalvular regurgitation of transcatheter aortic valve implantation. Int J Comput Methods. https://doi.org/10.1142/s0219876218420070
Chiu W-C et al (2014) Thromboresistance comparison of the HeartMate II ventricular assist device with the device thrombogenicity emulation-optimized HeartAssist 5 VAD. J Biomech Eng 136:021014–021019. https://doi.org/10.1115/1.4026254
Chung W, Cho J, Belytschko T (1998) On the dynamic effects of explicit FEM in sheet metal forming analysis. Eng Comput 15:750–776
Couture EL, Lepage S, Masson J-B, Daneault B (2017) Very late transcatheter heart valve thrombosis. World J Cardiol 9:196–199. https://doi.org/10.4330/wjc.v9.i2.196
De Jaegere P et al (2016) Patient-specific computer modeling to predict aortic regurgitation after transcatheter aortic valve replacement. JACC Cardiovasc Interv 9:508–512. https://doi.org/10.1016/j.jcin.2016.01.003
Dvir D et al (2012) Multicenter evaluation of Edwards SAPIEN positioning during transcatheter aortic valve implantation with correlates for device movement during final deployment. JACC Cardiovasc Interv 5:563–570. https://doi.org/10.1016/j.jcin.2012.03.005
Eggebrecht H, Doss M, Schmermund A, Nowak B, Krissel J, Voigtländer T (2012) Interventional options for severe aortic regurgitation after transcatheter aortic valve implantation: balloons, snares, valve-in-valve. Clin Res Cardiol 101:503–507. https://doi.org/10.1007/s00392-012-0434-4
Genereux P et al (2013) Paravalvular leak after transcatheter aortic valve replacement: the new Achilles’ heel? A comprehensive review of the literature. J Am Coll Cardiol 61:1125–1136. https://doi.org/10.1016/j.jacc.2012.08.1039
Gilbert ON et al (2018) Comparison of paravalvular aortic leak characteristics in the Medtronic CoreValve versus Edwards Sapien Valve: Paravalvular aortic leak characteristics. Catheter Cardiovasc Interv. https://doi.org/10.1002/ccd.27643
Girdhar G et al (2012) Device thrombogenicity emulation: a novel method for optimizing mechanical circulatory support device thromboresistance. PLoS One 7:e32463
Haj-Ali R, Marom G, Ben Zekry S, Rosenfeld M, Raanani E (2012) A general three-dimensional parametric geometry of the native aortic valve and root for biomechanical modeling. J Biomech 45:2392–2397. https://doi.org/10.1016/j.jbiomech.2012.07.017
Hatoum H, Yousefi A, Lilly S, Maureira P, Crestanello J, Dasi LP (2018) An in-vitro evaluation of turbulence after transcatheter aortic valve implantation. J Thorac Cardiovasc Surg 1:1. https://doi.org/10.1016/j.jtcvs.2018.05.042
Kanwar A, Thaden JJ, Nkomo VT (2018) Management of patients with aortic valve stenosis. Mayo Clin Proc 93:488–508. https://doi.org/10.1016/j.mayocp.2018.01.020
Kodali SK et al (2012) Two-year outcomes after transcatheter or surgical aortic-valve replacement. N Engl J Med 366:1686–1695
Leon MB et al (2016) Transcatheter or surgical aortic-valve replacement in intermediate-risk patients. New Engl J Med. https://doi.org/10.1056/nejmoa1514616
Maisano F, Taramasso M, Nietlispach F (2015) Prognostic influence of paravalvular leak following TAVI: is aortic regurgitation an active incremental risk factor or just a mere indicator? Eur Heart J 36:413–415. https://doi.org/10.1093/eurheartj/ehu410
Mao W, Wang Q, Kodali S, Sun W (2018) Numerical parametric study of paravalvular leak following a transcatheter aortic valve deployment into a patient-specific aortic root. J Biomech Eng 140:101007. https://doi.org/10.1115/1.4040457
Marom G, Bluestein D (2016) Lagrangian methods for blood damage estimation in cardiovascular devices–How numerical implementation affects the results. Expert Rev Med Devices 13:113–122. https://doi.org/10.1586/17434440.2016.1133283
Martin C, Sun W (2012) Biomechanical characterization of aortic valve tissue in humans and common animal models. J Biomed Mater Res Part A 100:1591–1599. https://doi.org/10.1002/jbm.a.34099
Martin C, Sun W (2015) Comparison of transcatheter aortic valve and surgical bioprosthetic valve durability: a fatigue simulation study. J Biomech 48:3026–3034. https://doi.org/10.1016/j.jbiomech.2015.07.031
Martin C, Pham T, Sun W (2011) Significant differences in the material properties between aged human and porcine aortic tissues. Eur J Cardio-Thor Surg 40:28–34. https://doi.org/10.1016/j.ejcts.2010.08.056
McGee OM, Gunning PS, McNamara A, McNamara LM (2018) The impact of implantation depth of the Lotus™ valve on mechanical stress in close proximity to the bundle of His. Biomech Model Mechanobiol 1:1. https://doi.org/10.1007/s10237-018-1069-9
Mensel B et al (2014) MRI-based determination of reference values of thoracic aortic wall thickness in a general population. Eur Radiol 24:2038–2044. https://doi.org/10.1007/s00330-014-3188-8
Morganti S, Conti M, Aiello M, Valentini A, Mazzola A, Reali A, Auricchio F (2014) Simulation of transcatheter aortic valve implantation through patient-specific finite element analysis: two clinical cases. J Biomech 47:2547–2555. https://doi.org/10.1016/j.jbiomech.2014.06.007
Morganti S, Brambilla N, Petronio AS, Reali A, Bedogni F, Auricchio F (2016) Prediction of patient-specific post-operative outcomes of TAVI procedure: The impact of the positioning strategy on valve performance. J Biomech 49:2513–2519. https://doi.org/10.1016/j.jbiomech.2015.10.048
Mummert J, Sirois E, Sun W (2013) Quantification of biomechanical interaction of transcatheter aortic valve stent deployed in porcine and ovine hearts. Ann Biomed Eng 41:577–586. https://doi.org/10.1007/s10439-012-0694-1
Nombela-Franco L et al (2012) Predictive factors, efficacy, and safety of balloon post-dilation after transcatheter aortic valve implantation with a balloon-expandable valve. JACC Cardiovasc Interv 5:499–512. https://doi.org/10.1016/j.jcin.2012.02.010
Pappano A, Wier W (2013) Cardiovascular physiology. In: Mosby physiology monograph series. vol 6, 10th edn. Elsevier, Philadelphia, PA, p 304
Pasic M, Unbehaun A, Buz S, Drews T, Hetzer R (2015) Annular rupture during transcatheter aortic valve replacement: classification, pathophysiology, diagnostics, treatment approaches, and prevention. JACC Cardiovasc Interv 8:1–9. https://doi.org/10.1016/j.jcin.2014.07.020
Petronio AS et al (2015) Optimal implantation depth and adherence to guidelines on permanent pacing to improve the results of transcatheter aortic valve replacement with the medtronic CoreValve system. JACC Cardiovasc Interv 8:837–846. https://doi.org/10.1016/j.jcin.2015.02.005
Pibarot P, Hahn RT, Weissman NJ, Monaghan MJ (2015) Assessment of paravalvular regurgitation following TAVR: a proposal of unifying grading scheme. JACC Cardiovasc Imag 8:340–360. https://doi.org/10.1016/j.jcmg.2015.01.008
Pilgrim T, Windecker S (2015) Transcatheter aortic valve replacement: lessons gained from extreme-risk patients. J Am Coll Cardiol 66:1335–1338. https://doi.org/10.1016/j.jacc.2015.04.085
Ribeiro HB et al (2013) Coronary obstruction following transcatheter aortic valve implantation: a systematic review. JACC Cardiovasc Interv 6:452–461. https://doi.org/10.1016/j.jcin.2012.11.014
Schultz C et al (2016) Patient-specific image-based computer simulation for theprediction of valve morphology and calcium displacement after TAVI with the Medtronic CoreValve and the Edwards SAPIEN valve. EuroIntervention 11:1044–1052. https://doi.org/10.4244/eijv11i9a212
Scotten LN, Siegel R (2014) Thrombogenic potential of transcatheter aortic valve implantation with trivial paravalvular leakage. Ann Transl Med 2:43. https://doi.org/10.3978/j.issn.2305-5839.2014.05.04
Smith RG, Leon MB, Mack MJ, Miller CD (2011) Transcatheter versus surgical aortic-valve replacement in high-risk patients. New Engl J Med 364:2187–2198
Still S, Szerlip M, Mack M (2018) TAVR Vs. SAVR in intermediate-risk patients: what influences our choice of therapy. Curr Cardiol Rep 20:82. https://doi.org/10.1007/s11886-018-1026-3
Sturla F et al (2016) Impact of different aortic valve calcification patterns on the outcome of Transcatheter Aortic Valve Implantation: a finite element study. Journal of biomechanics 1:1. https://doi.org/10.1016/j.jbiomech.2016.03.036
Takagi K et al (2011) Predictors of moderate-to-severe paravalvular aortic regurgitation immediately after CoreValve implantation and the impact of postdilatation. Catheter Cardiovasc Interve 78:432–443. https://doi.org/10.1002/ccd.23003
Tzamtzis S, Viquerat J, Yap J, Mullen MJ, Burriesci G (2013) Numerical analysis of the radial force produced by the Medtronic-CoreValve and Edwards-SAPIEN after transcatheter aortic valve implantation (TAVI). Med Eng Phys 35:125–130. https://doi.org/10.1016/j.medengphy.2012.04.009
Vahidkhah K, Azadani AN (2017) Supra-annular Valve-in-Valve implantation reduces blood stasis on the transcatheter aortic valve leaflets. J Biomech 58:114–122. https://doi.org/10.1016/j.jbiomech.2017.04.020
Van der Boon RM et al (2012) New conduction abnormalities after TAVI—frequency and causes. Nat Rev Cardiol 9:454–463
Wang Q, Kodali S, Primiano C, Sun W (2014) Simulations of transcatheter aortic valve implantation: implications for aortic root rupture. Biomech Model Mechanobiol 14:29–38. https://doi.org/10.1007/s10237-014-0583-7
Webb JG et al (2009) Transcatheter aortic valve implantation—impact on clinical and valve-related outcomes. Circ J 119:3009–3016. https://doi.org/10.1161/circulationaha.108.837807
Xenos M, Girdhar G, Alemu Y, Jesty J, Slepian MJ, Einav S, Bluestein D (2010) Device Thrombogenicity Emulator (DTE)—design optimization methodology for cardiovascular devices: a study in two bileaflet MHV designs. J Biomech 43:2400–2409
Yushkevich PA, Piven J, Hazlett HC, Smith RG, Ho S, Gee JC, Gerig G (2006) User-guided 3D active contour segmentation of anatomical structures: significantly improved efficiency and reliability. NeuroImage 31:1116–1128. https://doi.org/10.1016/j.neuroimage.2006.01.015
Acknowledgement
This work was financially supported by NIH-NIBIB (1U01EB026414-01, DB) and by a NIH-NIBIB Quantum award Phase II-C (1U01EB012487-0, DB). This work was supported by computing resources from the SeaWulf cluster at Stony Brook University. ANSYS Fluent was provided by an ANSYS Academic Partnership with Stony Brook University.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10237_2018_1094_MOESM1_ESM.tif
Figure S1: Schematic of the steps of the fluid domain extraction process and the setup of the flow analyses (TIFF 118 kb)
Rights and permissions
About this article
Cite this article
Bianchi, M., Marom, G., Ghosh, R.P. et al. Patient-specific simulation of transcatheter aortic valve replacement: impact of deployment options on paravalvular leakage. Biomech Model Mechanobiol 18, 435–451 (2019). https://doi.org/10.1007/s10237-018-1094-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-018-1094-8